|
 |
- Search
| Clin Exp Reprod Med > Volume 51(1); 2024 > Article |
|
Abstract
Objective
The purpose of this study was to identify factors associated with twin pregnancy following day 3 double embryo transfer (DET).
Methods
This retrospective cohort study incorporated data from 16,972 day 3 DET cycles. The participants were women aged between 18 and 45 years who underwent in vitro fertilization with intracytoplasmic sperm injection (IVF/ICSI) at My Duc Assisted Reproduction Technique Unit (IVFMD), My Duc Hospital, located in Ho Chi Minh City, Vietnam.
Results
Of the 16,972 day 3 DET cycles investigated, 8,812 (51.9%) resulted in pregnancy. Of these, 6,108 cycles led to clinical pregnancy, with 1,543 (25.3% of clinical pregnancies) being twin pregnancies. Factors associated with twin pregnancy included age under 35 years (odds ratio [OR], 1.5; 95% confidence interval [CI], 1.32 to 1.71; p<0.001) and cycles involving the transfer of at least one grade I embryo. Relative to the transfer of two grade III embryos, the risk of twin pregnancy was significantly elevated following the transfer of two grade I embryos (OR, 1.40; 95% CI, 1.16 to 1.69; p<0.001) or a combination of one grade I and one grade II embryo (OR, 1.27; 95% CI, 1.05 to 1.55; p=0.001).
Twin pregnancy is associated with a variety of adverse neonatal and obstetric outcomes [1,2] and has been shown to impose a psychological and economic burden on families and society [3]. Since the 1980s, the global twin birth rate has increased by approximately one-third, from 9.1 to 12.0 sets of twins per 1,000 live births. This equates to about 1.6 million twins born each year [4].
Most twin pregnancies are related to the use of assisted reproductive technology [5]. In 2016, Mancuso et al. [6] proved elective single embryo transfer (eSET) could be an solution to minimise multiple births while maintaining live birth rates comparable to those achieved double embryo transfers. However, the adoption of day 3 eSET varies across countries [7,8] and is often less common than day 3 double embryo transfer (DET) due to concerns that the eSET approach may decrease implantation and live birth rates [9]. For each transfer, eSET has been found to yield a live birth rate approximately one-third of that following DET [10].
To ensure positive treatment outcomes without increasing the risk of multiple pregnancy following day 3 DET, it is important to identify individuals who are at increased risk of twin pregnancy. Such identification could facilitate the development of a personalized embryo transfer strategy. In frozen embryo transfer (FET) cycles, available data suggest that certain embryo-specific factors and cycle characteristics may be associated with twin pregnancy after day 3 DET. Kaser et al. [11] identified six factors associated with live twin delivery in frozen-thawed day 3 DET. These included a patient age of less than 35 years at the time of cryopreserved embryo transfer, intact survival of the lead embryo, resumption of mitosis, the presence of seven or eight viable cells in the non-lead embryo, the transfer of a lead embryo with seven or more cells, and a total of 14 or more viable cells in the two transferred embryos [11]. In fresh day 3 DET cycles, young age and the transfer of good-quality embryos were found to relate to the occurrence of two gestational sacs [12,13]. A retrospective Dutch cohort study reported that a height greater than 1.74 m and the retrieval of eight or more oocytes were independent factors associated with an increased live twin delivery rate following day 3 DET [14].
The objective of this study was to identify factors associated with clinical twin pregnancy following day 3 DET in Vietnamese women undergoing IVF/intracytoplasmic sperm injection (ICSI).
This retrospective cohort study was conducted at My Duc Assisted Reproduction Technique Unit (IVFMD), My Duc Hospital, located in Ho Chi Minh City, Vietnam. The study was approved by the medical ethics committee of the My Duc Hospital (19/2021/MD-BVMD) on December 9, 2021. Informed consent was waived by the board.
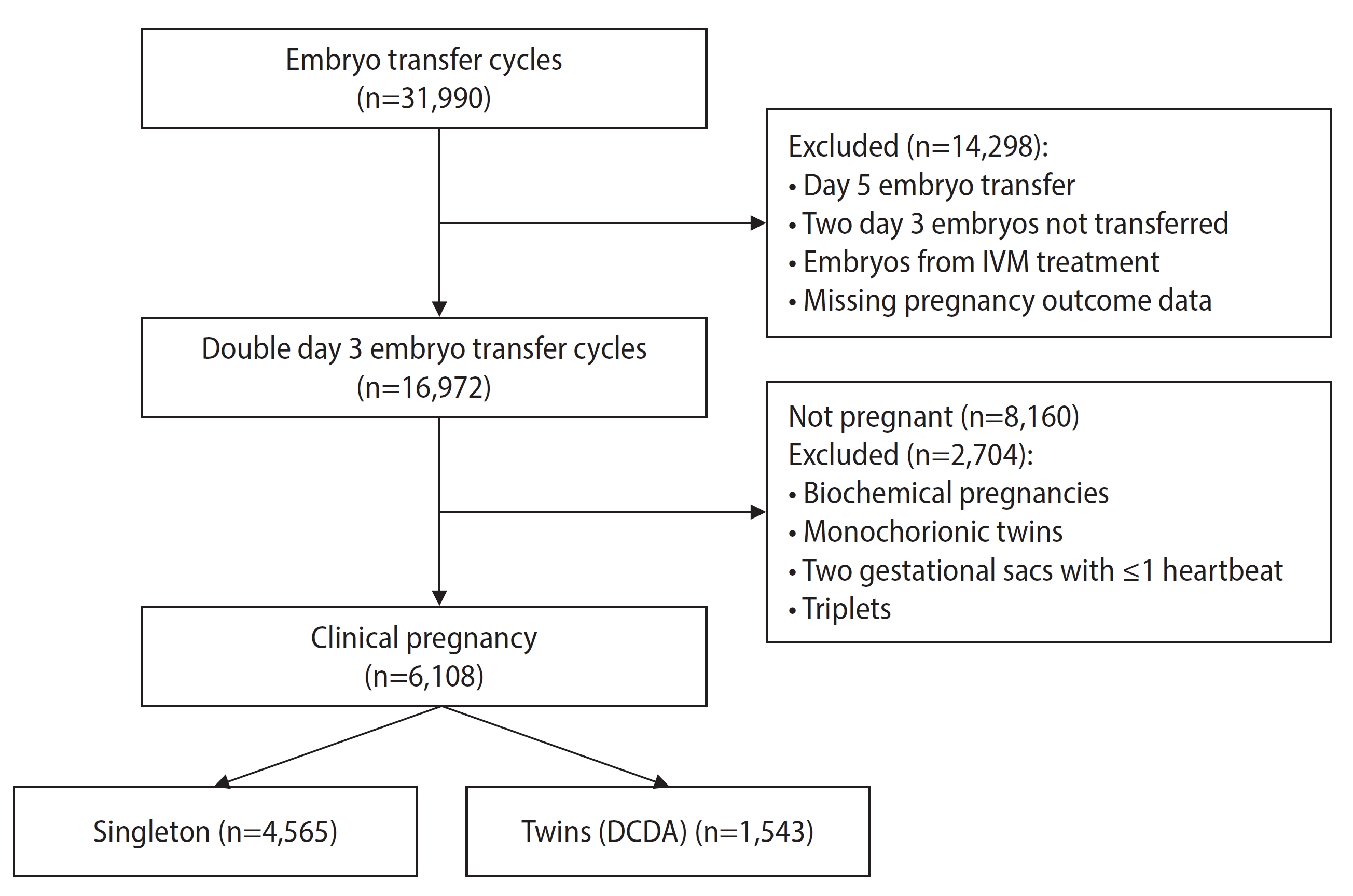
Data were extracted from the digital data management system of the clinic. All women who underwent IVF/ICSI and day 3 DET between January 2016 and September 2021 were evaluated for eligibility. Cycles involving blastocyst transfer, embryos resulting from in vitro maturation treatment, and women with uterine abnormalities, untreated hydrosalpinx, or missing pregnancy information were excluded. Biochemical pregnancies, monochorionic twins, instances of two gestational sacs with only one fetal heartbeat, and triplet or higher-order multiple pregnancies were also excluded from the analysis.
All patients underwent ovarian stimulation using a gonadotropin-releasing hormone antagonist protocol. Follicle-stimulating hormone (FSH) was administered for 5 days beginning on the 2nd or 3rd day of the menstrual cycle. The initial FSH dose was tailored for each patient based on anti-M├╝llerian hormone levels. Further dosage adjustments were made according to the clinical judgment of the treating physician. Follicular development was monitored using ultrasonography, as well as through measurement of serum estradiol and progesterone levels. Once the mean diameter of at least two leading follicles reached 17 mm, a 250-╬╝g dose of recombinant human chorionic gonadotropin (Ovitrelle; Merck Serono) was administered to trigger oocyte maturation. Oocyte retrieval was performed 36 hours later. Insemination was conducted using ICSI, 39 to 41 hours after triggering. Fertilization was assessed 16 to 18 hours after insemination. The day 3 embryos were evaluated and graded at a mean┬▒standard deviation of 66┬▒1 hours after fertilization, in accordance with the Istanbul consensus [15].
For fresh embryo transfer cycles, two day 3 embryos were transferred into the uterus under ultrasonographic guidance, 3 days after oocyte retrieval. For FET cycles, the endometrium was prepared using 6 to 8 mg/day of oral estradiol valerate starting on the 2nd or 3rd day of the menstrual cycle. The endometrial thickness was monitored from day 6 onwards, and vaginal progesterone was initiated when the thickness reached at least 7 mm. Two embryos were thawed on the day of embryo transfer, which was 3 days after the initiation of progesterone.
Endocrine support was administered through exogenous estradiol and vaginal progesterone, which was continued until week 11 of gestation. Pregnancy was tested by measuring the serum human chorionic gonadotropin level 2 weeks after embryo transfer. If the test was positive, gestational ultrasound scans were performed at 7 and 12 weeks of gestation.
The primary outcome examined was twin pregnancy following day 3 DET. A twin pregnancy was considered to be indicated by the presence of two gestational sacs, each containing an embryo and exhibiting a heartbeat, on ultrasound at 7 weeks of gestation.
Baseline data are presented using descriptive statistics, with the mean┬▒standard deviation used for normally distributed variables and the median and interquartile range used for variables with a skewed distribution. Categorical data are represented as a number and percentage. Eligible cycles were divided into two groups based on whether a singleton or twin clinical pregnancy was detected on ultrasound at 7 weeks of gestation. The differences between groups were analyzed using the Student t-test for variables with a normal distribution, the Mann-Whitney U test for variables with a skewed distribution, and the chi-square test for categorical variables. Univariate and multivariate logistic regression analyses were conducted to identify factors associated with twin pregnancy. All variables with a p-value of <0.05 in the univariate analysis were included in the multivariable analysis.
Between January 2016 and September 2021, a total of 16,972 cycles involving day 3 DET were screened, with 8,812 (51.9%) resulting in pregnancy. Of these, 6,108 cycles led to clinical pregnancy and were eligible for analysis, 1,543 of which (25.3%) involved twin pregnancies (Figure 1). The study population was relatively young, with a mean age of 33.81┬▒4.79 years, and lean, with a mean body mass index (BMI) of 21.32┬▒2.55 kg/m2 (Table 1). Anti-M├╝llerian hormone levels were comparable between the patients with singleton and twin pregnancies (3.18 ng/dL vs. 3.38 ng/dL, p=0.501; respectively) (Table 1).
In the multivariate analysis, independent predictors of twin pregnancy included being under 35 years of age at the time of IVF/ICSI, the transfer of two grade I embryos, or the transfer of one grade I embryo along with one grade II embryo (Table 2). BMI did not independently predict twin pregnancy (Table 2).
This large-scale retrospective study revealed that an age of under 35 years at the time of IVF/ICSI and the transfer of one or more good-quality (grade I) embryos were independently associated with a heightened risk of twin pregnancy following day 3 DET. These findings align with data from previous studies. An age of under 35 years has been reported to be associated with an elevated rate of live twin delivery in FET cycles [11]. Furthermore, relatively young age and the transfer of good-quality embryos have been linked to an increased risk of double gestational sacs following day 3 DET in fresh embryo transfer cycles [12,13]. However, these prior studies either did not categorize twin delivery as dichorionic diamniotic [11] or reported twin pregnancy based only on the presence of two gestational sacs on ultrasound, without confirming the existence of two fetal hearts inside the sacs [12,13]. In contrast, our study employed a clear definition for twin pregnancy: the presence of two gestational sacs, each containing an embryo and a fetal heart, as confirmed by ultrasound at the 7th week of gestation. Furthermore, our study is based on the largest retrospective dataset reported to date.
Following natural conception, both greater maternal height and maternal BMI are associated with a higher rate of fraternal twins. This is hypothesized to result from elevated FSH levels in obese women, which can lead to multi-follicular development and multi-ovulation [16,17]. In the context of IVF cycles, research has suggested that women of taller stature (>1.74 m) or greater body weight (>60 kg) have a higher likelihood of conceiving twins following day 3 DET [13,14]. In the present study, no significant differences were found in the height or weight of women who experienced twin pregnancies compared to those with singleton pregnancies. This was determined through multivariable logistic regression analysis, with height and weight as two independent variables, and remained the case when these two variables were replaced with BMI. Notably, however, the women in our study did not have a high BMI, and no significant BMI difference was found between the twin and singleton pregnancy groups. As such, the nature of the dataset may have limited our ability to clarify the influence of these factors on the risk of twin pregnancy.
A key limitation of this study is its retrospective design, which carries the potential for confounding factors to influence the results. Furthermore, the determination of twin pregnancy was based solely on clinical pregnancy at 7 weeks of gestation, without information regarding the rates of singleton and twin live births. Nonetheless, the studyŌĆÖs key strengths lie in its large sample size and the uniformity of data, which were sourced from a single center employing the same embryo culture and freezing process. Regular assessments ensured minimal variability in embryo evaluation among embryologists and reduced confounding due to asynchrony between embryo transfer cycles.
Numerous centers worldwide have begun implementing an eSET policy to decrease the rate of twin pregnancies associated with IVF treatment [18]. By identifying factors linked to twin pregnancy and establishing an appropriate embryo transfer policy for those at higher risk of conceiving twins, twin pregnancy rates could be lowered while maintaining favorable clinical outcomes for couples undergoing IVF. The findings of the current study indicate that patient age and the transfer of two good-quality embryos (either two grade I or one grade I and one grade II) were predictors of twin pregnancy in DET cycles. These findings could assist medical professionals in tailoring treatment strategies for couples with infertility. Future prospective studies are required to confirm whether transferring a single day 3 embryo to women with risk factors for twin pregnancy enhances IVF outcomes and reduces twin pregnancy rates.
Notes
Author contributions
Conceptualization: DLN, HLTH, TMH, LNV. Data curation: DLN, HLTH, NTN, VTTT. Formal analysis: DLN, VNAH, TDP. Funding acquisition: TMH. Methodology: HLTH, LNV. Project administration: DLN, HLTH. Visualization: DLN. Writing-original draft: DLN, HLTH, VNAH. Writing-review & editing: DLN, HLTH, VNAH, LNV.
Acknowledgments
We extend our gratitude to the staff of My Duc Assisted Reproduction Technique Unit (IVFMD), My Duc Hospital for their assistance. Additionally, we express our appreciation to all of the HOPE Research Center members who contributed to this research. Finally, this paper is dedicated to the memory of Hieu, our beloved friend and colleague, who designed this study.
Table┬Ā1.
Baseline characteristics of women included in the analysis
Table┬Ā2.
Univariate and multivariate logistic regression analysis of factors associated with the risk of twin pregnancy following double day 3 embryo transfer
References
1. Practice Committee of American Society for Reproductive Medicine. Multiple gestation associated with infertility therapy: an American Society for Reproductive Medicine Practice Committee opinion. Fertil Steril 2012;97:825-34.


2. Santana DS, Silveira C, Costa ML, Souza RT, Surita FG, Souza JP, et al. Perinatal outcomes in twin pregnancies complicated by maternal morbidity: evidence from the WHO Multicountry Survey on Maternal and Newborn Health. BMC Pregnancy Childbirth 2018;18:449.




3. Miller V, Diamond MP, Hansen KR, Steiner AZ, Cedars M, Legro RS, et al. Families with children resulting from ART: psychosocial and financial implications. Hum Reprod Open 2020;2020:hoaa010.




4. Monden C, Pison G, Smits J. Twin peaks: more twinning in humans than ever before. Hum Reprod 2021;36:1666-73.




5. De Geyter C, Calhaz-Jorge C, Kupka MS, Wyns C, Mocanu E, Motrenko T, et al. ART in Europe, 2014: results generated from European registries by ESHRE: the European IVF-monitoring Consortium (EIM) for the European Society of Human Reproduction and Embryology (ESHRE). Hum Reprod 2018;33:1586-601.

6. Mancuso AC, Boulet SL, Duran E, Munch E, Kissin DM, Van Voorhis BJ. Elective single embryo transfer in women less than age 38 years reduces multiple birth rates, but not live birth rates, in United States fertility clinics. Fertil Steril 2016;106:1107-14.



7. Practice Committee of the American Society for Reproductive Medicine and the Practice Committee for the Society for Assisted Reproductive Technologies. Guidance on the limits to the number of embryos to transfer: a committee opinion. Fertil Steril 2021;116:651-4.


8. Maheshwari A, Griffiths S, Bhattacharya S. Global variations in the uptake of single embryo transfer. Hum Reprod Update 2011;17:107-20.


9. Kamath MS, Mascarenhas M, Kirubakaran R, Bhattacharya S. Number of embryos for transfer following in vitro fertilisation or intra-cytoplasmic sperm injection. Cochrane Database Syst Rev 2020;8:CD003416.


10. Roberts SA, McGowan L, Mark Hirst W, Vail A, Rutherford A, Lieberman BA, et al. Reducing the incidence of twins from IVF treatments: predictive modelling from a retrospective cohort. Hum Reprod 2011;26:569-75.


11. Kaser DJ, Missmer SA, Correia KF, Ceyhan ST, Hornstein MD, Racowsky C. Predictors of twin live birth following cryopreserved double embryo transfer on day 3. J Assist Reprod Genet 2013;30:1023-30.




12. Abdollahi M, Omani Samani R, Hemat M, Arabipoor A, Shabani F, Eskandari F, et al. Factors that influence the occurrence of multiple pregnancies after intracytoplasmic injection cycles with two or three fresh embryo transfers. Int J Fertil Steril 2017;11:191-6.


13. Kim MS, Kim JH, Jee BC, Suh CS, Kim SH. Factors affecting occurrence of twin pregnancy after double embryo transfer on day 3. J Obstet Gynaecol Res 2015;41:1223-8.



14. Groeneveld E, Lambers MJ, Stakelbeek ME, Mooij TM, van den Belt-Dusebout AW, Heymans MW, et al. Factors associated with dizygotic twinning after IVF treatment with double embryo transfer. Hum Reprod 2012;27:2966-70.


15. Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology. The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod 2011;26:1270-83.


16. Basso O, Nohr EA, Christensen K, Olsen J. Risk of twinning as a function of maternal height and body mass index. JAMA 2004;291:1564-6.

17. Reddy UM, Branum AM, Klebanoff MA. Relationship of maternal body mass index and height to twinning. Obstet Gynecol 2005;105:593-7.


18. Gingold JA, Fazzari M, Gerber R, Kappy M, Goodman M, Lieman H, et al. Adherence to embryo transfer guidelines in favorable-prognosis patients aged less than 35 years using autologous oocytes and in recipients using donor oocytes: a Society for Assisted Reproductive Technology Clinic Outcome Reporting System study. Fertil Steril 2022;117:548-59.