|
 |
- Search
| Clin Exp Reprod Med > Volume 44(2); 2017 > Article |
Abstract
Objective
Hyperstimulation methods are broadly used for in vitro fertilization (IVF) in patients with infertility; however, the side effects associated with these therapies, such as ovarian hyperstimulation syndrome (OHSS), have not been well studied. N-glycoproteomes are subproteomes used for the remote sensing of ovarian stimulation in follicular growth. Glycoproteomic variation in human follicular fluid (hFF) has not been evaluated. In this study, we aimed to identify and quantify the glycoproteomes and N-glycoproteins (N-GPs) in natural and stimulated hFF using label-free nano-liquid chromatography/electrospray ionization-quad time-of-flight mass spectrometry.
Methods
For profiling of the total proteome and glycoproteome, pooled protein samples from natural and stimulated hFF samples were selectively isolated using hydrazide chemistry to obtain the total proteomes and glycoproteomes. N-GPs were validated by the consensus sequence N-X-S/T (92.2% specificity for the N-glycomotif at p<0.05). All data were compared between natural versus hyperstimulated hFF samples.
Results
We detected 41 and 44 N-GPs in the natural and stimulated hFF samples, respectively. Importantly, we identified 11 N-GPs with greater than two-fold upregulation in stimulated hFF samples compared to natural hFF samples. We also validated the novel N-GPs thyroxine-binding globulin, vitamin D-binding protein, and complement proteins C3 and C9.
Human infertility problems can often be overcome through in vitro fertilization (IVF) methods and artificial reproductive technologies (ARTs), which have been used since 1978 [1]. Hyperstimulation methods increase the retrieval of mature oocytes and improve IVF success rates; indeed, in the United States, the pregnancy rate of patients undergoing IVF has increased to 37.4% [2]. However, hyperstimulation via gonadotropin injection causes deterioration of the quality and maturity of oocytes in some patients, including patients with polycystic ovary syndrome (PCOS) and unexplained poor responders. Furthermore, in some patients, hyperstimulation increases the risk of ovarian hyperstimulation syndrome (OHSS), which is associated with multiple growing follicles responding to follicle-stimulating hormone (FSH) hyperstimulation. PCOS is characterized by hyperandrogenism, anovulation, and delayed growth in numerous small antral follicles without the formation of mature follicles that proceed to ovulation. After human chorionic gonadotrophin (hCG) administration, multiple follicular development increases, and some patients exhibit OHSS [3,4]. The mechanisms underlying the increased sensitivity of patients with PCOS and OHSS to exogenous gonadotropins have not been clearly elucidated.
The maturation and quality of oocytes affect fertilization and embryo development in relation to repeated IVF failure and miscarriage. Genomic approaches have been used to analyze the maturation and dysfunction of oocytes. Recently, the development of proteomic analysis techniques has facilitated many new studies in biomedical research. The protein compositions of follicular fluid [5,6,7,8,9,10], fallopian fluid [9,11], granulosa cells [12,13], uterine tissues [14,15], and male reproductive fluid [16,17,18] have been reported. Follicular fluid contains various factors that mediate the growth of follicles and oocytes, and is therefore associated with follicle growth [2,7,19].
Spitzer et al. [20] compared the complex protein patterns of human follicular fluid (hFF) in mature and immature human follicles by two-dimensional electrophoresis; subsequently, several mass spectrometry (MS) techniques, including surface-enhanced laser desorption/ionization time-of-flight (TOF) MS and nano-liquid chromatography (LC) matrix-assisted laser desorption/ionization TOF/TOF MS [21,22,23], have shown significantly increased numbers of proteins that were not previously reported in hFF. The majority of proteins identified have been plasma-matched proteins, primarily represented by acute-phase proteins and some low-copy proteins, including sex hormone-binding globulin and inhibin A [6,24]. However, those studies identified only variations in the protein composition of hFF samples and plasma collected from patients participating in IVF programs.
During translation, proteins undergo maturation processes, such as folding, bonding, and glycosylation. Glycosylation is an important enzymatic process that links saccharides to produce glycan molecules, which are attached to biological protein molecules, such as proteins, lipids, or other organic molecules. During or after translation, proteins are subject to N- and O-linked glycosylation, resulting in alterations in their functions and activities; for example, glycosylation has been shown to affect proteins involved in cell-cell adhesion, cell migration, protease protection, signaling, and protein structure [12,13,25,26]. These physiological events are pivotal in folliculogenesis and ovulation, and therefore female reproduction, in vertebrates. Furthermore, studies have reported that alterations in glycoproteins can be related to the acrosome reaction, fertility, preterm labor, and immune responses [27,28,29,30,31]. Therefore, the application of powerful glycoprotein profiling approaches, specifically N-glycoproteome analysis, in reproductive medical research may significantly contribute to a comprehensive understanding of OHSS and PCOS.
Despite the important roles of glycoproteins, few studies have performed glycoprotein profiling in the female reproductive system. Moreover, limited information is available concerning the characteristics and functions of the identified total proteins, which may include glycoproteins, in natural and stimulated hFF samples [8,9,10,23,32,33]. Additionally, hFF glycosylation in stimulated and natural cycles has not been assessed. Furthermore, among the glycoproteins found in hFF, the N-glycoproteins (N-GPs) in hFF have yet to be identified and their potential physiological functions have yet to be studied. Accordingly, in this study, we performed a glycoproteome analysis of natural and stimulated hFF samples using nano-LC/electrospray ionization-quad-TOF (Q-TOF) MS and by isolating and enriching glycoprotein digests using hydrazide chemistry, which is highly efficient for enriching N-linked glycoproteins [34]. The identified glycoproteins and N-GPs were subjected to pathway analysis to investigate the enriched signal pathways in each sample.
hFF samples from 17 women (aged 24–38 years) were obtained from the Fertility Center of Eulji Medical Center, Seoul, Republic of Korea under IRB EMC 2015-02-006. No differences in the general population characteristics were identified except for estradiol levels on the day of hCG administration. The patient groups had similar past obstetric histories in past pregnancies. (p>0.99). The primary infertility diagnoses were male factor or tubal infertility. Samples collected during natural cycles (n=4) were obtained from patients with tubal infertility, normal ovaries on ultrasound, and regular menstrual cycles, as confirmed by endometrial biopsy during the previous cycle. For the stimulated cycles (n=13), a standard IVF treatment protocol was applied, including controlled ovarian FSH hyperstimulation using a gonadotropin-releasing hormone antagonist with hCG administration to induce follicular/egg maturation. Oocyte transvaginal ultrasound retrieval was performed 36 hours after hCG administration. The patient groups had similar past obstetric histories (all pregnancies, p>0.99), with no significant differences in their medical histories regarding diabetes, eating disorders, depression, cancer, hypertension, hyperthyroidism, or uterine fibroids. The patient characteristics are shown in Table 1 and Supplement 1.
Each hFF sample was obtained via puncture of the dominant ovarian follicles (14–22 mm in diameter) into individual tubes, and macroscopically clear fluids without erythrocytes were collected. Following oocyte isolation, the hFF was centrifuged to remove cellular components and debris. Collected samples were transferred to sterile polypropylene tubes and frozen at
−70℃ for further analysis.
The sample preparation procedure is summarized in Figure 1. Prior to the analysis of hFF proteomics, six high-abundance proteins in the hFF samples were depleted using a multiple affinity removal (MARS) column (human 6, 4.6×50 mm; Agilent Technologies, Santa Clara, CA, USA) for enhancing the detection sensitivity of proteomic profiling in the hFF samples [35]. The proteins in pools of flow-through low-abundance fractions for each sample were collected and then loaded onto the column. The depleted sample solutions were concentrated using an Amicon membrane filter (Ultra-4 10000 NMWL; Millipore, Billerica, MA, USA) by centrifugation at 4,000× g for 16 minutes. After concentration, 8 M urea with 0.1 M Tris (pH 7.9) was added, and samples were centrifuged at 4,000× g for 16 minutes. Finally, the supernatants were collected for further analysis.
The concentrations of proteins from the MARS column were measured using a Quant kit (GE Healthcare, Piscataway, NJ, USA) with standard controls (bovine serum albumin: 0, 1, 2, 4, 8, and 16 µg). Protein samples (1 mg) were treated with 5 mM Tris (2-carboxyethyl) phosphine (TCEP; Pierce, Rockford, IL, USA) and then incubated at 37℃ at 300 rpm for 30 minutes for reduction. Next, 15 mM iodoacetamide (Sigma-Aldrich, St. Louis, MO, USA) was added to each sample, and samples were incubated at room temperature at 300 rpm for 1 hour in the dark. Following trypsinization (Promega, Madison, WI, USA) at a ratio of 1:20, samples were incubated overnight at 37℃ at 300 rpm. In order to clean up the samples, peptide mixtures were loaded and flushed on a C18 cartridge (Waters, Milford, MA, USA), and the peptides were then eluted with 80% acetonitrile/0.1% formic acid solution. The peptides were then completely dried using a centrifugal concentrator (Scanvac, Lynge, Denmark), and the dried peptides were dissolved in 20 µL of 0.1% formic acid for MS analysis.
An OFFGEL fractionator with a 12-well setup (3100 OFFGEL Low Res kit, pH 3–10; Agilent Technologies) was used according to the protocol for isoelectric point-based fractionation. The peptide samples were diluted in distilled water containing 1% ampholytes, and 0.15 mL of the diluted sample was loaded in each well. The default method used for the fractionation was as follows: limits of ‘focusing’ phase: 8,000 V, 50 µA, 200 mW, and 100 hours; limits of ‘hold’ phase: 500 V, 20 µA, and 50 mW. The temperature was set to 20℃.
A high-performance liquid chromatography (HPLC)-chip/Q-TOF system (Agilent Technologies) was used for sample analysis. The system consisted of an Agilent 1200 series nano-LC system and an Agilent 6520 Q-TOF coupled with a chip cube interface. The HPLC chip comprised a 160-nL enrichment column and a 75 µm (diameter) ×150 mm separation column packed with Zorbax 300SB-C18 (5 µm; Agilent Technologies, Santa Clara, CA, USA). The sample-loading capillary pump administered the solvent of 97% buffer A (water with 0.1% formic acid) and 3% buffer B (90% acetonitrile/0.1% formic acid) at a flow rate of 4 µL/min. A nano-pump was used to generate a separation-driven solvent composition gradient of 10% to 45% by pumping buffer B for 0 to 15 minutes. The buffer B percentage was then changed to 90% for 15 to 20 minutes and back to 10% for 20 to 30 minutes. The Q-TOF mass spectrometer was set to positive ionization mode. The drying gas (nitrogen gas) flow was set at 5 L/min and 300℃. Eluting peptides were selected for collision-induced dissociation during alternative procedures for MS scanning over the m/z range of 300 to 2,400 at a rate of 4 spectra/s and MS/MS scanning over the range of 100 to 3,000 m/z at 3 spectra/s. The isolation window was 4 m/z.
Protein samples (1 mg each, three replicates) were digested into peptides by trypsinization. Hydrazide resin (Affi-Prep beads; Bio-Rad, Hercules, CA, USA) was used to conduct glycocapturing as previously described [36]. Validation of the identified N-GPs was carried out based on the consensus sequence N-X-S/T (Spectrum Mill, Agilent Technologies), a unique acceptor sequence conserved in N-glycosylation. Prepared samples were analyzed using an HPLC-chip/Q-TOF system as previously described.
To identify peptides, all tandem mass spectra were analyzed using Spectrum Mill (Agilent Technologies) with the Swiss-Prot database. For the database search, the enzyme was set to trypsin with two missed cleavages, and mass tolerance was limited to 20 and 50 ppm for precursor and product ions, respectively. The modification parameters included static modification for cysteine carbamidomethylation, a dynamic modification for methionine oxidation, and N-terminal carbamylation. Deamidation of asparagine was included for glycopeptide identification. MassHunter Mass Profiler Professional software (Agilent Technologies) was used for label-free quantification by investigating similarities and differences in features across multiple samples. MetaCore ver. 6.4 (GeneGo, St. Joseph, MI, USA) was used to visualize and analyze molecular and protein interaction networks. The significance of biological pathways was evaluated through multiple testing using Benjamini-Hochberg false discovery rate analysis.
The pooled hFF samples were depleted using a MARS column, and protein concentrations were determined using Bradford protein assays (Bio-Rad). An equal amount of protein (50 µg) for each hFF was loaded onto 4% to 12% NuPAGE bis-Tris precast gels (Invitrogen, Carlsbad, CA, USA). Proteins were transferred to polyvinylidene difluoride membranes (Bio-Rad), and membranes were incubated with primary antibodies. All primary antibodies, including anti-thyroxine-binding globulin (TBG), anti-vitamin D-binding protein (VDBP), anticomplement C3, anticomplement C9, anti-prothrombin, and anti-actin, as well as secondary horseradish peroxidase-conjugated mouse immunoglobulin G (IgG) and horseradish peroxidase-conjugated rabbit IgG, were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA)
The summary statistics were expressed as means±standard errors. Data were processed with PRISM 5 (GraphPad Software, San Diego, CA, USA). The Mann-Whitney test was used for comparisons between the natural and stimulated groups. Differences with p-values of less than 0.05 were considered significant.
The total proteome analysis identified 126 and 167 unique proteins in the natural and stimulated hFF samples, respectively (Figure 2A, Supplements 2 and 3). A summary of differentially expressed total proteins between the natural and stimulated hFF samples of patients undergoing IVF is presented in Supplement 4. Among the 10 pathway maps identified as statistically significant, eight were more strongly expressed in the stimulated hFF samples than in the natural hFF samples, including immune response, blood coagulation, protein folding, and cell adhesion (Figure 2B). However, high-density lipoprotein transport processes and interleukin-6-induced acute-phase responses were more strongly expressed in the natural hFF samples.
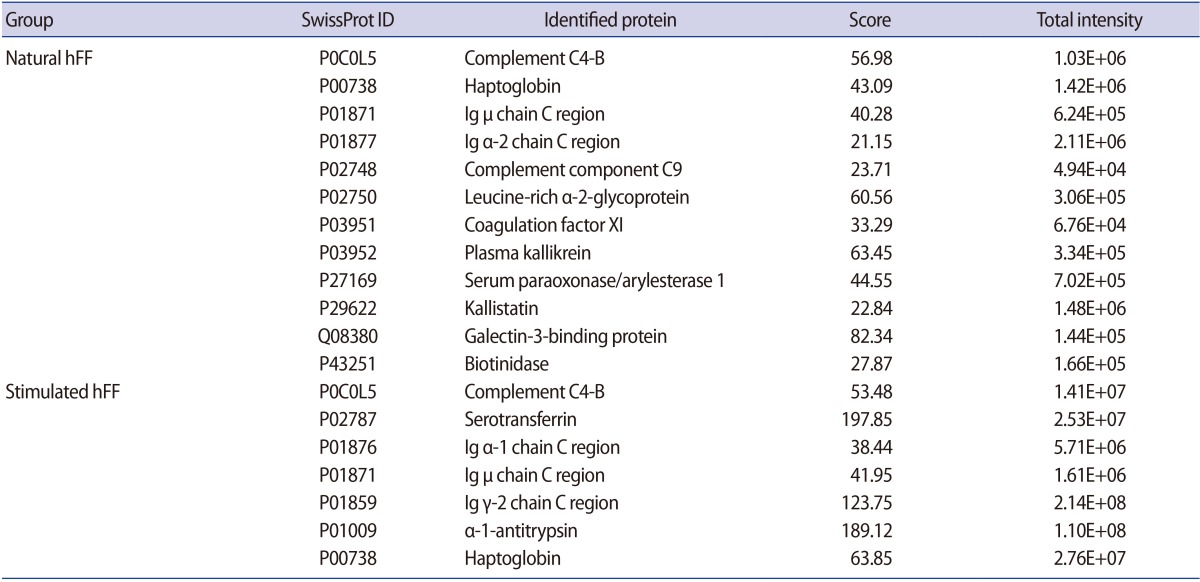
A total of 41 and 44 (approximately 32.5% and 26.5% of total proteins, respectively) hFF N-GPs were identified in the natural and stimulated hFF samples, respectively. The specificity for the N-glycomotif was 92.2%. The validated raw results of the consensus sequence N-X-S/T are shown in Supplement 5, and the list of identified glycocaptured peptides is given in Supplements 6 and 7, and summarized in Supplement 8. Twelve proteins (complement C4-B, haptoglobin, Ig µ chain C region, Ig α-2 chain C region, complement component C9, leucine-rich α-2-glycoprotein, coagulation factor 11, plasma kallikrein, serum paraoxonase/arylesterase 1, kallistatin, galectin-3-binding protein, and biotinidase) were identified only in the natural hFF N-GPs (Table 2, Figure 3A). Additionally, seven proteins (complement C4-B, serotransferrin, Ig α-1 chain C region, Ig µ chain C region, Ig γ-2 chain C region, α-1-antitrypsin, and haptoglobin) were identified only in the stimulated hFF N-GPs (Table 2, Figure 3B).
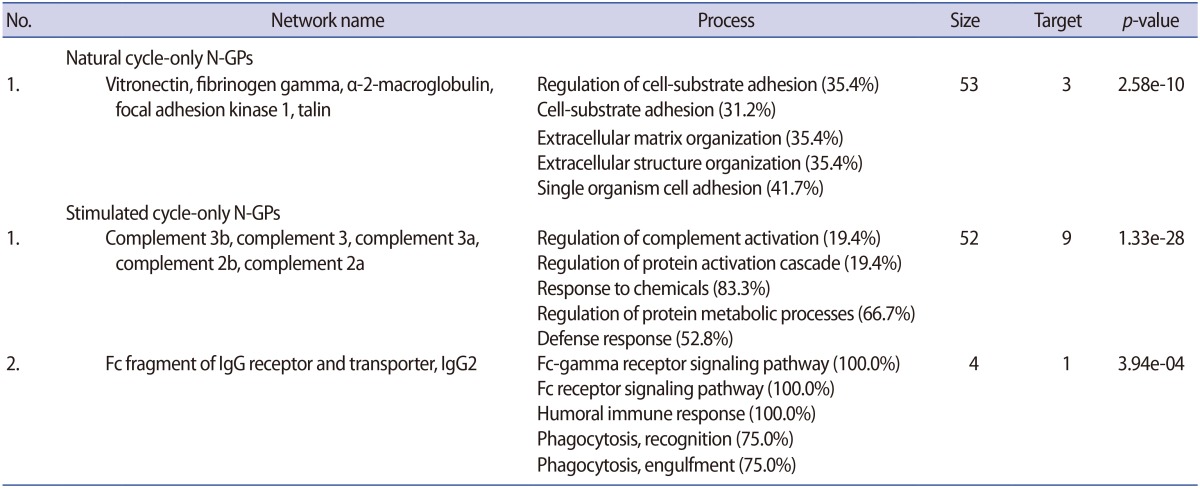
All statistically analyzed immune-related pathway maps, except the interleukin-6-induced acute-phase response by GeneGo, showed increased N-GP levels in the stimulated hFF samples (Figure 3D). However, protein folding- or cell adhesion-related N-GPs were more extensively expressed in the natural hFF samples (Figure 3D). Thirty-five N-GPs were commonly detected in both natural and stimulated hFF samples, while 6 and 9 N-GPs were identified only in the natural and stimulated hFF groups, respectively (Table 3, Figure 3C). The 3 N-GPs identified only in the natural hFF group, including α-2-macroglobulin, vitronectin, and fibrinogen γ, were related to cell adhesion and extracellular matrix structuring/organization (Table 4). In contrast, 10 N-GPs in the stimulated hFF samples were related to complement processes or immune reactions (Table 4). An N-GP process network analysis of the natural and stimulated hFF samples showed different functional changes in proteins involved in the immune response.
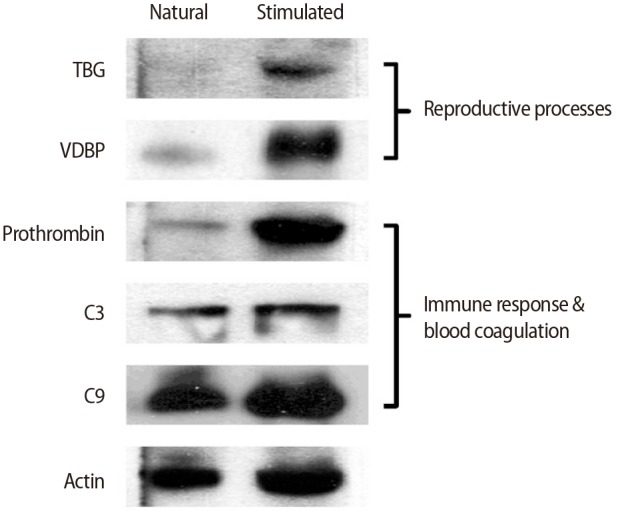
To confirm the differential expression of N-GPs, TBG and VDBP, which are known to be important in reproduction, were validated by western blotting and found to be significantly upregulated in the stimulated hFF samples compared with the natural hFF samples (Figure 4). Proteins related to the complement and coagulation pathways, including C3, C9, and prothrombin, were also validated by western blotting. The stimulated hFF samples showed increased expression of C3, C9, and prothrombin, consistent with our N-GP network (Figure 4).
The pregnancy rate of patients who undergo IVF is related to the patient's response to controlled ovarian hyperstimulation with an exogenous gonadotropin, and the composition of the hFF as a microenvironment for oocyte development and maturation [37]. Therefore, the composition of hFF is associated with the stage of oocyte development and the degree of follicle maturation [38]. However, the factors involved in follicular growth are largely unknown. Thus, investigating hFF content and function may provide insights into the physiological processes controlling follicular development and lead to increased ART success rates.
Previous studies have reported that the N-GP has important roles in cell migration, angiogenesis, and cell-cell adhesion [39]. Moreover, our data suggest that glycoprotein capturing may be a useful technique for understanding the differences in human folliculogenesis and oocyte maturation and for identifying methods to prevent OHSS or other side effects of hyperstimulation. These newly detected N-GPs perform pivotal physiological roles in the regulation of oocyte maturation and ovulation during folliculogenesis via various pathways, including the regulation of fibrinolysis, blood coagulation, cholesterol homeostasis, inflammation, and complement and antioxidant pathways. By glycocapturing, we found two interesting glycoproteins, TBG and VDBP, in the stimulated hFF group. TBG binds thyroid hormone in circulation and is one of the three proteins (along with transthyretin and albumin) responsible for carrying the thyroid hormones thyroxine (T4) and 3,5,3´-triiodothyronine (T3) in the bloodstream [40]. TBG production can be modified by steroid regulators, such as estrogen and corticosteroid [40,41,42]. In the late follicular phase, high levels of estrogen have stimulatory effects on the hypothalamus, leading it to release gonadotropin-releasing hormone, which in turn stimulates the expression of pituitary luteinizing hormone and FSH and stimulates late oocyte maturation [43]. Evaluation of the TBG concentration in hFF could be an alternative diagnostic tool for monitoring follicular growth and oocyte maturation during ART programs for ovarian disorders. VDBP belongs to the albumin gene family, together with human serum albumin and α-fetoprotein. VDBP is a multifunctional protein found in plasma, ascitic fluid, cerebrospinal fluid, and the surface of many cell types. Additionally, VDBP binds to vitamin D and its plasma metabolites and transports them to target tissues [44]. VDBP is known to be present in the human ovary and is thought to have an important role in the local regulation of ovulation and follicular growth [8,45].
Interestingly, we found that the expression of several complement proteins increased in stimulated hFF. Complement component C9 in stimulated hFF has been reported to have a vital role in follicular angiogenesis [6,46,47]. The levels of complement activity, including complement components C3, C4, and C9, in the hFF samples are associated with higher levels of vascular endothelial growth factor (VEGF) levels than are present in serum samples of women undergoing IVF. Thus, complement components are fundamentally involved in perifollicular angiogenesis and may significantly affect oocyte maturation and quality [48,49,50]. Previous reports have shown that VEGF expression is increased in patients with PCOS and is related to OHSS [51,52,53]. Moreover, several complement proteins and complement factors are related to VEGF and angiogenesis [54,55,56]. In this study, we detected and identified complement C3, C4A, and C7 in both groups of hFF samples. Interestingly, hFF N-GP analysis detected C4-B and C9 in both hFF groups, whereas C2 and C3 were only detected and identified in stimulated hFF samples.
Evidence has shown that inflammation and immune responses are highly activated in patients following abnormal hyperstimulation [57]. Consistent with this, our data showed that the upregulated N-GPs in the stimulated hFF samples were related to inflammation (haptoglobin and α-1-antitrypsin) and immune responses (Ig α-2 chain C region and Ig γ-1 chain C region). Inflammatory reactions have been reported to be associated with ovulation [58]. Additionally, the identified N-GPs in the stimulated hFF (prothrombin, hemopexin, galectin-3-binding protein, β-2-glycoprotein 1, inter-α-trypsin inhibitor, zinc-α-2-glycoprotein, and complement component C9) are involved in coagulation, cell-cell adhesion, and angiogenesis [24,59].
In summary, this is the first report describing the comparative N-GP profiling of natural and stimulated hFF samples from hyperstimulated ovaries in patients treated under an IVF program. Based on our results, we identified the N-GPs TBG, VDBP, C9, and prothrombin as useful for monitoring patients to ensure that they achieve successful hyperstimulation during IVF and to help prevent the side effects of IVF. Measurement of the changes in glycoproteins in hFF may provide prognostic value for a wide range of fertility problems and may therefore improve our understanding of oocyte quality and female reproduction. However, we were only able to obtain four natural hFF samples for this study because of problems with sampling. Accordingly, additional hFF samples are needed for further validation and elucidation of the relationship between oocytes and hFF components during IVF for the prevention of OHSS or other diseases.
This study is a novel approach, in which the N-GP was analyzed as a component of the functional protein analysis of hFF, but it has limitations in many areas. First, only a limited number of samples were used in the experiment due to the difficulty of sampling the natural cycle group. It was therefore difficult to find verified indicators and obtain a statistically verifiable result. For this reason, we are currently working on preparing a larger sample, taking into account age, hyperstimulation method, oocyte maturation, and pregnancy. This will help to validate the results of this study and ensure the objectivity of future studies. In addition, we will assess changes in the functional proteins in cases of OHSS induced by controlled ovarian hyperstimulation through the analysis of OHSS patients' hFF, although such samples were excluded from this study.
Notes
Supplementary materials
Supplement 1
Clinical information. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s001.pdf.
Supplement 2
List of the total proteomes identified in the natural hFF. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s002.pdf.
Supplement 3
List of the total proteomes identified in the stimulated hFF. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s003.pdf.
Supplement 4
Differentially expressed total proteins in natural and stimulated hFF samples. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s004.pdf.
Supplement 5
Eleven target peptide spectrum N-GPs in the stimulated hFF. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s005.pdf.
Supplement 6
List of the N-Glycopeptides identified in the natural hFF. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s006.pdf.
Supplement 7
List of the N-Glycopeptides identified in the stimulated hFF. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s007.pdf.
Supplement 8
N-GP list of natural and stimulated hFFs. Supplemental data can be found at: http://ecerm.org/src/sm/cerm-44-63-s008.pdf.
References
1. Nelson SM, Lawlor DA. Predicting live birth, preterm delivery, and low birth weight in infants born from in vitro fertilisation: a prospective study of 144,018 treatment cycles. PLoS Med 2011;8:e1000386PMID: 21245905.



2. Dumesic DA, Meldrum DR, Katz-Jaffe MG, Krisher RL, Schoolcraft WB. Oocyte environment: follicular fluid and cumulus cells are critical for oocyte health. Fertil Steril 2015;103:303-316.PMID: 25497448.


3. Schenker JG, Weinstein D. Ovarian hyperstimulation syndrome: a current survey. Fertil Steril 1978;30:255-268.PMID: 361440.


4. MacDougall MJ, Tan SL, Balen A, Jacobs HS. A controlled study comparing patients with and without polycystic ovaries undergoing in-vitro fertilization. Hum Reprod 1993;8:233-237.PMID: 8473426.



5. Angelucci S, Ciavardelli D, Di Giuseppe F, Eleuterio E, Sulpizio M, Tiboni GM, et al. Proteome analysis of human follicular fluid. Biochim Biophys Acta 2006;1764:1775-1785.PMID: 17067859.


6. Atiomo W, Khalid S, Parameshweran S, Houda M, Layfield R. Proteomic biomarkers for the diagnosis and risk stratification of polycystic ovary syndrome: a systematic review. BJOG 2009;116:137-143.PMID: 19076945.


7. Ducolomb Y, Gonzalez-Marquez H, Fierro R, Jimenez I, Casas E, Flores D, et al. Effect of porcine follicular fluid proteins and peptides on oocyte maturation and their subsequent effect on in vitro fertilization. Theriogenology 2013;79:896-904.PMID: 23453254.


8. Estes SJ, Ye B, Qiu W, Cramer D, Hornstein MD, Missmer SA. A proteomic analysis of IVF follicular fluid in women <or=32 years old. Fertil Steril 2009;92:1569-1578.PMID: 18980758.


9. Ferrero S, Gillott DJ, Remorgida V, Anserini P, Ragni N, Grudzinskas JG. Proteomic analysis of peritoneal fluid in fertile and infertile women with endometriosis. J Reprod Med 2009;54:32-40.PMID: 19263878.

10. Jarkovska K, Martinkova J, Liskova L, Halada P, Moos J, Rezabek K, et al. Proteome mining of human follicular fluid reveals a crucial role of complement cascade and key biological pathways in women undergoing in vitro fertilization. J Proteome Res 2010;9:1289-1301.PMID: 20058866.


11. Georgiou AS, Sostaric E, Wong CH, Snijders AP, Wright PC, Moore HD, et al. Gametes alter the oviductal secretory proteome. Mol Cell Proteomics 2005;4:1785-1796.PMID: 16105986.


12. Curry TE Jr, Dean DD, Sanders SL, Pedigo NG, Jones PB. The role of ovarian proteases and their inhibitors in ovulation. Steroids 1989;54:501-521.PMID: 2559499.


13. Yokota K, Hirano T, Urata N, Yamauchi N, Hattori MA. Upregulation of P-glycoprotein activity in porcine oocytes and granulosa cells during in vitro maturation. J Reprod Dev 2011;57:322-326.PMID: 21289467.


14. Clark GF. Functional glycosylation in the human and mammalian uterus. Fertil Res Pract 2015;1:17PMID: 28620522.



15. Daikoku T, Tranguch S, Friedman DB, Das SK, Smith DF, Dey SK. Proteomic analysis identifies immunophilin FK506 binding protein 4 (FKBP52) as a downstream target of Hoxa10 in the periimplantation mouse uterus. Mol Endocrinol 2005;19:683-697.PMID: 15528267.



16. Aitken RJ, Nixon B, Lin M, Koppers AJ, Lee YH, Baker MA. Proteomic changes in mammalian spermatozoa during epididymal maturation. Asian J Androl 2007;9:554-564.PMID: 17589795.


17. Peddinti D, Nanduri B, Kaya A, Feugang JM, Burgess SC, Memili E. Comprehensive proteomic analysis of bovine spermatozoa of varying fertility rates and identification of biomarkers associated with fertility. BMC Syst Biol 2008;2:19PMID: 18294385.



18. Yoon SJ, Rahman MS, Kwon WS, Ryu DY, Park YJ, Pang MG. Proteomic identification of cryostress in epididymal spermatozoa. J Anim Sci Biotechnol 2016;7:67PMID: 27895910.




19. Bedaiwy M, Shahin AY, AbulHassan AM, Goldberg JM, Sharma RK, Agarwal A, et al. Differential expression of follicular fluid cytokines: relationship to subsequent pregnancy in IVF cycles. Reprod Biomed Online 2007;15:321-325.PMID: 17854532.


20. Spitzer D, Murach KF, Lottspeich F, Staudach A, Illmensee K. Different protein patterns derived from follicular fluid of mature and immature human follicles. Hum Reprod 1996;11:798-807.PMID: 8671331.



21. Anderson NL, Anderson NG. Proteome and proteomics: new technologies, new concepts, and new words. Electrophoresis 1998;19:1853-1861.PMID: 9740045.


22. Anderson NL, Anderson NG. The human plasma proteome: history, character, and diagnostic prospects. Mol Cell Proteomics 2002;1:845-867.PMID: 12488461.


23. Hanrieder J, Nyakas A, Naessen T, Bergquist J. Proteomic analysis of human follicular fluid using an alternative bottom-up approach. J Proteome Res 2008;7:443-449.PMID: 18047273.


24. Lee HC, Lee SW, Lee KW, Lee SW, Cha KY, Kim KH, et al. Identification of new proteins in follicular fluid from mature human follicles by direct sample rehydration method of two-dimensional polyacrylamide gel electrophoresis. J Korean Med Sci 2005;20:456-460.PMID: 15953869.



25. Hughes SC, Mason HD, Franks S, Holly JM. The insulin-like growth factors (IGFs) in follicular fluid are predominantly bound in the ternary complex. J Endocrinol 1997;155:R1-R4.PMID: 9488005.


26. Wujek P, Kida E, Walus M, Wisniewski KE, Golabek AA. N-glycosylation is crucial for folding, trafficking, and stability of human tripeptidyl-peptidase I. J Biol Chem 2004;279:12827-12839.PMID: 14702339.


27. Schweigert FJ, Gericke B, Wolfram W, Kaisers U, Dudenhausen JW. Peptide and protein profiles in serum and follicular fluid of women undergoing IVF. Hum Reprod 2006;21:2960-2968.PMID: 16893915.



28. Ceciliani F, Giordano A, Spagnolo V. The systemic reaction during inflammation: the acute-phase proteins. Protein Pept Lett 2002;9:211-223.PMID: 12144517.


29. de Boer JP, Creasey AA, Chang A, Abbink JJ, Roem D, Eerenberg AJ, et al. Alpha-2-macroglobulin functions as an inhibitor of fibrinolytic, clotting, and neutrophilic proteinases in sepsis: studies using a baboon model. Infect Immun 1993;61:5035-5043.PMID: 7693593.



30. Tse JY, Chiu PC, Lee KF, Seppala M, Koistinen H, Koistinen R, et al. The synthesis and fate of glycodelin in human ovary during folliculogenesis. Mol Hum Reprod 2002;8:142-148.PMID: 11818517.



31. Das T, Chattopadhyay R, Ghosh S, Goswami S, Chattopadhyay D, Chakravarty B, et al. Role of an estrogen-upregulated 64.0-kDa uterine fluid glycoprotein in improving fertility in women. Fertil Steril 2007;87:343-350.PMID: 17097647.


32. Dai G, Lu G. Different protein expression patterns associated with polycystic ovary syndrome in human follicular fluid during controlled ovarian hyperstimulation. Reprod Fertil Dev 2012;24:893-904.PMID: 22935150.


33. Kim YS, Kim MS, Lee SH, Choi BC, Lim JM, Cha KY, et al. Proteomic analysis of recurrent spontaneous abortion: identification of an inadequately expressed set of proteins in human follicular fluid. Proteomics 2006;6:3445-3454.PMID: 16637005.


34. Wollscheid B, Bausch-Fluck D, Henderson C, O’Brien R, Bibel M, Schiess R, et al. Mass-spectrometric identification and relative quantification of N-linked cell surface glycoproteins. Nat Biotechnol 2009;27:378-386.PMID: 19349973.




35. Echan LA, Tang HY, Ali-Khan N, Lee K, Speicher DW. Depletion of multiple high-abundance proteins improves protein profiling capacities of human serum and plasma. Proteomics 2005;5:3292-3303.PMID: 16052620.


36. Larsen MR, Jensen SS, Jakobsen LA, Heegaard NH. Exploring the sialiome using titanium dioxide chromatography and mass spectrometry. Mol Cell Proteomics 2007;6:1778-1787.PMID: 17623646.


37. Fortune JE. Ovarian follicular growth and development in mammals. Biol Reprod 1994;50:225-232.PMID: 8142540.


38. Nayudu PL, Lopata A, Jones GM, Gook DA, Bourne HM, Sheather SJ, et al. An analysis of human oocytes and follicles from stimulated cycles: oocyte morphology and associated follicular fluid characteristics. Hum Reprod 1989;4:558-567.PMID: 2794015.



39. Kuo MW, Wang CH, Wu HC, Chang SJ, Chuang YJ. Soluble THSD7A is an N-glycoprotein that promotes endothelial cell migration and tube formation in angiogenesis. PLoS One 2011;6:e29000PMID: 22194972.



40. Tahboub R, Arafah BM. Sex steroids and the thyroid. Best Pract Res Clin Endocrinol Metab 2009;23:769-780.PMID: 19942152.


41. Poppe K, Velkeniers B, Glinoer D. The role of thyroid autoimmunity in fertility and pregnancy. Nat Clin Pract Endocrinol Metab 2008;4:394-405.PMID: 18506157.



42. Stuckey BG, Yeap D, Turner SR. Thyroxine replacement during super-ovulation for in vitro fertilization: a potential gap in management? Fertil Steril 2010;93:2414PMID: 20079900.

43. Lee S, Kang DW, Hudgins-Spivey S, Krust A, Lee EY, Koo Y, et al. Theca-specific estrogen receptor-alpha knockout mice lose fertility prematurely. Endocrinology 2009;150:3855-3862.PMID: 19423761.




44. Cooke NE, David EV. Serum vitamin D-binding protein is a third member of the albumin and alpha fetoprotein gene family. J Clin Invest 1985;76:2420-2424.PMID: 2416779.



45. Nielsen HK, Brixen K, Bouillon R, Mosekilde L. Changes in biochemical markers of osteoblastic activity during the menstrual cycle. J Clin Endocrinol Metab 1990;70:1431-1437.PMID: 2110577.



46. Kushner I. The acute phase response: an overview. Methods Enzymol 1988;163:373-383.PMID: 2467171.


47. Schepeler T, Mansilla F, Christensen LL, Orntoft TF, Andersen CL. Clusterin expression can be modulated by changes in TCF1-mediated Wnt signaling. J Mol Signal 2007;2:6PMID: 17634137.



48. Orvieto R. Controlled ovarian hyperstimulation: an inflammatory state. J Soc Gynecol Investig 2004;11:424-426.


49. Orvieto R, Zagatsky I, Yulzari-Roll V, La Marca A, Fisch B. Substituting human chorionic gonadotropin by gonadotropin-releasing hormone agonist to trigger final follicular maturation, during controlled ovarian hyperstimulation, results in less systemic inflammation. Gynecol Endocrinol 2006;22:437-440.PMID: 17012105.


50. Revelli A, Delle Piane L, Casano S, Molinari E, Massobrio M, Rinaudo P. Follicular fluid content and oocyte quality: from single biochemical markers to metabolomics. Reprod Biol Endocrinol 2009;7:40PMID: 19413899.



51. Ferrara N. Role of vascular endothelial growth factor in regulation of physiological angiogenesis. Am J Physiol Cell Physiol 2001;280:C1358-C1366.PMID: 11350730.


52. Fukuda R, Hirota K, Fan F, Jung YD, Ellis LM, Semenza GL. Insulin-like growth factor 1 induces hypoxia-inducible factor 1-mediated vascular endothelial growth factor expression, which is dependent on MAP kinase and phosphatidylinositol 3-kinase signaling in colon cancer cells. J Biol Chem 2002;277:38205-38211.PMID: 12149254.


53. Agrawal R, Sladkevicius P, Engmann L, Conway GS, Payne NN, Bekis J, et al. Serum vascular endothelial growth factor concentrations and ovarian stromal blood flow are increased in women with polycystic ovaries. Hum Reprod 1998;13:651-655.PMID: 9572428.



54. Bora NS, Kaliappan S, Jha P, Xu Q, Sohn JH, Dhaulakhandi DB, et al. Complement activation via alternative pathway is critical in the development of laser-induced choroidal neovascularization: role of factor B and factor H. J Immunol 2006;177:1872-1878.PMID: 16849499.


55. Girardi G, Yarilin D, Thurman JM, Holers VM, Salmon JE. Complement activation induces dysregulation of angiogenic factors and causes fetal rejection and growth restriction. J Exp Med 2006;203:2165-2175.PMID: 16923853.



56. Nozaki M, Raisler BJ, Sakurai E, Sarma JV, Barnum SR, Lambris JD, et al. Drusen complement components C3a and C5a promote choroidal neovascularization. Proc Natl Acad Sci U S A 2006;103:2328-2333.PMID: 16452172.



57. Jarkovska K, Kupcova Skalnikova H, Halada P, Hrabakova R, Moos J, Rezabek K, et al. Development of ovarian hyperstimulation syndrome: interrogation of key proteins and biological processes in human follicular fluid of women undergoing in vitro fertilization. Mol Hum Reprod 2011;17:679-692.PMID: 21697218.



58. Wu R, Van der Hoek KH, Ryan NK, Norman RJ, Robker RL. Macrophage contributions to ovarian function. Hum Reprod Update 2004;10:119-133.PMID: 15073142.



59. Kruger AJ, Yang C, Tam SW, Hinerfeld D, Evans JE, Green KM, et al. Haptoglobin as an early serum biomarker of virus-induced autoimmune type 1 diabetes in biobreeding diabetes resistant and LEW1WR1 rats. Exp Biol Med (Maywood) 2010;235:1328-1337.PMID: 20975081.



Figure 1
Experimental flow of the comparative analysis of natural-cycle versus stimulated-cycle human follicular fluid using a proteomics approach. MARS, multiple affinity removal; LC, liquid chromatography; MS, mass spectrometry.
Figure 2
Proteomic profiles of total proteins in natural and stimulated hFF samples. (A) Venn diagram describing the identified proteins, including 126 (natural-cycle hFF) and 167 (stimulated cycle hFF) proteins identified by LC-MS/MS. (B) Pathway map of the proteins identified in natural and stimulated hFF samples. Sorting was carried out for statistically significant pathway maps. HDL, high-density lipoprotein; IL, interleukin; hFF, human follicular fluid; LC, liquid chromatography; MS, mass spectrometry.

Figure 3
Proteomic profiles of N-glycoproteins in natural and stimulated human follicular fluid (hFF) samples. Venn diagrams showing (A) total proteins versus N-glycoproteins in natural-cycle hFF samples, (B) total proteins versus N-glycoproteins in stimulated-cycle hFF samples, and (C) the N-glycoproteins identified in natural and stimulated-cycle hFF samples. (D) Pathway map of the N-glycoproteins identified in natural and stimulated-cycle hFF samples. Sorting was carried out for statistically significant pathway maps. IL, interleukin; ECM, extracellular matrix.
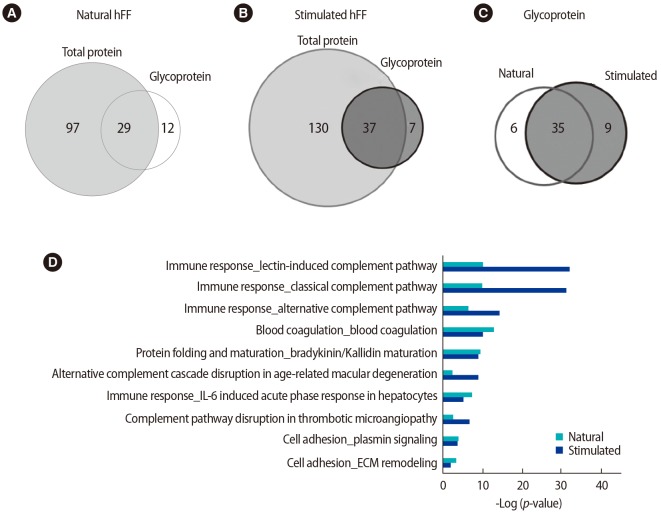
Figure 4
Validation of N-glycoproteins identified only in the stimulated-cycle human follicular fluid samples by western blotting. TBG, thyroxine-binding globulin; VDBP, vitamin D-binding protein.
- TOOLS