 |
 |
- Search
| Clin Exp Reprod Med > Volume 44(4); 2017 > Article |
Abstract
Objective
Methods
Results
Conclusion
References
Figure 1
Photographs of quarter laser zona thinning-assisted hatching. Twenty-five percent of the zona pellucida of a 2-cell and 8-cell fresh embryo is thinned to a depth of 70%–80% (×400). (A) Two-cell embryo before vitrification, (B) 8-cell embryo before vitrification.
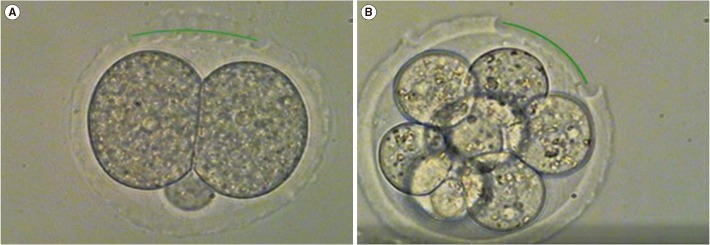
Figure 2
Photograph of cells differentially stained in a mouse hatched blastocyst (×400, double-fluorescence staining; inner cell mass, blue; trophectoderm, pink). (A–D) Two-cell mouse embryo, (E–H) 8-cell mouse embryo. (A) Control-8, 8 minutes of vitrification without quarter laser zona thinning-assisted hatching (qLZT-AH), 72 hours after thawing. (B) 2LAH-ES8, 8 minutes of vitrification after qLZT-AH, 72 hours after thawing. (C) 2LAH-ES6, 6 minutes of vitrification after qLZT-AH, 72 hours after thawing. (D) 2LAH-ES4, 4 minutes of vitrification after qLZT-AH, 72 hours after thawing. (E) Control-10, 10 minutes of vitrification without qLZT-AH, 48 hours after thawing. (F) 8LAH-ES10, 10 minutes of vitrification after qLZT-AH, 48 hours after thawing. (G) 8LAH-ES7, 7 minutes of vitrification after qLZT-AH, 48 hours after thawing. (H) 8LAH-ES4, 4 minutes of vitrification after qLZT-AH, 48 hours after thawing.

Table 1
Effects of laser-assisted hatching and exposure time to vitrification solution on blastocyst formation in mouse 2-cell embryos
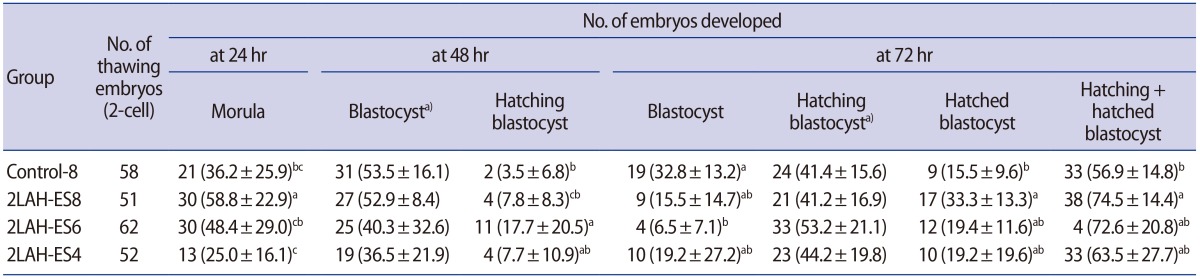
Values are presented as number (%, mean±standard deviation).
Control-8, 8 minutes of vitrification without quarter laser zona thinning-assisted hatching; 2LAH-ES8, 8 minutes of vitrification after quarter laser zona thinningassisted hatching; 2LAH-ES6, 6 minutes of vitrification after quarter laser zona thinning-assisted hatching; 2LAH-ES4, 4 minutes of vitrification after quarter laser zona thinning-assisted hatching.
a)No significant differences between groups; a-cStatistically significant differences (p<0.05).
Table 2
Effects of laser-assisted hatching and exposure time to vitrification solution on blastocyst formation in mouse 8-cell embryos
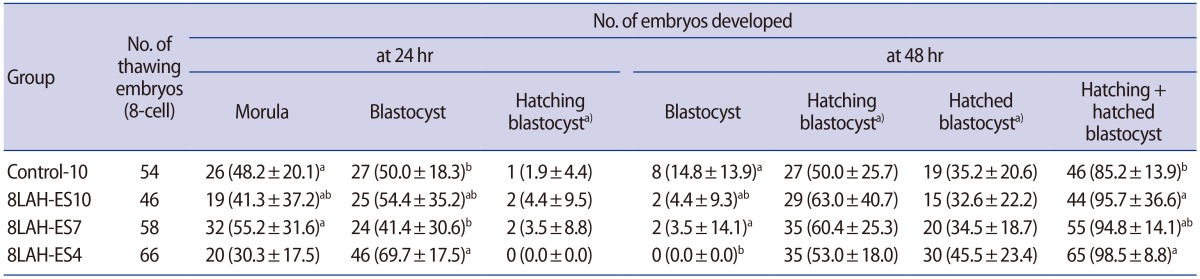
Values are presented as number (%, mean±standard deviation).
Control-10, 10 minutes of vitrification without quarter laser zona thinning-assisted hatching; 8LAH-ES10, 10 minutes of vitrification after quarter laser zona thinning-assisted hatching; 8LAH-ES7, 7 minutes of vitrification after quarter laser zona thinning-assisted hatching; 8LAH-ES4, 4 minutes of vitrification after quarter laser zona thinning-assisted hatching.
a)No significant differences between groups; a,bStatistically significant difference (p<0.05).
Table 3
Effects of laser-assisted hatching and exposure time to vitrification solution on cell number in mouse 2-cell embryos
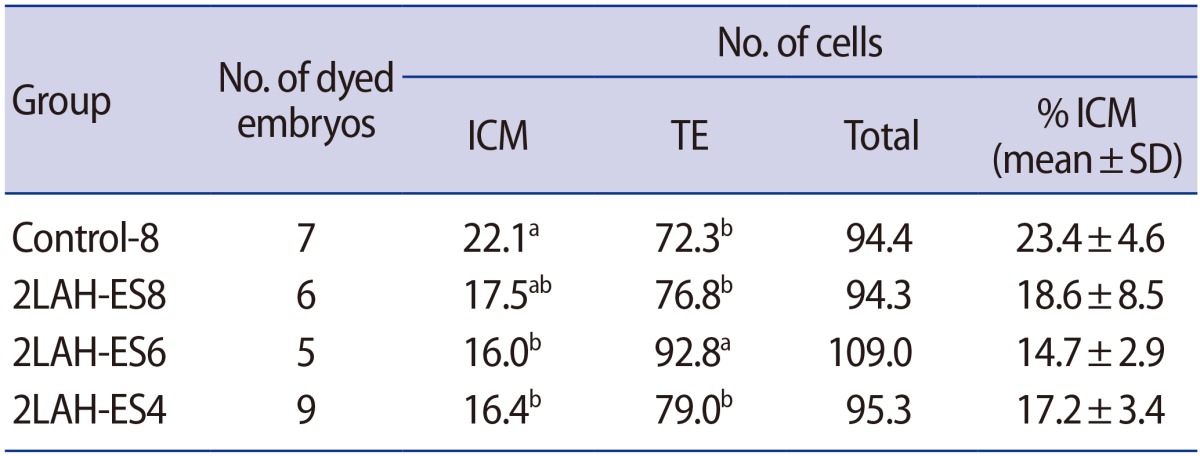
ICM, inner cell mass; TE, trophectoderm; SD, standard deviation; Control-8, 8 minutes of vitrification without quarter laser zona thinning-assisted hatching; 2LAH-ES8, 8 minutes of vitrification after quarter laser zona thinning-assisted hatching; 2LAH-ES6, 6 minutes of vitrification after quarter laser zona thinning-assisted hatching; 2LAH-ES4, 4 minutes of vitrification after quarter laser zona thinning-assisted hatching.
a,bStatistically significant difference (p<0.05).
Table 4
Effects of laser-assisted hatching and exposure time to vitrification solution on cell number in mouse 8-cell embryos
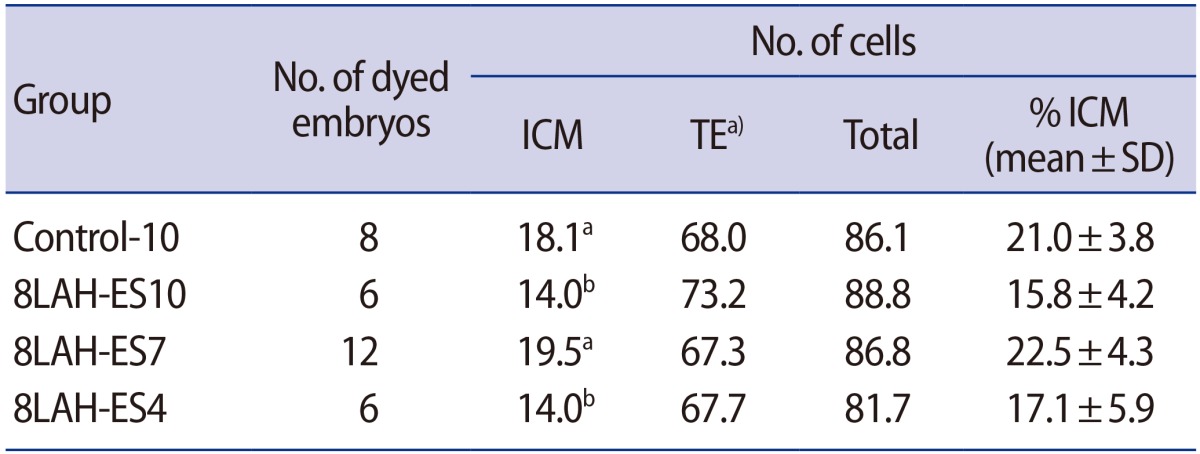
ICM, inner cell mass; TE, trophectoderm; SD, standard deviation; Control-10, 10 minutes of vitrification without quarter laser zona thinning-assisted hatching; 8LAH-ES10, 10 minutes of vitrification after quarter laser zona thinning-assisted hatching; 8LAH-ES7, 7 minutes of vitrification after quarter laser zona thinning-assisted hatching; 8LAH-ES4, 4 minutes of vitrification after quarter laser zona thinning-assisted hatching.
a)No significant differences between groups (p<0.05); a,bStatistically significant difference.
-
METRICS

- Related articles in Clin Exp Reprod Med
-
The Effects of Vero Cell Co-culture on Mouse Embryo Development.1997 August;24(2)
Effect of Partial Laser Assisted Hatching on Mouse Embryos.2001 June;28(2)





