 |
 |
- Search
| Clin Exp Reprod Med > Volume 39(2); 2012 > Article |
Abstract
Objective
This study was undertaken to determine the effect of hypoxia inducible factor (HIF)-1α on the cell death, autophagy, and invasion of trophoblasts.
Methods
To understand the effect of HIF-1α, we inhibited HIF-1α using siRNA under normoxia and hypoxia conditions. Invasion assay and zymography were performed to determine changes in the invasion ability of HIF-1α. Western blotting and immunofluorescence were performed to determine some of the signal events involved in apoptosis and autophagy.
Results
There was no difference in cell death through the inhibition of HIF-1α expression by siRNA; however, the expression of LC3 and autophagosome formation increased. On the other hand, autophagy was increased, and the invasive ability of trophoblast cells decreased according to the inhibition of HIF-1α expression by siRNA. These experimental results mean that HIF-1α genes regulate the invasive ability of trophoblasts by increasing autophagy.
Trophoblasts, which originate from the trophectoderm and are located in the marginal zone of the blastocyst, are the most typical cells in the placenta and influence the fetal and placental development by infiltrating into the maternal endometrium during early implantation [1]. It has been determined that the invasive ability of trophoblast cells is regulated by various environmental factors including signaling of the adhesion and growth factors regulated by the interactions of decidua and trophoblasts in the endometrium. Furthermore, the maternal immune system, such as the activity of T cells and macrophages, is also known to regulate the invasion activity of trophbolast cells [2,3]. In particular, hypoxic conditions, which are low oxygen partial pressure, are known as the typical extrinsic factor that regulates the invasive ability of trophoblast cells. During early pregnancy, the partial pressure of oxygen in the uterus (17.9 mm Hg) is lower than normal oxygen pressure (55 mm Hg), and the trophoblast cells migrate and invade the surrounding blood vessels of the endometrium in the maternal uterus in persisting hypoxic conditions. Eventually, blood flow is increased by the expansion of the spiral arteries in the endometrium, and normoxic conditions in the invaded trophoblast cells are maintained. In addition, the various growth factors that are secreted by the trophoblast cells play important roles in placental angiogenesis [4-6]. It was also reported that trophoblast cells differentiated into extravillous trophoblasts under hypoxic conditions and influenced placental development by regulating the differentiation of trophoblast cells [7]. It was found that the extravillous trophoblasts that invaded the endometrial stroma and myometrium under hypoxic conditions played a vital role in pregnancy. After the mid-term of pregnancy, normoxic conditions were restored when the maternal-fetal blood flow increased with the invasion of trophoblasts into the endometrium, and then the trophoblast invasion ceased. On the other hand, it has been reported that the abnormal invasive activity of trophoblasts caused typical obstetric diseases such as pre-eclampsia and intrauterine growth retardation [8,9].
Hypoxia inducible factor (HIF)-1, a transcription factor, regulated the cellular transcription gene expression depending on the oxygen concentration, and acted as a heterodimeric complex consisting of subunit HIF-1α and subunit HIF-1β. In particular, it was reported that HIF-1α was expressed primarily in hypoxic conditions. In normal oxygen conditions, HIF-1α binds to von Hippel Lindau (VHL), a tumor suppressor gene, through the activation of prolyl hydroxylase (PHD), and degrades by ubiquitination. However, HIF-1α migrated into the nucleus, bound to HIF-1α and p300-CBP, and became stabilized in hypoxic conditions by suppressing the activity of PHD [10]. Recently, it was found that the invasion ability of trophoblasts was regulated by the expression of HIF-1α [11]. It was found that these hypoxic conditions regulated the invasion activity of trophoblasts by influencing the proliferation and apoptosis of trophoblasts [12]. On the other hand, it was also determined that autophagy, unlike apoptosis and necrosis, induced cell death by phagocytosis, forming the autophagosome, which fused with a lysosome to recycle aged organelles and proteins [13]. It was reported that the autophagosome, the typical marker of autophagy, was formed by activating PI3K III complex, Beclin-1, autophagy-related protein (ATG) 5-12, LC3, and ATG-8, and that mammalian target of rapamycin (mTOR) suppressed the formation of autophagosomes by suppressing ATG protein 5-12 [14,15].
Recently, it was demonstrated that autophagy was higher in the pre-eclamptic placenta than the normal placenta [16]. It was reported that the higher autophagy promoted the rupture of the amnionic membrane that surrounds the fetus during pregnancy [17]. In addition, it was reported that the autophagy increased the cellular stress level with apoptosis and influenced cellular differentiation [18].
However, few studies have addressed the mechanisms of autophagy regulation and the invasion of trophoblasts according to HIF-1α expression. Therefore, the present study was undertaken to analyze the autophagy of the trophoblast in hypoxic conditions and to demonstrate the relationship between the invasion activity of trophoblasts, which decreased HIF-1α, and autophagy using siRNA.
Cells from the human extravillous trophoblast cell line HTR-8/SVneo, which were kindly donated by Dr. Charles H. Graham (Queen's University, Kingston, Canada), were cultured in RPMI-1640 medium supplemented with 1% penicillin/streptomycin (Gibco BRL, Logan, UT, USA) and 5% fetal bovine serum (FBS, Gibco BRL) at 37℃ in a 5% CO2 incubator. To increase the efficiency of the hypoxia treatment, the cells were cultured in a hypoxia chamber (C-chamber, BioSpherix Ltd., Lacona, NY, USA) by lowering the oxygen concentration to 1%.
HTR-8/SVneo trophoblast cells (1×106) were plated into a 100 mm dish (Thermo Fisher Scientific, Roskilde, Denmark) and mixed with 20 nM of siRNA-HIF-1α (Genolution Pharmaceuticals, Seoul, Korea) and 4 µL of Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) for 20 minutes at room temperature. The cells were cultured for 24 hours and 48 hours in normoxia and hypoxia conditions, respectively.
To analyze the cell proliferation activity under the conditions of the treatment with siRNA-HIF-1α and hypoxia, the trophoblast cells were plated onto a 6-well plate (Becton Dickinson Labware, Bedford, MA, USA) at a concentration of 5×104 cells/well and treated with 20 nM of siRNA-HIF-1α. After culture in normoxia or hypoxia conditions, the cells were stained with trypan blue (Gibco BRL) and the number of cells was counted. This experiment was repeated three times.
The invasion of trophoblasts was analyzed by using a 24-well insert system (8 µm pore size; Becton Dickinson Labware). In the upper insert, 2.5×104 cells/well were plated, and the culture medium-supplemented FBS was added in the lower one, and then both were cultured for 24 hours. After incubation, the trophoblast cells remaining in the upper insert were removed using a swab and the trophoblast cells infiltrated into the lower insert were fixed with methanol and stained with Mayer's hematoxylin (Dako, CA, USA) for nuclear staining and counted. The experiments were repeated three times.
Under conditions of normoxia and hypoxia, the trophoblast cells were placed onto the gelatin-coated glass slide in a 24-well plate and cultured for 24 hours and 48 hours, respectively. The cultured cells were washed with PBS (Gibco BRL) and blocked with protein block serum-free buffer (Dako) for 30 minutes at 37℃. The cells were then incubated overnight with mouse anti-human LC3 (Novus, Littleton, CO, USA) diluted with blocking buffer to 1:200. The next day, after washing with PBS, the cells were incubated with secondary antibody (1:400) conjugated to Alexa 586 (Invitrogen) for 40 minutes at room temperature. Next, the slides were mounted using the inclusion solution (Dako) containing 4', 6-diamidino-2-phenylindole for nuclear counterstaining and observed under a fluorescence microscope (Axioskop2, Carl Zeiss Microimaging, Germany) to identify the formation of the cytoplasmic autophagosomes. In case of monodansylcadaverine (MDC) staining, the cells were incubated under the same culture conditions and then reacted with 0.05 mM MDC (Sigma, St. Louis, MO, USA) for 30 minutes at 37℃. After washing with PBS, the cells were fixed with 4% paraformaldehyde for 30 minutes. Then, the slides were mounted with fluorescence mounting medium (Dako) and observed under a fluorescence microscope (Axioskop2).
In order to determine the changes in protein expression according to normoxia and hypoxia conditions, 100 µL of RIPA buffer (50 mM/L Tris-HCl, pH 7.6, 150 mM/L NaCl, 1% NP40, 0.25% deoxycholic acid, 0.1% SDS, 1 mM/L EGTA, 1 mM/L EDTA, 10 mM/L NaF, 1 mM/L NA3 VO4) containing protease inhibitor cocktail solutions was added to the 24-hours and 48-hours cultured trophoblast cells and left on ice for 5 minutes, and then scraped off. The supernatant was obtained by centrifugation at 15,000 rpm for 15 minutes (Sorvall Fresco, Thermo Fisher Scientific). The concentration of protein was quantified using a BCA assay kit (Pierce, Rockford, IL, USA). A total of 40 µg of protein was incubated for 5 minutes at 95℃, electrophoresed on 10%, 12%, and 15% of SDS-PAGE, and then transferred onto PVDF membrane (Bio-Rad, Hercules, CA, USA). The PVDF membranes were incubated in 5% skim milk (BD, Sparks, MD, USA) for 40 minutes at room temperature, and then reacted with mouse-anti HIF-1α (1:1,000, Cell signal, MA, USA), rabbit-anti Bcl2 (1:1,000, Cell signal), rabbit-anti Bax (1:1,000, Cell signal), rabbit-anti phosphorylated mTOR (p-mTOR; 1:1,000, Santa Cruz Biotechnology, CA, USA), mouse-anti PI3KIII (1:1,000, Cell signal), rabbit-anti beclin1 (1:1,000, Cell signal), rabbit-anti LC3 (1:2,000, Cell signal) and rabbit-anti GAPDH (1:3,000, Cell signal) at 4℃ overnight. After the reaction, the membranes were incubated with horseradish peroxidase (HRP)-conjugated rabbit (1:25,000) (Bio-Rad) secondary antibody or mouse secondary antibody (1:25,000) (Bio-Rad) for 1 hour at room temperature. Protein expressions were identified using an ECL Advance western blot detection kit (Amersham, Piscataway, NJ, USA) to visualize the peroxidase activity.
Zymography was conducted to analyze matrix metalloproteinases (MMPs) activity in HTR-8/SVneo trophoblast cells according to siRNA-HIF-1α transfection. 20 nM of siRNA-HIF1α were treated in 1×106 cells plated on a 10 mm culture dish, and each supernatant was obtained after 24-hours and 48-hours culture under normoxia and hypoxia conditions. Each 30 µL of the obtained medium was electrophoresed on 12% SDS-PAGE gel containing 1 mg/mL of gelatin (Sigma). After electrophoresis, the gels were washed twice for 15 minutes using renaturation buffer (Bio-Rad) and incubated overnight with development buffer (Bio-Rad) at 37℃. Then, the gels were stained with Coomassie brilliant blue R-250 solution (Bio-Rad) for 40 minutes at room temperature and destained using destaining solution (10% acetic acid/40% methanol) until the white bands became visible. The gels were dried and MMP activity was analyzed according the intensity of the bands.
To analyze the cytotoxicity and invasion activity induced by siRNA-HIF-1α, statistical analysis was conducted using the Student's t-test and analysis of variance with SAS software (SAS Institute Inc., Cary, NC, USA). The data values were expressed as mean±SE and considered significant when p-values were lower than 0.05.
The expressions of HIF-1α and Bax proteins were increased, whereas, those of Bcl2 were decreased in trophoblast cells cultured in normoxia and hypoxia conditions for 24 and 48 hours (Figure 1).
Under hypoxic conditions, apoptosis of trophoblast cells increased, so the expression of ATG was analyzed. Phosphorylation of mTOR, which inhibits autophagy, and the expression of PI3KIII and Beclin1 were high under normoxic conditions. However, their expressions under hypoxic conditions decreased and the expression of type II LC3, one of the autophagy-related markers, was higher than under normoxia (Figure 2A). In addition, in the case of MDC and LC3 immunostaining, which was performed to confirm the formation of cytoplasmic lysosomes, the horseshoe-shaped autophagosomes in the cytoplasm were increased in proportion to the extent of time of hypoxic conditions (Figure 2B).
By analysis of the trophoblast invasion ability according to the hypoxic and normoxic conditions, it was shown that the invasion ability of trophoblast cells under hypoxic conditions was significantly higher than that of normoxic conditions (p<0.05) (Figure 3A). In addition, in the analysis of MMP activity by zymography, the activity of MMP-2 was found to be slightly decreased under hypoxic conditions than in a normoxic state; however, the activity of MMP-9 in hypoxic conditions was more increased than in normoxia (Figure 3B).
To identify the effects of HIF-1α expression changes on the apoptosis and the proliferation of trophoblast cells under hypoxic conditions, we compared the expression of apoptosis-related proteins before and after their treatment with siRNA-HIF-1α, and under normoxia and hypoxia conditions. After siRNA-HIF-1α treatment, it was shown that the expression of HIF-1α in cultured cells for 24 hours and 48 hours under hypoxic conditions was significantly decreased. There was no difference in Bax protein expression. However, in the case of Bcl2, its expression was slightly decreased after siRNA-HIF-1α treatment under nomoxia condition at 24 hours, and there were no differences in the other conditions (Figure 4A). In the present analysis of the effect of HIF-1α expression on trophoblast proliferation, no statistical differences were found in either normoxia or hypoxia conditions (p>0.05) (Figure 4B).
In the analysis of autophagy of trophoblast cells according to decreased HIF-1α expression by siRNA, the expression of the p-mTOR protein was decreased and that of PI3KIII was increased as HIF-1α expression decreased under normoxic culture conditions. In addition, in the case of Beclin1 protein, it was shown that its expression was increased after 48-hours treatment in both normoxia and hypoxia, while it was shown that the expression of Beclin1 protein was increased after 48-hours treatment under both normoxia and hypoxia conditions. It was noted that type II LC3 protein expression increased with decreasing HIF-1α expression in all of the experimental conditions except the 24-hours cultivation group (Figure 5A). In particular, the expression of type II LC3 protein increased with decreasing HIF-1α expression under hypoxic conditions. In the case of autophagosome formation, which was identified by MDC staining, cytoplasmic autophagosome formation was increased when HIF-1α expression was decreased under both normoxia and hypoxia conditions (Figure 5B). The expression of type II LC3 was increased under both normoxia and hypoxia conditions after siRNA-HIF-1α treatment (Figure 5C).
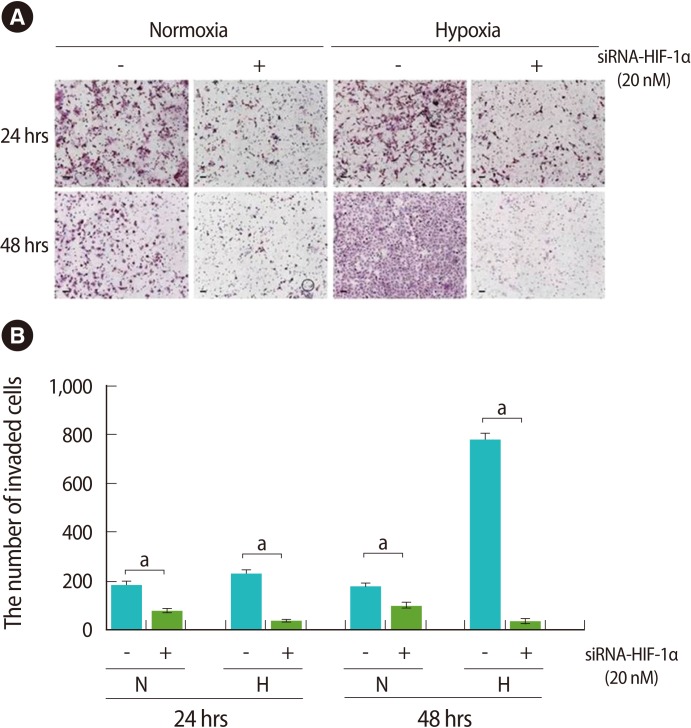
To analyze the invasion ability according to HIF-1α expression, we performed an invasion assay. The invasion ability of trophoblast cells under hypoxic conditions was more significantly increased than under normoxia (Figure 6A). The invasion ability of trophoblast cells was significantly decreased as HIF-1α expression decreased when compared to the control (p<0.005) (Figure 6B).
In early pregnancy, hypoxia is the typical extrinsic factor that increases the invasion ability and the alteration of various genes including increased integrin α4 by hypoxia. It has pivotal roles in early implantation by regulating the invasion ability of trophoblast cells [19]. In addition, it has been reported that the expression changes of matricellular protein CYR61 (CCN1), NOV (CCN3), and TGF-β, which are induced by hypoxia, regulate the invasion of JEG3, a choriocarcinoma cell line [20]. It is also known that the change in MMP-2 expression, which is increased under hypoxia, inhibits the invasion of the extravillous trophoblast; otherwise, the expression of MMP-9 and tissue inhibitor of metalloproteases-1 (TIMP-1) increases the invasion of trophoblast cells [21,22]. It has also been reported that hypoxia induces the invasion as well as apoptosis of trophoblast cells. These events are regulated by expressions of Bax and Bcl2 [23]. Also, hypoxia induces the proliferation, differentiation, and changes in physiological function of the trophoblast cells by regulating the expressions of various genes [24,25]. However, there is controversy about the effects of HIF-1α, which is expressed under conditions of hypoxia on trophoblast cells. In this study, we analyzed the effects of HIF-1α on the invasion, apoptosis, and autophagy of the trophoblast cells.
It has been reported that hypoxia, which acts as a factor involved in the invasion of tumor cells, increases the expression of MMP-1 via the regulation of CXCR4/SDF1 expression and also increases the invasion of sarcomere tumor cells via ERK signaling [26]. In addition, it is known that hypoxia regulates HIF-1α expression via mTOR signaling [27]. It is known that the activated HIF-1α by hypoxia has effects on the invasion of trophoblast cells as well as playing a role in the growth regulation of trophoblast cells [28]. In the present study, it was determined that decreased HIF-1α expression by siRNA promoted the apoptosis of trophoblast cells; however, there were no direct effects on cell division and proliferation of trophoblast cells (Figure 4). Apoptosis is the process of programmed cell death, which is controlled by specific genes and classified into apoptosis, necrosis, and autophagy. Autophagy, unlike pathologic cell death such as necrosis and apoptosis, plays an important role in simple cell death as well as immune protection [29]. It is known that autophagy is a process of phagocytosis of aged and damaged cells by fusion with lysosomes and a recycling mechanism of the degraded cellular constituents (cellular building blocks), and unlike simple cell death, it augments the cellular survival rate independent of HIF-1α under hypoxic conditions [30]. Trophoblast cells under hypoxic conditions show an increase in autophagic events, and this is regulated by the increase in LC3 and autophagy factor, and a decrease in Beclin1. It is reported that the increase in autophagy is related to preeclampsia [16]. Recently, it has been reported that the expression of HIF-1α regulates apoptosis by controlling the expression of type II LC3 and Beclin1, which were early autophagy factors [31,32]. In the present results, it was identified that apoptosis and autophagy were increased, the expression of the p-mTOR and PI3KIII were significantly reduced, and the expression of type II LC3 and the formation of autophagosome were increased in the trophoblast cells that were cultured under hypoxic conditions. However, it was interesting that there were no differences in apoptosis by siRNA-HIF-1α, whereas expression of LC3 II and formation of autophagosome were increased. From these results, it can be concluded that the expression of HIF-1α in trophoblast cells might be involved in autophagy rather than apoptosis, and further research is needed to prove the exact mechanism.
It has been reported that MMP expression increased during the invasion of trophoblast cells, and the expressions of MMP-2 and MMP-9 have especially important roles in the invasion of trophoblasts into the maternal blood vessels in the endometrium through the decidual layer. However, there is a great deal of controversy in the regulation mechanism of trophoblast invasion by MMP-2 and MMP-9 activity [33]. In this study, we observed that trophoblast invasion activity increased with the increase in the activity of MMP-9, rather than MMP-2.
It has also been reported that HIF-1α expression regulates MDA-MB231 invasion by controlling type II LC3 expression [34]. In addition, the NOV protein, which is regulated by HIF-1α expression, also regulates the invasion of trophoblast cells [20]. In the present study, however, it was found that the inhibition of HIF-1α augmented autophagy, thereby reducing the invasion of trophoblast cells.
In conclusion, the decreased HIF-1α expression using siRNA increases autophagy in conditions of both normoxia and hypoxia and thereby reduces the invasion ability of trophoblast cells.
Taken together, these results might be used as important data in the diagnosis and treatment of obstetric diseases through control of the invasion ability with the understanding of HIF-1α gene expression regulation and pre-eclampsia, which are induced by the abnormal early implantation and malfunction of trophoblast cells.
Acknowledgments
We thank Professor Dr. Charles H. Graham (Queen's University, Kingston, Canada) for the kind donation of the early pregnancy trophoblast cell line, HTR-8/SVneo.
Notes
References
1. Horne AW, White JO, Lalani EN. The endometrium and embryo implantation. A receptive endometrium depends on more than hormonal influences. BMJ 2000;321:1301-1302.PMID: 11090499.



2. Flaminio MJ, Antczak DF. Inhibition of lymphocyte proliferation and activation: a mechanism used by equine invasive trophoblast to escape the maternal immune response. Placenta 2005;26:148-159.PMID: 15708116.


3. Harris LK. Review: Trophoblast-vascular cell interactions in early pregnancy: how to remodel a vessel. Placenta 2010;31(Suppl): S93-S98.PMID: 20060584.


4. Caniggia I, Winter JL. Adriana and Luisa Castellucci Award lecture 2001. Hypoxia inducible factor-1: oxygen regulation of trophoblast differentiation in normal and pre-eclamptic pregnancies: a review. Placenta 2002;23(Suppl A): S47-S57.PMID: 11978059.


5. Mano Y, Kotani T, Shibata K, Matsumura H, Tsuda H, Sumigama S, et al. The loss of endoglin promotes the invasion of extravillous trophoblasts. Endocrinology 2011;152:4386-4394.PMID: 21914777.


6. Dilly M, Hambruch N, Haeger JD, Pfarrer C. Epidermal growth factor (EGF) induces motility and upregulates MMP-9 and TIMP-1 in bovine trophoblast cells. Mol Reprod Dev 2010;77:622-629.PMID: 20578063.


7. De Marco CS, Caniggia I. Mechanisms of oxygen sensing in human trophoblast cells. Placenta 2002;23(Suppl A): S58-S68.PMID: 11978060.


8. Lian IA, Toft JH, Olsen GD, Langaas M, Bjorge L, Eide IP, et al. Matrix metalloproteinase 1 in pre-eclampsia and fetal growth restriction: reduced gene expression in decidual tissue and protein expression in extravillous trophoblasts. Placenta 2010;31:615-620.PMID: 20452670.


9. Jeyabalan A, McGonigal S, Gilmour C, Hubel CA, Rajakumar A. Circulating and placental endoglin concentrations in pregnancies complicated by intrauterine growth restriction and preeclampsia. Placenta 2008;29:555-563.PMID: 18462791.



10. Nizet V, Johnson RS. Interdependence of hypoxic and innate immune responses. Nat Rev Immunol 2009;9:609-617.PMID: 19704417.



11. Dubinsky V, Poehlmann TG, Suman P, Gentile T, Markert UR, Gutierrez G. Role of regulatory and angiogenic cytokines in invasion of trophoblastic cells. Am J Reprod Immunol 2010;63:193-199.PMID: 20047586.

12. Genbacev O, DiFederico E, McMaster M, Fisher SJ. Invasive cytotrophoblast apoptosis in pre-eclampsia. Hum Reprod 1999;14(Suppl 2): 59-66.PMID: 10690801.


13. Rajawat YS, Hilioti Z, Bossis I. Aging: central role for autophagy and the lysosomal degradative system. Ageing Res Rev 2009;8:199-213.PMID: 19427410.


14. Mizushima N. The pleiotropic role of autophagy: from protein metabolism to bactericide. Cell Death Differ 2005;12(Suppl 2): 1535-1541.PMID: 16247501.


15. Eskelinen EL. Maturation of autophagic vacuoles in mammalian cells. Autophagy 2005;1:1-10.PMID: 16874026.


16. Oh SY, Choi SJ, Kim KH, Cho EY, Kim JH, Roh CR. Autophagy-related proteins, LC3 and Beclin-1, in placentas from pregnancies complicated by preeclampsia. Reprod Sci 2008;15:912-920.PMID: 19050324.


17. Shen ZY, Li EM, Lu SQ, Shen J, Cai YM, Wu YE, et al. Autophagic and apoptotic cell death in amniotic epithelial cells. Placenta 2008;29:956-961.PMID: 18926571.


18. Zhang J, Liu J, Liu L, McKeehan WL, Wang F. The fibroblast growth factor signaling axis controls cardiac stem cell differentiation through regulating autophagy. Autophagy 2012;8.

19. Na KH, Lee HJ, Choi JH, Eun JW, Nam SW, Yoon TK, et al. Dynamic alterations in integrin alpha4 expression by hypoxia are involved in trophoblast invasion during early implantation. J Cell Biochem 2012;113:685-694.PMID: 21976114.


20. Wolf N, Yang W, Dunk CE, Gashaw I, Lye SJ, Ring T, et al. Regulation of the matricellular proteins CYR61 (CCN1) and NOV (CCN3) by hypoxia-inducible factor-1{alpha} and transforming-growth factor-{beta}3 in the human trophoblast. Endocrinology 2010;151:2835-2845.PMID: 20237132.


21. Onogi A, Naruse K, Sado T, Tsunemi T, Shigetomi H, Noguchi T, et al. Hypoxia inhibits invasion of extravillous trophoblast cells through reduction of matrix metalloproteinase (MMP)-2 activation in the early first trimester of human pregnancy. Placenta 2011;32:665-670.PMID: 21764444.


22. Luo J, Qiao F, Yin X. Hypoxia induces FGF2 production by vascular endothelial cells and alters MMP9 and TIMP1 expression in extravillous trophoblasts and their invasiveness in a cocultured model. J Reprod Dev 2011;57:84-91.PMID: 21041986.


23. Chen X, Liu Y, Xu X, Chen H. Decreased Cyr61 under hypoxia induces extravillous trophoblasts apoptosis and preeclampsia. J Huazhong Univ Sci Technolog Med Sci 2011;31:235-240.PMID: 21505992.


24. Genbacev O, Joslin R, Damsky CH, Polliotti BM, Fisher SJ. Hypoxia alters early gestation human cytotrophoblast differentiation/invasion in vitro and models the placental defects that occur in preeclampsia. J Clin Invest 1996;97:540-550.PMID: 8567979.



25. Genbacev O. To proliferate or to divide - to be or not to be. Early Pregnancy 2001;5:63-64.PMID: 11753517.

26. Sun X, Wei L, Chen Q, Terek RM. CXCR4/SDF1 mediate hypoxia induced chondrosarcoma cell invasion through ERK signaling and increased MMP1 expression. Mol Cancer 2010;9:17PMID: 20102637.




27. Land SC, Tee AR. Hypoxia-inducible factor 1alpha is regulated by the mammalian target of rapamycin (mTOR) via an mTOR signaling motif. J Biol Chem 2007;282:20534-20543.PMID: 17502379.


28. Maltepe E, Krampitz GW, Okazaki KM, Red-Horse K, Mak W, Simon MC, et al. Hypoxia-inducible factor-dependent histone deacetylase activity determines stem cell fate in the placenta. Development 2005;132:3393-3403.PMID: 15987772.


29. Verhagen AM, Coulson EJ, Vaux DL. Inhibitor of apoptosis proteins and their relatives: IAPs and other BIRPs. Genome Biol 2001;2:REVIEWS3009PMID: 11516343.


30. Wilkinson S, O'Prey J, Fricker M, Ryan KM. Hypoxia-selective macroautophagy and cell survival signaled by autocrine PDGFR activity. Genes Dev 2009;23:1283-1288.PMID: 19487569.



31. Bohensky J, Shapiro IM, Leshinsky S, Terkhorn SP, Adams CS, Srinivas V. HIF-1 regulation of chondrocyte apoptosis: induction of the autophagic pathway. Autophagy 2007;3:207-214.PMID: 17224629.


32. Belibi F, Zafar I, Ravichandran K, Segvic AB, Jani A, Ljubanovic DG, et al. Hypoxia-inducible factor-1alpha (HIF-1alpha) and autophagy in polycystic kidney disease (PKD). Am J Physiol Renal Physiol 2011;300:F1235-F1243.PMID: 21270095.



33. Isaka K, Usuda S, Ito H, Sagawa Y, Nakamura H, Nishi H, et al. Expression and activity of matrix metalloproteinase 2 and 9 in human trophoblasts. Placenta 2003;24:53-64.PMID: 12495660.


34. Indelicato M, Pucci B, Schito L, Reali V, Aventaggiato M, Mazzarino MC, et al. Role of hypoxia and autophagy in MDA-MB-231 invasiveness. J Cell Physiol 2010;223:359-368.PMID: 20112292.


Figure 1
Effect of hypoxia on expression of hypoxia inducible factor-1α (HIF-1α) and apoptosis-associated factors in trophoblast (HTR-8/SVneo) cells according to hypoxia treatment. Cells exposed to hypoxia for 24 hours and 48 hours were analyzed for the expression HIF-1α, B-cell lymphoma (Bcl2), Bcl-2 associated X protein (Bax), and Glyceraldehyde 3-phophate dehydrogenase (GAPDH) by western blot analysis. GAPDH was used as a loading control.

Figure 2
Effect of hypoxia on expression of autophagy-associated factors in trophoblast (HTR-8/SVneo) cells according to hypoxia treatment. (A) Cells exposed to hypoxia for 24 hours and 48 hours were analyzed for the expression of phosphorylated mammalian target of rapamycin (mTOR), phosphoinositide 3-kinase III (PI3K III), Beclin 1, little computer 3 (LC3) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) by western blot analysis. GAPDH was used as a loading control. (B) The cells were analyzed for the formation of autophagosomes using monodansylcadaverine (MDC; upper) and the expression of active LC3 by immunofluorescence (lower). A larger view is represented in the right lower corner. Propodium iodide was used for nuclear staining. Scale bar=80 µm.
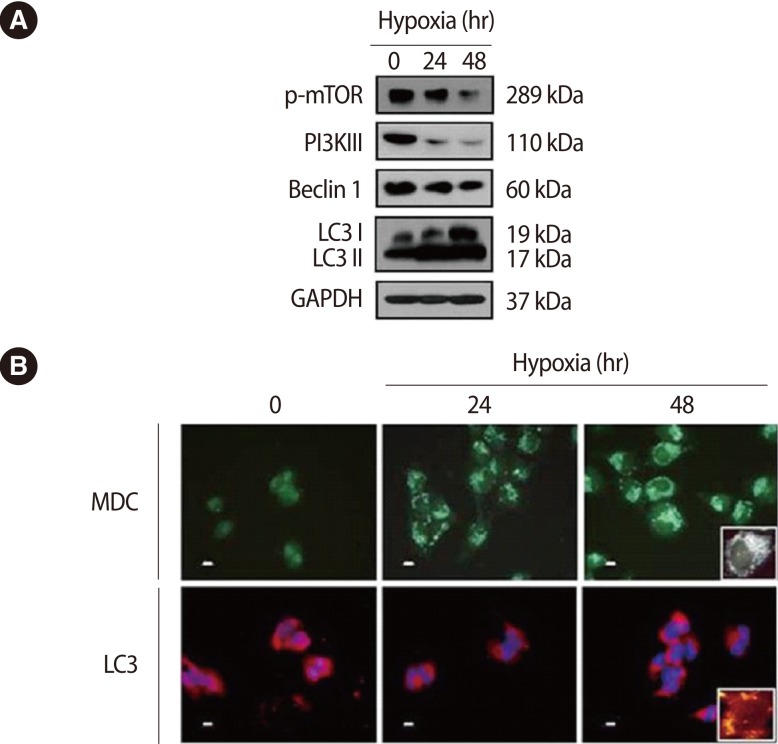
Figure 3
Hypoxia-dependent invasion analysis of trophoblast (HTR-8/SVneo) cells. (A) The number of invading cells in HTR-8 cells was analzyed using a trans insert system according to hypoxia treatment (n=3). Data are expressed as the mean±SE. aDepicts significant differences between the normoxia and hypoxia groups (p<0.001). (B) Enzyme activities of matrix metalloproteinase-2 (MMP-2; lower) and matrix metalloproteinase-9 (MMP-9; upper) in HTR-8 cells under hypoxia were determined by gelatin zymography analysis. The activities of MMP-9 in the hypoxia groups were higher than that of normoxia. The columns in the graphs represent the intensity of MMP-2 and MMP-9.

Figure 4
The effect of siRNA-hypoxia inducible facrot-1α (HIF-1α) and apoptosis related factors, and the proliferation activity of trophoblast (HTR-8/SVneo) cells. (A) The cells treated with siRNA-HIF-1α under mormoxia or hypoxia for 24 hours and 48 hours were analyzed for the expressions of HIF-1α, Bcl-2 associated X protein (Bax), B-cell lymphoma-2 (Bcl-2), and Glyceraldehyde 3-phophate dehydrogenase (GAPDH) in HTR-8 cells using western blot. GAPDH was used as a loading control. (B) The proliferation activity of HTR-8 cells treated with siRNA-HIF-1α under mormoxia or hypoxia for 24 hours and 48 hours were analyzed (n=3). The columns in the graphs represent the count analysis. N, normoxia; H, hypoxia. Data are expressed as the mean±SE; p>0.05, when compared with respective controls.
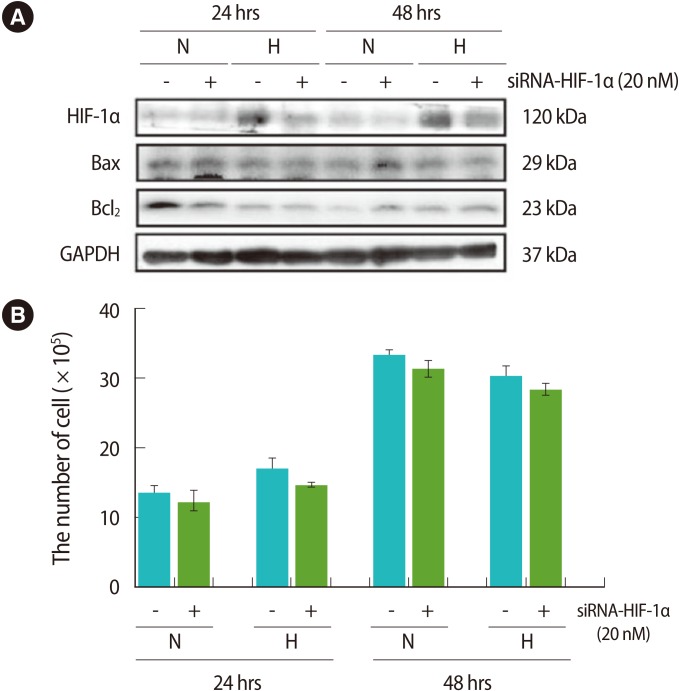
Figure 5
Effect of hypoxia and siRNA-hypoxia inducible factor-1α (HIF-1α) on autophagy in trophoblast (HTR-8/SVneo) cells. (A) HTR-8 cells were analyzed for the expression of autophagy-associated factors after hypoxia and siRNA-HIF-1a treatment. Western blot analysis was performed in same membrane after stripping. GAPDH was used as a loading control. (B) The cells were analyzed for the formation of autophagosomes using MDC staining after hypoxia and siRNA-HIF-1α treatment. (C) The cells were analyzed for the expression of active LC3 in HTR-8 cells using immunofluorescence after hypoxia and siRNA-HIF-1α treatment. Propodium iodide was used for nuclear staining. Scale bar=80 µm. N, Normoxia; H, Hypoxia; mTOR, mammalian target of rapamycin; PI3K III, phosphoinositide 3-kinase III; LC3, little computer 3; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; MDC, monodansylcadaverine.
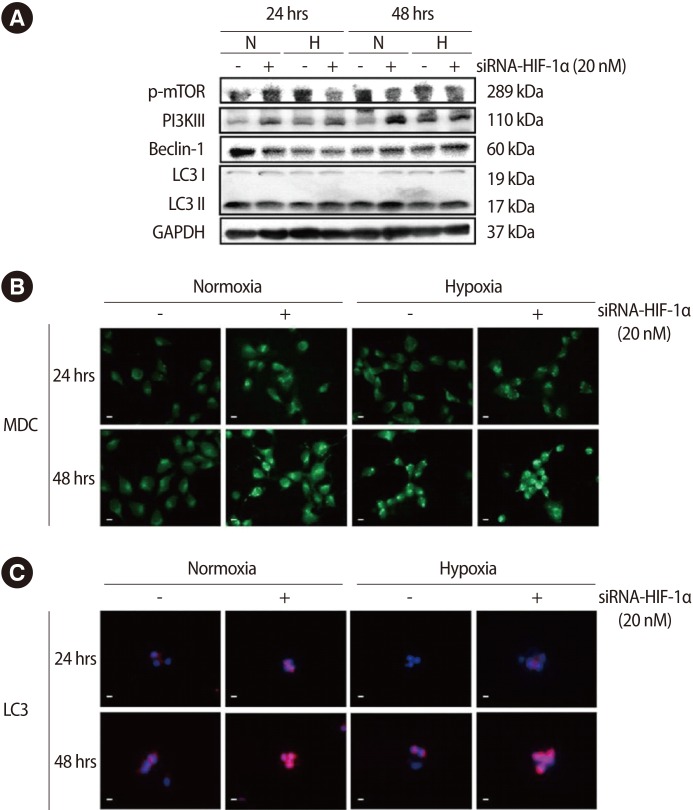
Figure 6
Effect of hypoxia and siRNA-hypoxia inducible factor-1α (HIF-1α) on invasion activity of trophoblast (HTR-8/SVneo) cells according to hypoxia and 20 nM siRNA-HIF-1α treatment. (A) Invading cells were analyzed using a trans insert system after hypoxia and siRNA-HIF-1α treatment. The cells were stained with hematoxylin and eosin (H&E) for the counting of the number of invading cells. (n=3). Scale bar=80 µm. (B) Columns in the graphs are the diagram of the invading cells of HTR-8 cells. N, normoxia; H, hypoxia. Data are expressed as the mean±SE. Significant differences are depicted between siRNA-HIF-1α treatment or none (ap<0.001).
-
METRICS

- Related articles in Clin Exp Reprod Med
-
Effects of selenium on the survival and invasion of trophoblasts2018 March;45(1)





