 |
 |
- Search
| Clin Exp Reprod Med > Volume 50(1); 2023 > Article |
|
Abstract
Objective
Sleep deprivation (SD) is a common problem in todayŌĆÖs stressful lifestyle and have physiological consequences, including reproductive dysfunction and infertility. As an antioxidant, olive oil may be effective in reducing testicular and spermatological damage by decreasing the production of free radicals.
Methods
This study investigated the effects of olive oil on sperm quality and testicular structure using stereological methods to assess rats with SD.
Results
When comparing SD group to grid floor+distilled water (GR) group, we found that the sperm count and motility, as well as the percentage of slow progressive sperm was significantly lower in SD group (p<0.05), but the percentage of immotile sperm was higher (p<0.01). However, no improvement was observed in sperm count or motility after concomitant treatment of SD group with olive oil. Stereological examinations revealed no significant change in the total volumes of the seminiferous tubules, interstitial tissue, and germinal epithelium in the study groups. Conversely, the total number of testicular cell types was significantly lower in SD group than in GR group. Although the total number of Sertoli and Leydig cells was significantly higher in the SD+olive oil group than in the untreated SD group, no significant difference in the total number of other testicular cell types was observed between the two groups.
Current lifestyle factors and psychological stress contribute to sleep deprivation (SD), a common condition today [1]. SD has been associated with changes in the immune [2] and neuroendocrine systems [3] as well as with memory deficiency [4,5]. SD, as a harmful stressor, can also affect the structure and function of the male reproductive system. A previous study found a correlation between SD and disruption in the secretion of gonadotropin-releasing hormone, which plays a critical role in the reproductive system and fertility [6]. Sleep disorders, including insufficient sleep syndrome, obstructive sleep apnea syndrome, shift work disorder, and insomnia, have also been found to affect testosterone levels [7] and semen quality [8], which impact male fertility. Sleep disorders can also lead to urinary and sexual dysfunction, such as erectile dysfunction [9]. In addition, night sleep duration has been shown to affect sexual behavior [10]. The prevalence of SD and its complications, especially sexual dysfunction, has been a major concern in recent years. SD is also responsible for a significant induction of reactive oxygen species and production of free radicals [11]. Since testicular cell membranes are rich in polyunsaturated fatty acids, they are vulnerable to oxidative stress [12].
Olive oil is composed of monounsaturated fatty acids (e.g., oleic acid), saturated fatty acids, polyunsaturated fatty acids, and alpha-tocopherol [13]. Olive oil also possesses several bioactive compounds, including polyphenols, flavonoids, and other bioactive organic molecules (e.g., squalene) [14]. Virgin olive oil has antioxidant properties due to its high phenolic compound content [15]. Olive oil has anti-inflammatory, antithrombotic, anticancer, and vasodilator effects [14,16] and has been reported to exert beneficial effects on reproductive capability [17]. Olive oil has been shown to lessen testicular and spermatological damage by reducing the production of free radicals [18].
Based on this information, the present study investigated the protective effects of olive oil on sperm quality and quantitative changes in testicular structure in adult male rats with SD. To examine this hypothesis, unbiased stereological methods were used to obtain quantitative, reliable, and comparable data.
Thirty male Sprague-Dawley rats (210ŌĆō270 g) were purchased from the Animal Laboratory Center of Shiraz University of Medical Sciences (SUMS). All animal procedures were performed under the standard rules established by the Animal Care and Ethics Committee of SUMS (No. 94-7624). The animals were kept under standard conditions at room temperature (25┬░C┬▒2┬░C) with normal humidity and 12-hour light-dark cycles. They also had free access to standard food and water. The rats were randomly divided into six groups (n=5), including control (CON), grid floor+distilled water (GR), olive oil (OO), grid floor+olive oil (GR+OO), sleep deprivation+distilled water (SD), and sleep deprivation+olive oil (SD+OO). The animals in the sham and olive oil groups, respectively, received 1.5 mL of distilled water and 1.5 mL of olive oil by daily oral gavage for 21 days.
In this study, a modified plastic multi-platform box measuring 125├Ś45├Ś45 cm3 was used to induce SD. The box was made of 14 small cylindrical platforms (6.5 cm in diameter and 8.0 cm in height), which were located on the box floor. The platform box was filled with water at a temperature of 22┬░C┬▒2┬░C. The SD protocol was set for 18 hours per day for 21 days [19,20]. Concurrently, rats in the GR groups were placed on wire mesh grids in the same box as controls for the SD animals.
The tail of the epididymis was removed, cleaned quickly, then placed in a petri dish containing 3 mL of phosphate-buffered saline (PBS). The petri dish was put on a hot plate at 37┬░C until the semen diffused into the PBS solution. The diluted semen was gently shaken to achieve uniform suspension and to spread the spermatozoa [21].
The diluted semen was spread on a hemocytometer (a Neubauer chamber) and the heads of spermatozoa were counted manually using a light microscope at a final magnification of ├Ś400. Approximately 100ŌĆō300 heads of sperm were counted in each animal and the total numbers were expressed as sperm/mL [21].
The microscope slides were warmed on a hot plate to 37┬░C, and diluted semen was loaded onto each slide. The microscope slides were examined at a final magnification of ├Ś400 in 10 random microscopic fields, and 100ŌĆō300 sperm per animal were assessed. The motility of the spermatozoa was classified as (1) rapid progressive (spermatozoa moved quickly in direct lines), (2) slow progressive (spermatozoa moved slowly), (3) non-progressive (spermatozoa did not move in a linear direction, but in a circular motion), and (4) immotile (spermatozoa had no movement at all). Briefly, the percentage of motile sperm was calculated as the number of motile sperm├Ś100 /total number of sperm [21].
The testis was removed and weighed. The volume of the testis was measured by immersion [22], then it was fixed in 4% buffered formaldehyde solution for stereological studies. The orientator method was applied to obtain isotropic uniform random sections [22]. Approximately 8ŌĆō12 slabs were collected from each testis using this procedure. Using a trocar, a circle was punched in a random testis slab to estimate tissue shrinkage. After tissue processing and paraffin embedding, 5- and 25-╬╝m sections were cut with a microtome and stained using hematoxylin and eosin and HeidenhainŌĆÖs Azan stains. The areas of the circles were measured before processing (not shrunken) and after processing (shrunken) and finally, the degree of shrinkage ŌĆ£d(shr)ŌĆØ was calculated by the following formula:
d(shr)=1ŌĆō(area [after]/area [before])1.5
Next, the total volume of the testis was calculated based on tissue shrinkage (V[shrunken]) using the following formula:
V (shrunken)=V (not shrunken)├Ś[1ŌłÆd(shr)]
The volume density of the testis section was analyzed by video microscopy. A point grid was superimposed on live microscopic images of each section (5-╬╝m thickness) and displayed on a monitor by software designed at the Histomorphometry and Stereology Research Center. The volume density, abbreviated as ŌĆ£Vv (structure/testis),ŌĆØ of each testicular component, including the seminiferous tubules, interstitial tissue, and germinal epithelium was estimated using the point-counting method [23]. Finally, the total volume of each component was obtained using the following formula:
V (structure)=Vv (structure/testis)├ŚV (shrunken)
A computer linked to a light microscope (Nikon E200, Tokyo, Japan) with a ├Ś40 oil lens (numerical aperture=1.4) was used to assess the total number of testicular cell types, including spermatogonia, spermatocytes, spermatids (round and long), and Sertoli and Leydig cells. The total number of testicular cell types was calculated by applying the optical dissector method to the 25-┬Ąm-thick sections [24]. To do so, the microscopic fields were scanned by moving the microscope stage equal distances in the X and Y directions based on systematic uniform random sampling. Movement in the Z direction was also performed using a microcator (MT12; Heidenhain Corp., Traunreut, Germany) fixed on the microscope stage. The Z-axis distribution of cells sampled in different focal planes was plotted to determine the guard zones and dissectorŌĆÖs height [25]. The numerical density (Nv) was estimated using the following formula:
Nv=╬ŻQ/(╬ŻA├Śhr)├Ś(t/BA)
, where ╬ŻQ was the number of nuclei for each cell type coming into focus. ╬ŻA indicated the total area of the unbiased counting frame, and h represented the dissectorŌĆÖs height, t was the mean section thickness, and BA was the microtome block advance. Finally, the total number of testicular cell types was calculated by multiplying the Nv by V (structure):
N (cells)=Nv├ŚV (structure)
, where V (structure) was the total volume of the germinal epithelium for the germinal layer cells and the total volume of the interstitial tissue for the Leydig cells.
As shown in Table 1, the sperm count and motility percentage were lower in the SD group than in the GR group (p<0.05). Conversely, the percentage of immotile sperm was higher in the SD group than in the GR group (p<0.01). The estimated number of non-progressive sperm was lower in the animals with SD than the GR group (p<0.03). Furthermore, no improvement was observed in sperm count and motility after concomitant treatment of the SD group with olive oil. There were also no significant differences among other experimental groups, including CON, GR, OO, and GR+OO, in this regard.
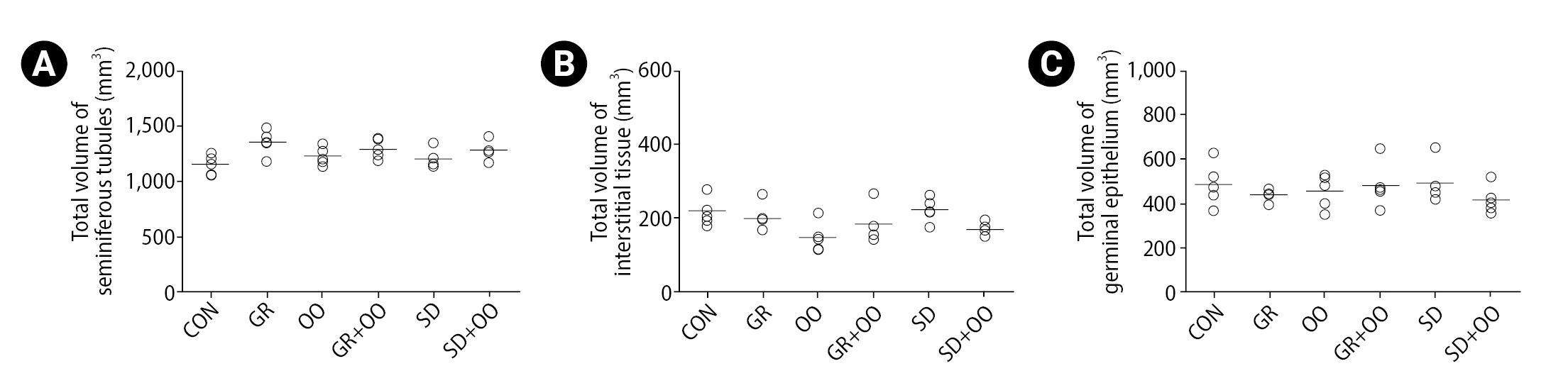
The stereological study results showed no significant change in the total volumes of the seminiferous tubules, interstitial tissue, or germinal epithelium in all study groups (Figure 1). The total numbers of testicular cell types in the SD group, including spermatogonia, spermatocytes, round spermatids, long spermatids, Sertoli cells, and Leydig cells were 54%, 33%, 30%, 36%, 45%, and 37% lower, respectively, than in the GR group. Interestingly, the total number of Sertoli and Leydig cells was significantly higher in the SD rats treated with olive oil than in the untreated SD group. Despite an increase in the total number of other testicular cell types, no significant differences were detected. Similarly, no significant difference was observed among the other experimental groups (Figure 2).
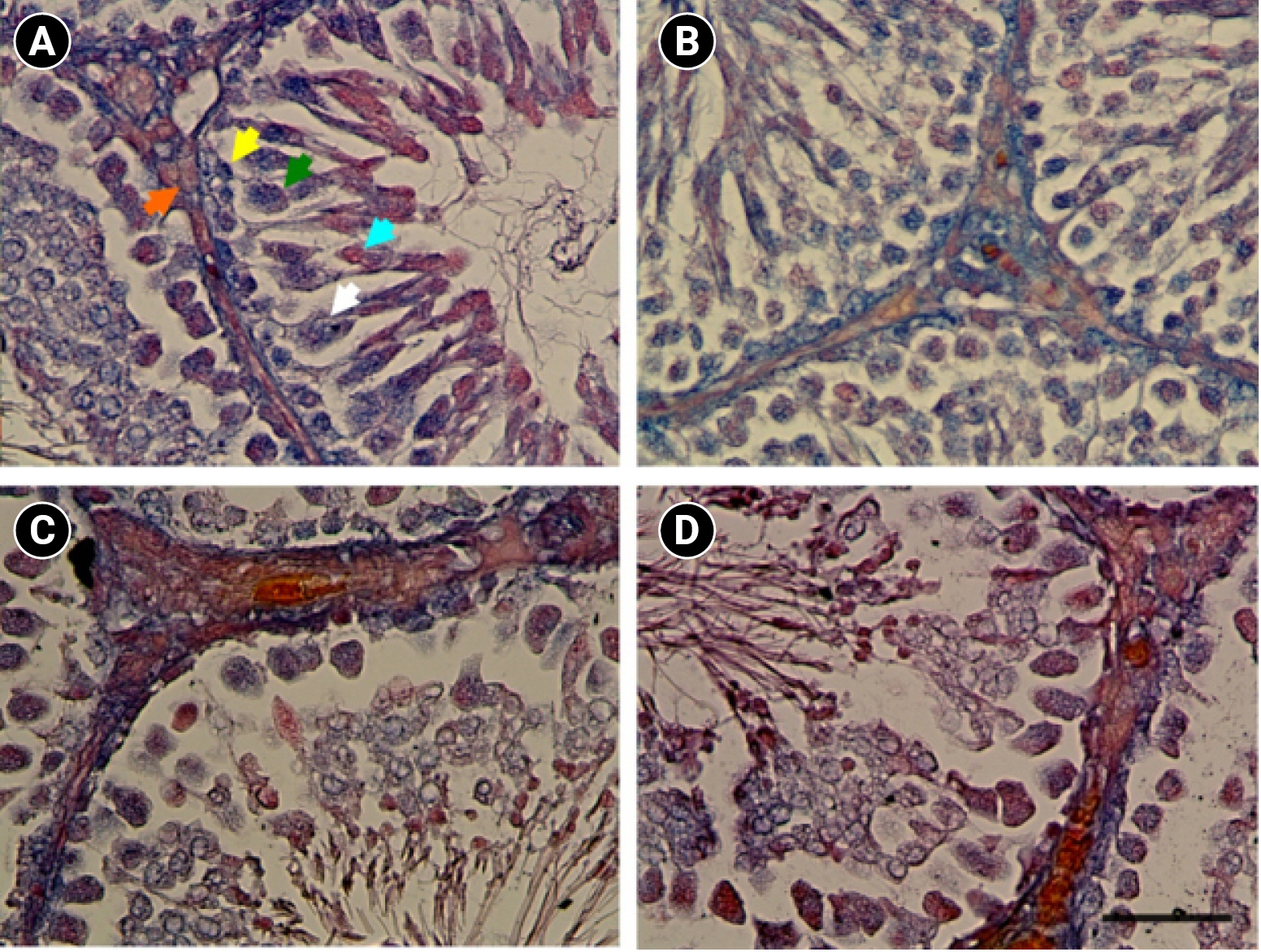
A qualitative evaluation of the testicular tissues is presented in Figure 3. The total number of testicular cell types seen in the histological sections of the SD group was lower than in the GR group. Concomitant treatment of the SD group with olive oil only improved the number of Sertoli and Leydig cells, while not altering other testicular cell types.
Infertility is affected by the quality, timing, and duration of sleep. Human and animal models have shown that SD alters reproductive hormone levels that play a key role in determining fertility. To study sleep effects, the induction of SD in animal models is often used, as was done in this study.
The present study used stereological techniques to investigate the protective effects of olive oil on sperm quality and testicular structural changes in adult male rats with SD. The first stage of the study revealed that SD significantly decreased the total number of testicular cell types, including spermatogonia, spermatocytes, round spermatids, long spermatids, Sertoli cells, and Leydig cells by 54%, 33%, 30%, 36%, 45%, and 37%, respectively. Nevertheless, no significant changes were observed in the total volumes of the seminiferous tubules, interstitial tissue, or germinal epithelium in the study groups. A decrease in the number of the testosterone-producing Leydig cells and the supporting Sertoli cells could affect the proliferation and differentiation of germ cells as well as the spermatogenesis process [26]. Therefore, the decrease in the number of spermatogonia, spermatocytes, and spermatids cells could be a result of the decrease in Leydig and Sertoli cells. These effects of SD on the histological changes in testicular tissues were further confirmed by our sperm quality findings. Accordingly, SD decreased sperm count and motility (p<0.05) and increased the percentage of immotile sperm (p<0.01), probably due to impaired spermatogenesis. These findings were consistent with the results of other studies demonstrating the detrimental effects of SD on the reproductive system of male rats [27,28]. It has been reported that SD decreased sperm motility and induced abnormal testicular histopathology. Atrophy of the seminiferous tubules and spermatid retention were also found with SD [27]. It has been suggested that decreased testosterone levels are the main reason for testicular abnormalities following SD. Elevated corticosteroid levels in stressful conditions, such as SD, could be responsible for decreased testosterone secretion through inhibition of the hypothalamic-pituitary-gonadal axis [27,29]. Consequently, oxidative stress has been presumed to cause the elevated glucocorticoid levels in SD. It has been proposed that sleep recovery could decrease malondialdehyde levels and increase catalase and glutathione levels, which might then reduce the high glucocorticoid levels induced by SD [30,31]. Stress-induced corticosterone levels could induce apoptosis in Leydig cells and decrease the production of testosterone [32]. In response to luteinizing hormone stimulation, Leydig cells produce testosterone and regulate spermatogenesis [26]. Another probable mechanism for decreased testosterone levels is associated with serotonin levels. Binding of serotonin to the 5-HT2 receptor in Leydig cells could inhibit testosterone synthesis [33]. Since animal studies have reported an increase in serotonin concentrations during SD [34,35], it could be hypothesized that the SD-induced lowering of testosterone might be associated with the release of serotonin. Although these mechanisms may be involved in the reduction of testosterone and the consequent disturbance of spermatogenesis, the exact mechanism underlying the anatomical alterations associated with SD is not fully understood and should be clarified in future studies.
The potential of olive oil to reverse testicular dysfunction has been demonstrated by several studies [18,36]. Because of its antioxidant and anti-inflammatory properties, olive oil can alleviate testicular damage and restore spermatogenesis [18]. Nevertheless, no studies have evaluated the protective effects of olive oil on the testicular damage induced by SD in rats.
The second stage of the present study demonstrated that olive oil significantly increased the total number of Sertoli and Leydig cells in the SD group. Despite an increase in the total number of other testicular cell types, no significant differences were observed. Additionally, no improvement was detected in sperm count and motility after concomitant treatment of the SD group with olive oil for 21 days. A longer period may be required for the Sertoli and Leydig cells to affect the proliferation and differentiation of germ cells as well as the spermatogenesis process. Moreover, an increase in the number of spermatogonia, spermatocytes, and spermatid cells could reach significant levels in the SD+OO group by using higher doses of olive oil. Further studies are recommended to assess the effect of higher doses of olive oil for longer time periods.
One of the limitations of our study was that corticosterone and the main hormones involved in the hypothalamic-pituitary-gonadal axis were not measured. We also did not examine the impact of olive oil following SD on oxidative stress in the adult rat testis. The current study demonstrated that SD can induce structural changes in testicular tissue, which could then affect sperm count and motility. Interestingly, olive oil only improved the total number of Sertoli and Leydig cells in the SD group, without improving sperm count and motility.
Acknowledgments
The authors would like to thank Ms. A. Keivanshekouh at the Research Consultation Center of Shiraz University of Medical Sciences for improving the use of English in the manuscript.
Figure┬Ā1.
The dot plots represent the total volume of (A) seminiferous tubules, (B) interstitial tissue, and (C) germinal epithelium in the control (CON), grid floor+distilled water (GR), olive oil (OO), grid floor+olive oil (GR+OO), sleep deprivation+distilled water (SD), and sleep deprivation+olive oil (SD+OO) groups. The horizontal bars show the means of the parameters in the experimental groups. The p-values <0.05 were considered statistically significant.
Figure┬Ā2.
The dot plots show the total number of (A) spermatogonia, (B) spermatocytes, (C) round spermatids, (D) long spermatids, (E) Sertoli cells, and (F) Leydig cells in the control (CON), grid floor+distilled water (GR), olive oil (OO), grid floor+olive oil (GR+OO), sleep deprivation+distilled water (SD), and sleep deprivation+olive oil (SD+OO) groups. The horizontal bars show the means of the parameters in the experimental groups. a)pŌēż0.05; b)pŌēż0.01.

Figure┬Ā3.
Microscopic evaluation of the testicular tissue in the control (CON; A), grid floor+distilled water (GR; B), sleep deprivation+distilled water (SD; C), and sleep deprivation+olive oil (SD+OO; D) groups. The spermatogenic cells, including spermatogonia (yellow arrow), spermatocytes (green arrow), spermatids (blue arrow), Sertoli cells (supporting cells, white arrow), and Leydig cells (interstitial cells, orange arrow), were represented in the CON group. It should be noted that the olive oil (OO) and grid floor+olive oil (GR+OO) groups showed the same characteristics as the CON and GR groups; therefore, these groups are not shown here. A reduction in the population of the spermatogenic, Sertoli, and Leydig cells can be seen in the SD group. Concurrent treatment of the sleep deprived rats with olive oil only improved the Sertoli and Leydig cells, and did not lead to significant changes in other testicular cell types. Scale bar=30 ╬╝m.
Table┬Ā1.
Comparison of sperm count and motility in the study groups (n=5)
| Group | Sperm count (├Ś106) |
Motility by type (%) |
Total motility (%) | Immotile (%) | ||
|---|---|---|---|---|---|---|
| Rapid progressive | Slow progressive | Non-progressive | ||||
| CON | 8.9┬▒4.9 | 5.2┬▒3.5 | 22.0┬▒15.3 | 61.7┬▒10.9 | 88.9┬▒10.1 | 11.1┬▒10.1 |
| GR | 10.4┬▒1.6 | 4.3┬▒4.0 | 27.7┬▒9.0 | 55.1┬▒17.9 | 87.1┬▒3.8 | 12.9┬▒3.8 |
| OO | 8.3┬▒5.0 | 3.0┬▒2.7 | 25.3┬▒17.8 | 58.9┬▒26.4 | 87.2┬▒34.2 | 12.8┬▒4.2 |
| GR+OO | 6.3┬▒3.2 | 4.1┬▒4.0 | 21.3┬▒20.3 | 63.1┬▒22.0 | 88.5┬▒4.1 | 11.5┬▒ 4.1 |
| SD | 5.6┬▒3.1a) | 2.4┬▒2.1 | 15.8┬▒12.5a) | 51.2┬▒17.5 | 69.4┬▒23.1a) | 30.6┬▒13.9b) |
| SD+OO | 7.2┬▒2.2 | 4.6┬▒3.3 | 19.7┬▒10.9 | 47.4┬▒15.3 | 71.7┬▒9.4 | 28.3┬▒7.3 |
References
2. Ibarra-Coronado EG, Pantaleon-Martinez AM, Velazquez-Moctezuma J, Prospero-Garcia O, Mendez-Diaz M, Perez-Tapia M, et al. The bidirectional relationship between sleep and immunity against infections. J Immunol Res 2015;2015:678164.




3. Aldabal L, Bahammam AS. Metabolic, endocrine, and immune consequences of sleep deprivation. Open Respir Med J 2011;5:31-43.



4. Tartar JL, Ward CP, McKenna JT, Thakkar M, Arrigoni E, McCarley RW, et al. Hippocampal synaptic plasticity and spatial learning are impaired in a rat model of sleep fragmentation. Eur J Neurosci 2006;23:2739-48.



5. Kim EY, Mahmoud GS, Grover LM. REM sleep deprivation inhibits LTP in vivo in area CA1 of rat hippocampus. Neurosci Lett 2005;388:163-7.


6. Lee DS, Choi JB, Sohn DW. Impact of sleep deprivation on the hypothalamic-pituitary-gonadal axis and erectile tissue. J Sex Med 2019;16:5-16.


7. Wittert G. The relationship between sleep disorders and testosterone in men. Asian J Androl 2014;16:262-5.



8. Jensen TK, Andersson AM, Skakkebaek NE, Joensen UN, Blomberg Jensen M, Lassen TH, et al. Association of sleep disturbances with reduced semen quality: a cross-sectional study among 953 healthy young Danish men. Am J Epidemiol 2013;177:1027-37.


9. Cho JW, Duffy JF. Sleep, sleep disorders, and sexual dysfunction. World J Mens Health 2019;37:261-75.



10. Kalmbach DA, Arnedt JT, Pillai V, Ciesla JA. The impact of sleep on female sexual response and behavior: a pilot study. J Sex Med 2015;12:1221-32.


11. Villafuerte G, Miguel-Puga A, Rodriguez EM, Machado S, Manjarrez E, Arias-Carrion O. Sleep deprivation and oxidative stress in animal models: a systematic review. Oxid Med Cell Longev 2015;2015:234952.




12. Ghosh D, Das Sarkar S, Maiti R, Jana D, Das UB. Testicular toxicity in sodium fluoride treated rats: association with oxidative stress. Reprod Toxicol 2002;16:385-90.


13. Waterman E, Lockwood B. Active components and clinical applications of olive oil. Altern Med Rev 2007;12:331-42.

15. Franco MN, Galeano-Diaz T, Lopez O, Fernandez-Bolanos JG, Sanchez J, De Miguel C, et al. Phenolic compounds and antioxidant capacity of virgin olive oil. Food Chem 2014;163:289-98.


16. Owen RW, Giacosa A, Hull WE, Haubner R, Spiegelhalder B, Bartsch H. The antioxidant/anticancer potential of phenolic compounds isolated from olive oil. Eur J Cancer 2000;36:1235-47.


17. El-Kholy TA, Al-Abbadi HA, Qahwaji D, Al-Ghamdi AK, Shelat VG, Sobhy HM, et al. Ameliorating effect of olive oil on fertility of male rats fed on genetically modified soya bean. Food Nutr Res 2015;59:27758.


18. AL-Ani KH. Protective influence of olive oil on reproductive parameters in male rat treated with cadmium. J Biosci Biotechnol 2013;2:500-5.
19. Kamali AM, Noorafshan A, Karimi F, Karbalay-Doust S. Methodological aspects of REM sleep-deprivation and stereological protocols in the brain-stem respiratory nuclei. J Adv Med Sci Appl Technol 2016;2:283-6.

20. Noorafshan A, Karimi F, Karbalay-Doust S, Kamali AM. Using curcumin to prevent structural and behavioral changes of medial prefrontal cortex induced by sleep deprivation in rats. EXCLI J 2017;16:510-20.


21. Aminsharifi A, Hekmati P, Noorafshan A, Karbalay-Doost S, Nadimi E, Aryafar A, et al. Scrotal cooling to protect against cisplatin-induced spermatogenesis toxicity: preliminary outcome of an experimental controlled trial. Urology 2016;91:90-8.


22. Mandarim-de-Lacerda CA. Stereological tools in biomedical research. An Acad Bras Cienc 2003;75:469-86.


23. Tschanz S, Schneider JP, Knudsen L. Design-based stereology: planning, volumetry and sampling are crucial steps for a successful study. Ann Anat 2014;196:3-11.


24. Wreford NG. Theory and practice of stereological techniques applied to the estimation of cell number and nuclear volume in the testis. Microsc Res Tech 1995;32:423-36.


25. von Bartheld CS. Distribution of particles in the Z-axis of tissue sections: relevance for counting methods. Neuroquantology 2012;10:66-75.


26. Smith LB, Walker WH. The regulation of spermatogenesis by androgens. Semin Cell Dev Biol 2014;30:2-13.


27. Choi JH, Lee SH, Bae JH, Shim JS, Park HS, Kim YS, et al. Effect of sleep deprivation on the male reproductive system in rats. J Korean Med Sci 2016;31:1624-30.




28. Ajibare AJ, Ayodele OD, Olayaki LA. Mifepristone ameliorates sleep deprivation-induced oxidative stress in the testis of rats. Afr J Biomed Res 2020;23:239-45.
29. Breen KM, Karsch FJ. New insights regarding glucocorticoids, stress and gonadotropin suppression. Front Neuroendocrinol 2006;27:233-45.


30. Olayaki LA, Sulaiman SO, Anoba NB. Vitamin C prevents sleep deprivation-induced elevation in cortisol and lipid peroxidation in the rat plasma. Niger J Physiol Sci 2015;30:5-9.

31. Abd El-Aziz EA, Mostafa DG. Impact of sleep deprivation and sleep recovery on reproductive hormones and testicular oxidative stress in adult male rats. AAMJ 2012;10:1.
32. Gao HB, Tong MH, Hu YQ, Guo QS, Ge R, Hardy MP. Glucocorticoid induces apoptosis in rat leydig cells. Endocrinology 2002;143:130-8.


33. Frungieri MB, Zitta K, Pignataro OP, Gonzalez-Calvar SI, Calandra RS. Interactions between testicular serotoninergic, catecholaminergic, and corticotropin-releasing hormone systems modulating cAMP and testosterone production in the golden hamster. Neuroendocrinology 2002;76:35-46.



34. Meerlo P, Sgoifo A, Suchecki D. Restricted and disrupted sleep: effects on autonomic function, neuroendocrine stress systems and stress responsivity. Sleep Med Rev 2008;12:197-210.


- TOOLS







