1. Practice Committee of American Society for Reproductive Medicine. Ovarian hyperstimulation syndrome. Fertil Steril 2008;90(5 Suppl): S188-S193.PMID:
19007627.


2. Boothroyd C, Karia S, Andreadis N, Rombauts L, Johnson N, Chapman M, et al. Consensus statement on prevention and detection of ovarian hyperstimulation syndrome. Aust N Z J Obstet Gynaecol 2015;55:523-534.PMID:
26597569.


3. Fritz MA, Speroff L. Clinical gynecologic endocrinology and infertility. 8th ed. Philadelphia: Lippincott Williams & Wilkins; 2011.
4. Humaidan P, Quartarolo J, Papanikolaou EG. Preventing ovarian hyperstimulation syndrome: guidance for the clinician. Fertil Steril 2010;94:389-400.PMID:
20416867.


5. Bellver J, Munoz EA, Ballesteros A, Soares SR, Bosch E, Simon C, et al. Intravenous albumin does not prevent moderate-severe ovarian hyperstimulation syndrome in high-risk IVF patients: a randomized controlled study. Hum Reprod 2003;18:2283-2288.PMID:
14585874.



6. Gokmen O, Ugur M, Ekin M, Keles G, Turan C, Oral H. Intravenous albumin versus hydroxyethyl starch for the prevention of ovarian hyperstimulation in an in-vitro fertilization programme: a prospective randomized placebo controlled study. Eur J Obstet Gynecol Reprod Biol 2001;96:187-192.PMID:
11384805.


7. Graf MA, Fischer R, Naether OG, Baukloh V, Tafel J, Nuckel M. Reduced incidence of ovarian hyperstimulation syndrome by prophylactic infusion of hydroxyaethyl starch solution in an in-vitro fertilization programme. Hum Reprod 1997;12:2599-2602.PMID:
9455820.



8. Westphal M, James MF, Kozek-Langenecker S, Stocker R, Guidet B, Van Aken H. Hydroxyethyl starches: different products--different effects. Anesthesiology 2009;111:187-202.PMID:
19512862.


9. Abbara A, Jayasena CN, Christopoulos G, Narayanaswamy S, Izzi-Engbeaya C, Nijher GM, et al. Efficacy of kisspeptin-54 to trigger oocyte maturation in women at high risk of ovarian hyperstimulation syndrome (OHSS) during in vitro fertilization (IVF) therapy. J Clin Endocrinol Metab 2015;100:3322-3331.PMID:
26192876.



10. Practice Committee of the American Society for Reproductive Medicine. Prevention and treatment of moderate and severe ovarian hyperstimulation syndrome: a guideline. Fertil Steril 2016;106:1634-1647.PMID:
27678032.


11. Hill MJ, Chason RJ, Payson MD, Segars JH, Csokmay JM. GnRH antagonist rescue in high responders at risk for OHSS results in excellent assisted reproduction outcomes. Reprod Biomed Online 2012;25:284-291.PMID:
22796230.



12. Gilliam ML. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Obstet Gynecol 2011;118:706-707.PMID:
21860304.


13. Youssef MA, Van der Veen F, Al-Inany HG, Mochtar MH, Griesinger G, Nagi Mohesen M, et al. Gonadotropin-releasing hormone agonist versus HCG for oocyte triggering in antagonist-assisted reproductive technology. Cochrane Database Syst Rev 2014;(10): CD008046PMID:
25358904.


14. Fatemi HM, Garcia-Velasco J. Avoiding ovarian hyperstimulation syndrome with the use of gonadotropin-releasing hormone agonist trigger. Fertil Steril 2015;103:870-873.PMID:
25724740.


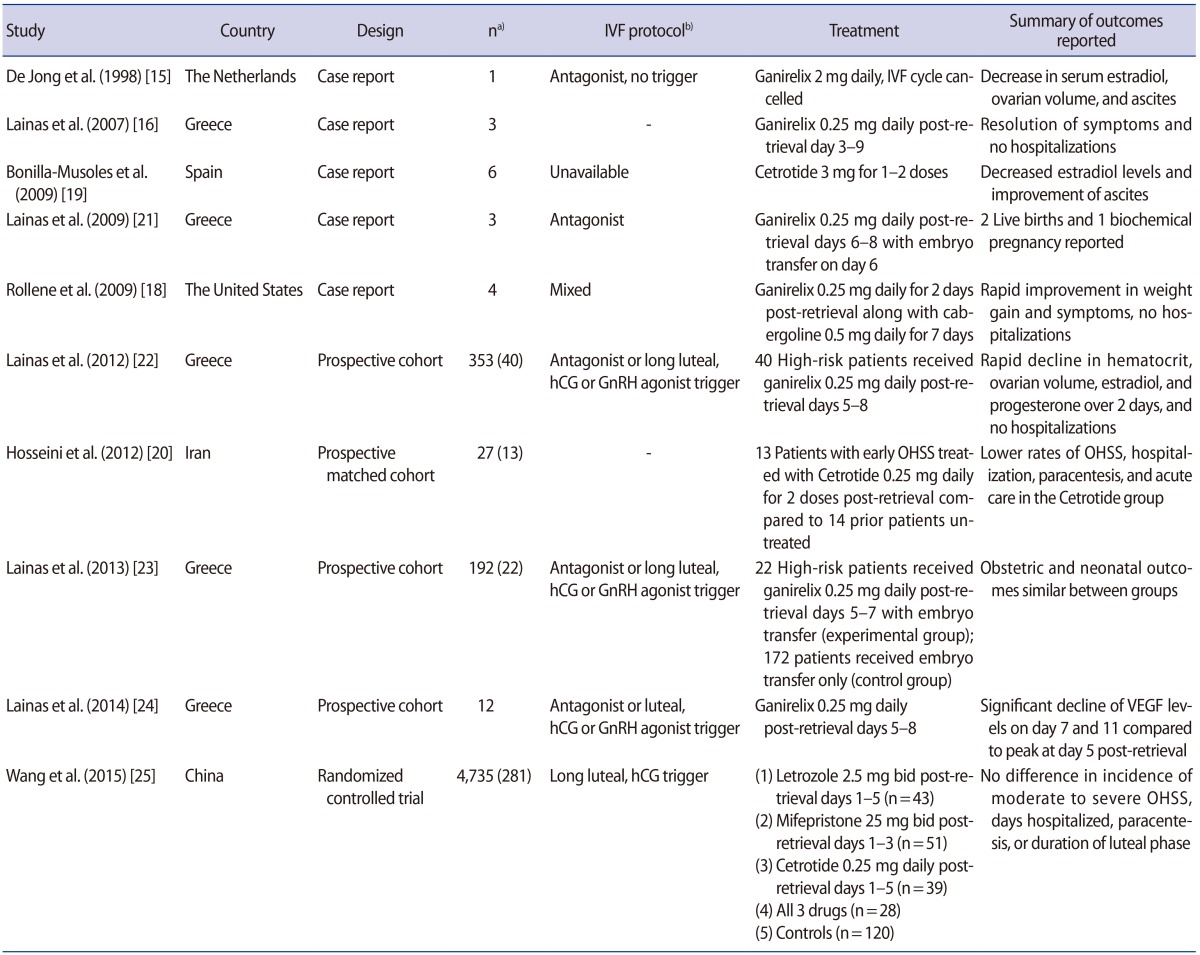
15. de Jong D, Macklon NS, Mannaerts BM, Coelingh Bennink HJ, Fauser BC. High dose gonadotrophin-releasing hormone antagonist (ganirelix) may prevent ovarian hyperstimulation syndrome caused by ovarian stimulation for in-vitro fertilization. Hum Reprod 1998;13:573-575.PMID:
9572414.



16. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Kolibianakis EM. Management of severe early ovarian hyperstimulation syndrome by re-initiation of GnRH antagonist. Reprod Biomed Online 2007;15:408-412.PMID:
17908403.


17. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Iliadis GS, et al. Management of severe OHSS using GnRH antagonist and blastocyst cryopreservation in PCOS patients treated with long protocol. Reprod Biomed Online 2009;18:15-20.

18. Rollene NL, Amols MH, Hudson SB, Coddington CC. Treatment of ovarian hyperstimulation syndrome using a dopamine agonist and gonadotropin releasing hormone antagonist: a case series. Fertil Steril 2009;92:1169.e15-1169.e17.


19. Bonilla-Musoles FM, Raga F, Castillo JC, Sanz M, Dolz M, Osborne N. High doses of GnRH antagonists are efficient in the management of severe ovarian hyperstimulation syndrome. Clin Exp Obstet Gynecol 2009;36:78-81.PMID:
19688946.

20. Hosseini MA, Mahdavi A, Aleyasin A, Safdarian L, Bahmaee F. Treatment of ovarian hyperstimulation syndrome using gonadotropin releasing hormone antagonist: a pilot study. Gynecol Endocrinol 2012;28:853-855.PMID:
22697477.


21. Lainas TG, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas GT, Alexopoulou E, et al. Live births after management of severe OHSS by GnRH antagonist administration in the luteal phase. Reprod Biomed Online 2009;19:789-795.PMID:
20031018.


22. Lainas GT, Kolibianakis EM, Sfontouris IA, Zorzovilis IZ, Petsas GK, Tarlatzi TB, et al. Outpatient management of severe early OHSS by administration of GnRH antagonist in the luteal phase: an observational cohort study. Reprod Biol Endocrinol 2012;10:69PMID:
22938051.



23. Lainas GT, Kolibianakis EM, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas TG, et al. Pregnancy and neonatal outcomes following luteal GnRH antagonist administration in patients with severe early OHSS. Hum Reprod 2013;28:1929-1942.PMID:
23624582.



24. Lainas GT, Kolibianakis EM, Sfontouris IA, Zorzovilis IZ, Petsas GK, Lainas TG, et al. Serum vascular endothelial growth factor levels following luteal gonadotrophin-releasing hormone antagonist administration in women with severe early ovarian hyperstimulation syndrome. BJOG 2014;121:848-855.PMID:
24621101.


25. Wang YQ, Luo J, Xu WM, Xie QZ, Yan WJ, Wu GX, et al. Can steroidal ovarian suppression during the luteal phase after oocyte retrieval reduce the risk of severe OHSS? J Ovarian Res 2015;8:63PMID:
26400057.



26. Mais V, Kazer RR, Cetel NS, Rivier J, Vale W, Yen SS. The dependency of folliculogenesis and corpus luteum function on pulsatile gonadotropin secretion in cycling women using a gonadotropin-releasing hormone antagonist as a probe. J Clin Endocrinol Metab 1986;62:1250-1255.PMID:
3084538.



27. Nelson LR, Fujimoto VY, Jaffe RB, Monroe SE. Suppression of follicular phase pituitary-gonadal function by a potent new gonadotropin-releasing hormone antagonist with reduced histamine-releasing properties (ganirelix). Fertil Steril 1995;63:963-969.PMID:
7536693.


28. Ortmann O, Weiss JM, Diedrich K. Embryo implantation and GnRH antagonists: ovarian actions of GnRH antagonists. Hum Reprod 2001;16:608-611.PMID:
11278203.


29. Peng C, Fan NC, Ligier M, Vaananen J, Leung PC. Expression and regulation of gonadotropin-releasing hormone (GnRH) and GnRH receptor messenger ribonucleic acids in human granulosa-luteal cells. Endocrinology 1994;135:1740-1746.PMID:
7956897.



30. Asimakopoulos B, Nikolettos N, Nehls B, Diedrich K, Al-Hasani S, Metzen E. Gonadotropin-releasing hormone antagonists do not influence the secretion of steroid hormones but affect the secretion of vascular endothelial growth factor from human granulosa luteinized cell cultures. Fertil Steril 2006;86:636-641.PMID:
16904114.


31. Tong XM, Zhang SY, Song T, Xu WH, Lin XN, Shu J, et al. Effects of gonadotropin-releasing hormone antagonists on the expression of vascular endothelial growth factor and its receptors in a rat model of ovarian hyperstimulation syndrome. Chin Med J (Engl) 2008;121:2434-2439.PMID:
19102964.


32. Navot D, Bergh PA, Laufer N. Ovarian hyperstimulation syndrome in novel reproductive technologies: prevention and treatment. Fertil Steril 1992;58:249-261.PMID:
1633889.