 |
 |
- Search
| Clin Exp Reprod Med > Epub ahead of print |
Abstract
Objective
Autophagy is a major intracellular catabolic pathway governed by the sequential actions of proteins encoded by autophagy-related genes (Atg). ATG9, the only transmembrane protein involved in this process, regulates phospholipid translocation to autophagosomes during the early phases of autophagy. In mammals, two Atg9 isoforms have been reported: Atg9a and Atg9b. In this study, we examined whether the molecular and cellular characteristics of these two isoforms differed in mice.
Methods
Whole uteri were collected on days 1, 4, and 8 of pregnancy and from ovariectomized mice injected with vehicle, progesterone, or 17β-estradiol. Cells from reproductive tissues, such as granulosa cells, uterine epithelial cells (UECs), uterine stromal cells (USCs), and oocytes were collected. Two human uterine cell lines were also used in this analysis. Reverse transcription-polymerase chain reaction tests, Western blotting, and immunofluorescence staining were performed. Serum starvation conditions were used to induce autophagy in primary cells.
Results
Atg9a and Atg9b were expressed in multiple mouse tissues and reproductive cells. Neither Atg9A nor Atg9B significantly changed in response to steroid hormones. Immunofluorescence staining of the UECs and USCs showed that ATG9A was distributed in a punctate-like pattern, whereas ATG9B exhibited a pattern of elongated tubular shapes in the cytoplasm. In human cancer cell lines, ATG9B was undetectable, whereas ATG9A was found in all cell types examined.
Autophagy, a self-digesting cellular process in metazoans, is a catabolic subcellular response used to produce the intracellular metabolites needed for cell survival [1]. During this process, macromolecules and organelles are enclosed within vesicular membranes that then fuse with lysosomes for degradation and recycling. The processed materials can be used as an energy source or building blocks for macromolecules [2,3]. While many proteins encoded by autophagy-related genes (Atg) and other factors participate in a series of sequential steps in autophagy, ATG9 is the only autophagy-related protein with transmembrane domains and lipid scrambling activity [4,5].
The lipid scrambling activity of ATG9 is required for the early steps of autophagosome formation in yeast [6]. Two isoforms of Atg9 have been reported in mammals: ATG9A and ATG9B (Figure 1A) [7]. These isoforms exhibit distinct expression patterns; whereas Atg9a is expressed in multiple tissues in mice, Atg9b is highly expressed in the placenta and at lower levels in the uterus and testis [7]. ATG9A is widely used in cell analyses to investigate the role of ATG9 in mammalian cells [8-10]. Mammalian ATG9A is localized to seed vesicles and expands autophagosomes, suggesting that it is involved in the initial stages of autophagosome formation. ATG9B is thought to function similarly during autophagy, but potentially in more restricted cell types [7].
In reproductive tissues, cells respond to changing environments and activate autophagy. We previously showed that autophagy is induced above basal levels in the uterus during hormonal deprivation [11] and in mouse oocytes during vitrification [12]. In the uterus, autophagy is involved in the turnover of vasoactive factors during dynamic tissue remodeling [11].
Several studies have reported changes in Atg9b expression under pathophysiological conditions. Dysregulation and mutations of ATG9B have been reported in human gastrointestinal cancers [13,14]. Reduced expression of Atg9b has also been observed in the hearts of aging mice [15]. Since Atg9b appears to exhibit more restricted patterns of expression in reproductive tissues [7], we investigated whether these isoforms are differentially expressed and regulated in the reproductive cells of mice. In the present study, we examined the expression of Atg9a and Atg9b in uteri, several primary cells, and oocytes. Herein, we report the distinct expression patterns of these two isoforms.
Institute of Cancer Research (ICR) mice were purchased from Raon Bio. The mice were housed in a semi-specific pathogen-free facility and maintained in accordance with the Institutional Animal Care and Use Committee (IACUC) guidelines until use. The mouse experiments were approved by the IACUC at Konkuk University (KU22030). To obtain uterine tissues from pregnant mice, 7-week-old ICR mice were injected with 2.5 IU of pregnant mare serum gonadotropin (PMSG) (Daesung Microbiological Labs Ltd.) and 2.5 IU of human chorionic gonadotropin (hCG) (Sigma-Aldrich) at 48-hour intervals. After the hCG injection, the female mice were caged with stud male mice for mating. Female mice with vaginal plugs the following morning were designated day 1 of pregnancy. Uteri were collected on days 1, 4, and 8 of pregnancy. The implantation sites from day 8 pregnant mice were separated into embryo-containing decidua and myometrium [16].
To examine the effects of steroid hormones on Atg9a and Atg9b, mice underwent ovariectomy (OVX) and rested for 10 to 12 days. The mice received a single injection of vehicle (sesame oil 0.1 mL; Sigma-Aldrich), progesterone (P) (1 mg/0.1 mL sesame oil; Sigma-Aldrich), or 17β-estradiol (E) (100 ng/0.1 mL sesame oil; Sigma-Aldrich) subcutaneously. The uteri were collected after 24 hours and subjected to RNA extraction or lysate preparation.
Four-week-old ICR mice were injected with 5 IU PMSG, and germinal vesicle (GV)-stage oocytes were collected from fully grown follicles 48 hours post-PMSG. To collect polar body (PB)-stage oocytes, PMSG-primed mice received an injection of 5 IU hCG at 48 hours, and ovulated oocytes were collected from the oviducts at 16 hours after hCG injection. One hundred oocytes were pooled from seven to eight mice and subjected to RNA extraction.
Seven-week-old ICR mice (three to five mice per group) received a single subcutaneous injection of E (100 ng/0.1 mL sesame oil) to increase epithelial cell proliferation. Twenty-four hours later, the uteri were collected, longitudinally incised, and cut into 3 to 4 mm long pieces. The uterine pieces were washed in phosphate-buffered saline (PBS) (Gibco, Thermo Fisher Scientific) and enzymatically treated with pancreatin (Sigma-Aldrich) and dispase (Gibco, Thermo Fisher Scientific) following a previously described protocol [17]. The uterine epithelial cells (UECs) in the pelleted fraction after centrifugation were filtered with a 70 μm nylon mesh filter (Corning, Sigma-Aldrich) and counted. The UECs were subjected to RNA extraction and immunofluorescence staining.
Ovaries were collected from 10 IU PMSG-primed 7-week-old mice (four mice per group). The isolated ovaries were treated with 0.5 M ethylene glycol tetraacetic acid (EGTA) solution (AG Scientific) for 15 minutes followed by incubation in hypertonic sucrose solution containing 0.5 M sucrose (Thermo Fisher Scientific), 0.2% bovine serum albumin (BSA) (Gibco, Thermo Fisher Scientific), and 1.8 mM EGTA in Dulbecco’s Modified Eagle Medium (DMEM/F12) (Gibco, Thermo Fisher Scientific) for 15 minutes. Large follicles were then punctured in DMEM/F12 containing 10% fetal bovine serum (FBS) (Gibco, Thermo Fisher Scientific) and 1% penicillin-streptomycin (Gibco, Thermo Fisher Scientific) to obtain granulosa cells (GCs). The cells were then centrifuged, washed with PBS, and subjected to reverse transcription-polymerase chain reaction (RT-PCR) and immunofluorescence staining.
AN3CA (human uterine adenocarcinoma cells) and T HESC (human telomerase reserve transcriptase)-immortalized uterine stromal cells (USCs; CRL-4003) were obtained from the American Type Culture Collection and cultured in DMEM/F-12 containing 10% FBS. The cells were grown to 80% confluence, either on 10-cm culture plates or in 6-well plates, for use in experiments.
Total RNA was extracted from oocytes, UECs, GCs, uterine lysates, and human uterine cells using TRIzol Reagent (Invitrogen, Thermo Fisher Scientific) following the manufacturer’s protocol. To separate DNA, RNA was treated with DNase (Promega Corp.) for 20 minutes at room temperature, then incubated at 75 °C for 10 minutes to inactivate the DNase. The RNA concentration and quality were assessed using a NanoDrop spectrophotometer (ND-1000; Thermo Fisher Scientific). Complementary DNA (cDNA) was synthesized from 2 μg of RNA by using random hexamer primers (Invitrogen) and moloney murine leukemia virus reverse transcriptase (Promega Corp.). Equal amounts of cDNA were used as templates for the PCR analysis. The primers used in this study are listed in Table 1, and their locations on the exons are indicated in Figure 1A (black arrows).
Each uterine sample was prepared from a single uterine horn of one mouse. The uterine horn was cut into small pieces and lysed in a radioimmunoprecipitation assay buffer: 10 mM Tris (pH 7.2; HanLAB), 150 mM NaCl (Fisher Scientific), 0.1% Triton X-100 (Sigma-Aldrich), 5 mM ethylenediaminetetraacetic acid (EDTA; HanLAB), 1% sodium dodecyl sulfate (Bio-Rad Laboratories Inc.), 1 mM dithiothreitol (Sigma-Aldrich), 1 mM phenylmethylsulfonyl fluoride (PMSF; MP Biomedicals), and 1X protease inhibitor (Roche). We used a Kinematica Polytron homogenizer (Kinematica AG), and centrifuged the samples at 15,928 ×g for 20 minutes at 4 °C. Protein concentrations were determined by bicinchoninic acid protein assay (Thermo Fisher Scientific). Lysates (10 μg) were loaded onto 10% sodium dodecyl sulfate-polyacrylamide gels and then transferred onto nitrocellulose membranes (Bio-Rad). After Western blotting, the chemiluminescence signals were detected using a West Femto kit (SuperSignal, Thermo Fisher Scientific). The intensity of the signal was normalized against the β-tubulin signal. The data were presented as mean±standard error of the mean. The primary antibodies used were rabbit polyclonal anti-ATG9A antibody (26276-1-AP; Proteintech Genomics), rabbit polyclonal anti-ATG9B (ab117591; Abcam), and rabbit polyclonal anti-β-tubulin antibody (ab6046; Abcam).
UECs and GCs were fixed with 4% paraformaldehyde (Biosesang) in PBS, permeabilized with 0.25% Triton X-100 (Sigma-Aldrich), and then blocked with 2% BSA in PBS for 1 hour. The primary antibodies used were rabbit polyclonal anti-ATG9A (26276-1-AP; Proteintech Genomics) and rabbit polyclonal anti-ATG9B (BS-4011R; Bioss Inc.) antibodies. The secondary antibody was Alexa Fluor 488-conjugated antibody (Invitrogen). The DNA was visualized using TO-PRO-3-iodide (Invitrogen). The cells were mounted on glass slides using Vectashield mounting medium (Vector Laboratories Inc.). The samples were observed with a confocal microscope (Zeiss LMS900), and images were obtained and analyzed using ZEN Blue software (Carl Zeiss).
The data were analyzed and plotted using GraphPad Prism ver. 5 software (GraphPad Software Inc.). The bands of Western blots were analyzed using ImageJ software (National Institutes of Health). All data were analyzed using the one-tailed t-test. Statistical significance is indicated in the figure legends by p<0.05, p<0.01, and p<0.001.
We first examined the expression of Atg9a and Atg9b in multiple tissues, including the liver, kidney, brain, lung, ovary, and uterus using RT-PCR. As shown in Figure 1B, the expression of both Atg9a and Atg9b was confirmed in all tissues examined. Although previous studies have suggested that Atg9b exhibits a restricted expression pattern [7], it also exhibits a wide distribution similar to Atg9a. The expression of these two genes was also confirmed in primary cells isolated from the ovary and uterus, including ovarian GCs, UECs, and USCs (Figure 1C). Oocytes in the GV and PB stages expressed both isoforms (Figure 1D).
During the first 8 days of pregnancy, the uterus undergoes dynamic changes in cell proliferation and differentiation. On day 1 of pregnancy, the uterus is influenced by estrogens, whereas on day 4, rising levels of P and E prepare the uterus for embryo implantation. On day 8, the stromal cells transform into decidual tissue around the developing embryo for support and protection [18]. As shown in Figure 2A, both ATG9A and ATG9B were observed in uterine tissues on all days of pregnancy. Notably, ATG9B appeared to be more prominent in the decidual tissues of day-8 implantation sites, while ATG9A levels were very low to undetectable throughout all days of pregnancy (Figure 2A).
To examine whether steroid hormones regulate the expression of ATG9A and ATG9B, we used an OVX model with steroid hormone injections. Based on RT-PCR analysis, Atg9a and Atg9b were expressed in all samples without notable variation (Figure 2B). Based on Western blotting, ATG9B appeared to be more abundant in the uterus than ATG9A. Neither protein exhibited fluctuations based on the type of hormone administered (Figure 2C), and there was no statistical significance among groups (data not shown).
We examined the subcellular localization of ATG9A and ATG9B in primary cells collected from ovaries and uteri. In cells and tissues, serum deprivation generally results in limited ambient nutrients, which leads to autophagy [19]. Three primary cell lines were used: ovarian GC, UEC, and USC. Under normal conditions (Figure 3, +FBS), all three primary cell lines exhibited scattered punctate patterns of ATG9A distribution. When the cells were serum-deprived for 6 hours (Figure 3, –FBS), the GCs and UECs exhibited increased ATG9A signal in punctate patterns in a subset of cells. A subpopulation of serum-deprived USCs exhibited a non-punctate pattern of cytoplasmic ATG9A localization.
The ATG9B signal was more responsive to serum starvation conditions in GCs, UECs, and USCs. As shown in Figure 4, ATG9B was abundant in GCs and exhibited a cytoplasmic punctate pattern. ATG9B also exhibited a strong cytoplasmic punctate pattern in the UECs. In USCs, the Atg9B signal was observed in an elongated tubular pattern. In all of the cells examined, serum starvation slightly increased the ATG9B signal.
We examined the expression of these two genes in human cell lines. AN3CA is a human uterine adenocarcinoma cell line of epithelial origin [20], and CRL-4003 is a human telomerase reverse transcriptase (hTERT)-immortalized USC line isolated from a myoma patient [21]. MCF7 is a widely used breast cancer cell line and is included for comparison. As shown in Figure 5A, ATG9A was expressed in AN3CA, CRL-4003, and MCF7 cell lines, whereas ATG9B expression was undetectable in all three cancer cell lines. Immunofluorescence staining indicated low levels of ATG9A expression in AN3CA and CRL-4003 cells, but it did not appear to respond to starvation conditions (Figure 5B).
The sources of phospholipids that produce membrane-enclosed autophagosomes have been extensively investigated over the years. It has become clear that phospholipid molecules are channeled from the endoplasmic reticulum and other membranous structures to growing autophagosomes via ATG2A [22]. When ATG2A delivers phospholipids to growing vesicles that contain ATG9, the ATG9 scrambles and redistributes these building blocks within the growing membrane [4,5]. The transport of lipid molecules between lipid bilayers is generally referred to as lipid transport, and various lipid translocases mediate this process. Among these, scramblases such as ATG9 transport lipid molecules bidirectionally without using ATP, whereas flippases and floppases move lipid molecules unidirectionally at the expense of ATP [23]. The identification of ATG9 as the sole lipid scramblase in the autophagic process has elucidated the mechanism underlying the initial growth of autophagosomes.
In mammals, two ATG9 isoforms have been identified: ATG9A and ATG9B. While it is generally considered that ATG9A is the main isoform, ATG9B has drawn the attention of reproductive biologists because of its skewed distribution in placental tissues and trophoblasts [7]. The lipid scrambling activity of ATG9 has been investigated using the ATG9A isoform in various studies, and it is generally assumed that ATG9B has a similar activity. However, a previous report on their disparate expression patterns led us to investigate the expression and regulation of Atg9A and Atg9B in mouse reproductive tissues.
Our results indicated that both Atg9a and Atg9b were expressed in GCs and primary uterine cells. Western blotting suggested that ATG9B was more abundant in the mouse uterus (Figure 2). As previously reported [24], mice also express Atg9 isoforms. In mouse primary cells, ATG9A and ATG9B responded to serum starvation and exhibited increased signals (Figures 3 and 4). While many autophagy marker proteins exhibit scattered punctate patterns of subcellular localization, ATG9B exhibited a unique elongated tubular pattern in USCs (Figure 4). ATG9-positive cytoplasmic structures have been observed in elongated tubular patterns, as Atg9 can be localized to Golgi membranes during fragmentation under serum starvation conditions [25]. Our observations reflect a similar phenomenon in serum-starved USCs.
The uterus is a versatile organ that undergoes cyclic vascular changes [26]. Autophagy is continuously monitored to meet nutritional needs and to remove metabolic waste. We previously showed that autophagy is required for the turnover of vasoactive factors during cyclical changes [11], and that OVX to remove the source of steroid hormones increases the autophagic rate [16]. Autophagic activation also increases in the uterus on day 1 of pregnancy, when there is a heightened inflammatory response after mating [16]. Autophagic flux is commonly assessed based on the ATG8 (Microtubule-associated protein 1A/1B-light chain 3 [LC3]) lipidation status. Our results showed that the levels of Atg9A and Atg9B were not influenced by differences in the hormonal status of the uterus or days of pregnancy (Figure 2). This may be because ATG9 proteins are involved in the initial autophagic process, and their levels per se do not reflect the active autophagic flux observed in ATG8 lipidation.
In the three human cell lines used to analyze the expression of ATG9 isoforms, ATG9B was undetectable, whereas the expression of ATG9A was readily detectable (Figure 5). ATG9B and other ATG genes are downregulated in sporadic breast carcinoma by aberrant methylation in their promoter regions [27]. As epigenetic silencing of specific genes is a widespread phenomenon in various cancers [28], ATG9B may be silenced by similar mechanisms in AN3CA and MCF7 cancer cells. The absence of ATG9B was also noted in CRL-4003, a hTERT-immortalized USC line. Further investigation is warranted to elucidate the mechanisms underlying ATG9B silencing.
Our work shows, for the first time, that both Atg9a and Atg9b are present in mouse reproductive tissues. To gain insight into the autophagy pathway and its role in cellular homeostasis, it is essential to understand the mechanism of autophagy initiation as mediated by ATG9 in reproductive tissues.
Notes
Author contributions
Conceptualization: HJL, HS. Methodology: ML, SS. Formal analysis: ML, SS, HJL. Data curation: ML, SS, HJL. Funding acquisition: HJL. Visualization: ML, SS. Software: ML, SS. Validation: ML, SS, HJL, HS. Investigation: ML, SS, HJL, HS. Writing-original draft: ML, SS. Writing-review & editing: HJL, HS. Approval of final manuscript: ML, SS, HJL, HS.
Figure 1.
Expression of Atg9a and Atg9b in multiple tissues and reproductive cells in mice. (A) Gene structures of the mouse Atg9a and Atg9b. They are encoded by two distinct genes on different chromosomes. Locations of the primers used in this study are marked with arrows. Green square, exon; black line, intron. The total number of amino acids in each protein is shown at the right. (B, C, D) Expression of Atg9a and Atg9b mRNAs in multiple tissues, primary cells from the ovary and uterus, and oocytes (germinal vesicle [GV]-stage and polar body [PB]-stage). Atg, autophagy-related gene; L, liver; K, kidney; B, brain; Lu, lung; O, ovary; U, uterus; –, no reverse transcription; GC, granulosa cell; UEC, uterine epithelial cell; USC, uterine stromal cell; Rpl7, ribosomal protein L7.
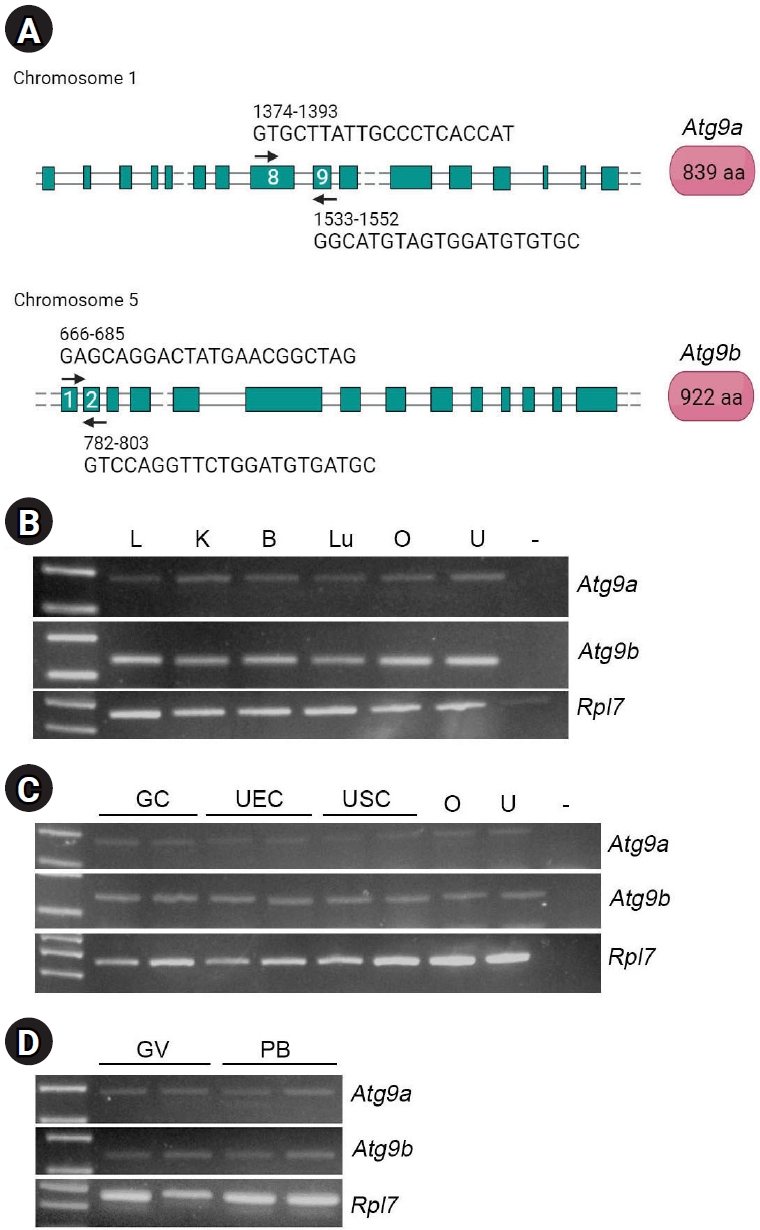
Figure 2.
Expression analysis of ATG9A and ATG9B in mouse uteri during the peri-implantation period and in ovariectomized mice. (A) Uterine lysates from days 1, 4, and 8 of pregnancy were subjected to Western blot analysis. Day 8 implantation sites were separated into two groups: the decidua containing an implanted embryo and surrounding decidual tissue (Decidua) and the myometrium (Myo). Brain (B) served as a positive control, and β-tubulin (TUBB), as a loading control. (B) Reverse transcription-polymerase chain reaction analysis of Atg9a and Atg9b in the uteri of mice who underwent ovariectomy (OVX) and were injected with steroid hormones. After OVX, mice were injected with sesame oil (vehicle, 0.1 mL), progesterone (P, 1 mg/0.1 mL vehicle), or 17β-estradiol (E, 100 ng/0.1 mL vehicle). Twenty-four hours after injection, the uterus from each mouse was collected and subjected to RNA extraction. For each group, two to three individual samples were used. (C) Western blotting of ATG9A and ATG9B in the uteri of vehicle-, P-, and E-injected mice. Both ATG9A and ATG9B are expressed in all samples regardless of hormonal treatments, suggesting that their levels are not influenced by ovarian steroid hormone. ATG, autophagy-related gene.
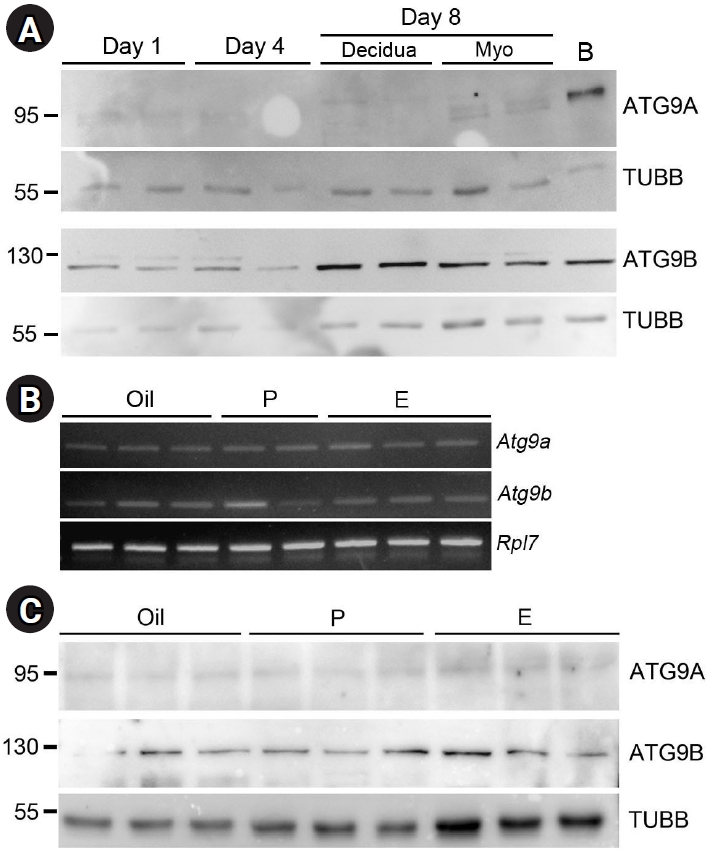
Figure 3.
Immunofluorescence staining of ATG9A in primary cells from mouse ovary and uterus. Immunofluorescence staining was performed using a rabbit polyclonal antibody against ATG9A and probed with Alexa Fluor 488-conjugated secondary antibodies (green) and TO-PRO-3-iodide to counterstain the DNA (blue). Confocal microscopy was used to obtain the images. Cells were incubated in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) or without FBS (serum starvation) for 6 hours. Rabbit immunoglobulin G (IgG) was used as a negative control. White scale bar, 20 µm. ATG, autophagy-related gene; GC, granulosa cell; UEC, uterine epithelial cell; USC, uterine stromal cell.

Figure 4.
Immunofluorescence staining of ATG9B in primary cells from mouse ovary and uterus. Immunofluorescence staining was performed using a rabbit polyclonal antibody against ATG9B and probed with Alexa Fluor 488-conjugated secondary antibodies (green) and TO-PRO-3-iodide to counterstain the DNA (blue). Confocal microscopy was used to obtain the images. Cells were incubated in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS) or without FBS (serum starvation) for 6 hours. Rabbit immunoglobulin G (IgG) was used as a negative control. White scale bar, 20 µm. ATG, autophagy-related gene; GC, granulosa cell; UEC, uterine epithelial cell; USC, uterine stromal cell.

Figure 5.
Expression of ATG9A and ATG9B in human cell lines. (A) Reverse transcription-polymerase chain reaction analysis of ATG9A and ATG9B in AN3CA (a human endometrial adenocarcinoma cell line) and CRL-4003 (a human telomerase reverse transcriptase [hTERT]-immortalized human uterine stromal cell line). MCF7 (a human breast cancer cell line) was also used for comparison. (B) Immunofluorescence staining of ATG9A in AN3CA and CRL-4003 cells. White scale bar, 20 µm. ATG, autophagy-related gene; FBS, fetal bovine serum; IgG, immunoglobulin G.
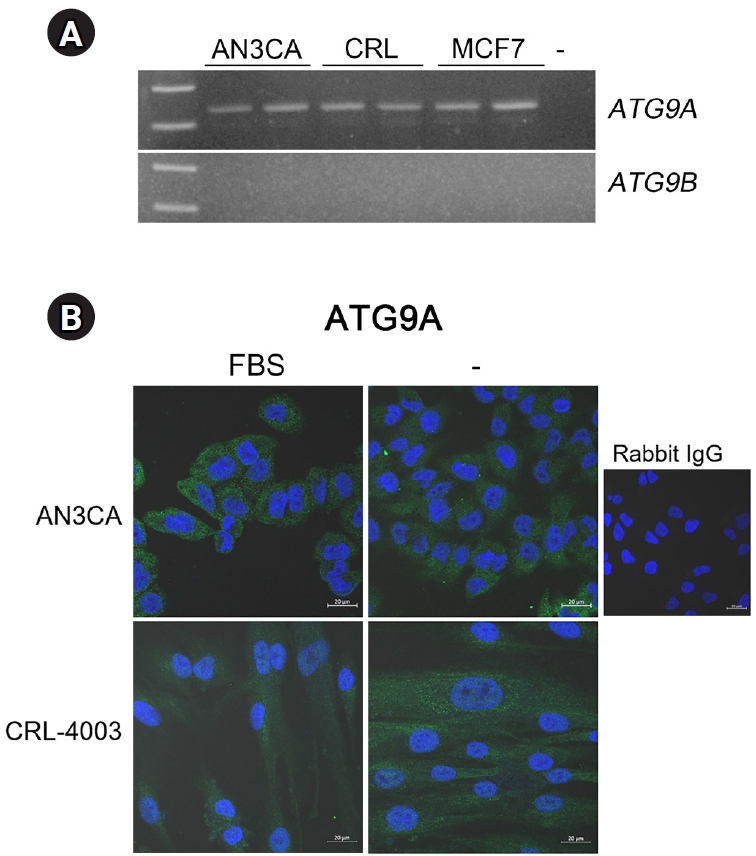
Table 1.
Primers Used for Polymerase Chain Reaction Analyses
References
1. Lum JJ, DeBerardinis RJ, Thompson CB. Autophagy in metazoans: cell survival in the land of plenty. Nat Rev Mol Cell Biol 2005;6:439-48.



2. Mizushima N, Levine B. Autophagy in mammalian development and differentiation. Nat Cell Biol 2010;12:823-30.




3. Mizushima N. A brief history of autophagy from cell biology to physiology and disease. Nat Cell Biol 2018;20:521-7.



4. Maeda S, Yamamoto H, Kinch LN, Garza CM, Takahashi S, Otomo C, et al. Structure, lipid scrambling activity and role in autophagosome formation of ATG9A. Nat Struct Mol Biol 2020;27:1194-201.




5. Matoba K, Kotani T, Tsutsumi A, Tsuji T, Mori T, Noshiro D, et al. Atg9 is a lipid scramblase that mediates autophagosomal membrane expansion. Nat Struct Mol Biol 2020;27:1185-93.



6. Yamamoto H, Kakuta S, Watanabe TM, Kitamura A, Sekito T, Kondo-Kakuta C, et al. Atg9 vesicles are an important membrane source during early steps of autophagosome formation. J Cell Biol 2012;198:219-33.




7. Yamada T, Carson AR, Caniggia I, Umebayashi K, Yoshimori T, Nakabayashi K, et al. Endothelial nitric-oxide synthase antisense (NOS3AS) gene encodes an autophagy-related protein (APG9-like2) highly expressed in trophoblast. J Biol Chem 2005;280:18283-90.


8. Mailler E, Guardia CM, Bai X, Jarnik M, Williamson CD, Li Y, et al. The autophagy protein ATG9A enables lipid mobilization from lipid droplets. Nat Commun 2021;12:6750.




9. Broadbent DG, Barnaba C, Perez GI, Schmidt JC. Quantitative analysis of autophagy reveals the role of ATG9 and ATG2 in autophagosome formation. J Cell Biol 2023;222:e202210078.




10. Olivas TJ, Wu Y, Yu S, Luan L, Choi P, Guinn ED, et al. ATG9 vesicles comprise the seed membrane of mammalian autophagosomes. J Cell Biol 2023;222:e202208088.



11. Lee B, Shin H, Oh JE, Park J, Park M, Yang SC, et al. An autophagic deficit in the uterine vessel microenvironment provokes hyperpermeability through deregulated VEGFA, NOS1, and CTNNB1. Autophagy 2021;17:1649-66.



12. Bang S, Shin H, Song H, Suh CS, Lim HJ. Autophagic activation in vitrified-warmed mouse oocytes. Reproduction 2014;148:11-9.


13. Kang MR, Kim MS, Oh JE, Kim YR, Song SY, Kim SS, et al. Frameshift mutations of autophagy-related genes ATG2B, ATG5, ATG9B and ATG12 in gastric and colorectal cancers with microsatellite instability. J Pathol 2009;217:702-6.



14. Gil J, Karpinski P, Sasiadek MM. Transcriptomic profiling for the autophagy pathway in colorectal cancer. Int J Mol Sci 2020;21:7101.



15. Liang W, Moyzis AG, Lampert MA, Diao RY, Najor RH, Gustafsson AB. Aging is associated with a decline in Atg9b-mediated autophagosome formation and appearance of enlarged mitochondria in the heart. Aging Cell 2020;19:e13187.




16. Choi S, Shin H, Song H, Lim HJ. Suppression of autophagic activation in the mouse uterus by estrogen and progesterone. J Endocrinol 2014;221:39-50.


17. Chung D, Das SK. Mouse primary uterine cell coculture system revisited: ovarian hormones mimic the aspects of in vivo uterine cell proliferation. Endocrinology 2011;152:3246-58.




18. Cha J, Lim H, Dey SK. Embryo implantation. In: Plant TM, Zeleznik AJ, editors. Knobil and Neill's physiology of reproduction. 4th ed. Elsevier; 2015. p. 1697-739.
20. Lim HJ, Moon I, Han K. Transcriptional cofactors exhibit differential preference toward peroxisome proliferator-activated receptors alpha and delta in uterine cells. Endocrinology 2004;145:2886-95.


21. Ono YJ, Terai Y, Tanabe A, Hayashi A, Hayashi M, Yamashita Y, et al. Decorin induced by progesterone plays a crucial role in suppressing endometriosis. J Endocrinol 2014;223:203-16.



22. Maeda S, Otomo C, Otomo T. The autophagic membrane tether ATG2A transfers lipids between membranes. Elife 2019;8:e45777.




23. Matoba K, Noda NN. Secret of Atg9: lipid scramblase activity drives de novo autophagosome biogenesis. Cell Death Differ 2020;27:3386-8.




24. Jin X, Wang K, Wang L, Liu W, Zhang C, Qiu Y, et al. RAB7 activity is required for the regulation of mitophagy in oocyte meiosis and oocyte quality control during ovarian aging. Autophagy 2022;18:643-60.



25. Takahashi Y, Meyerkord CL, Hori T, Runkle K, Fox TE, Kester M, et al. Bif-1 regulates Atg9 trafficking by mediating the fission of Golgi membranes during autophagy. Autophagy 2011;7:61-73.



26. Massri N, Loia R, Sones JL, Arora R, Douglas NC. Vascular changes in the cycling and early pregnant uterus. JCI Insight 2023;8:e163422.










