 |
 |
- Search
| Clin Exp Reprod Med > Volume 50(1); 2023 > Article |
|
Abstract
Objective
The DNA integrity of spermatozoa that attach to fallopian tube (FT) cells is higher than spermatozoa that do not attach. FT epithelial cells can distinguish normal and abnormal sperm chromatin. This study investigated the effects of sperm with a high-DNA fragmentation index (DFI) from men with unexplained repeated implantation failure (RIF) on the Toll-like receptor (TLR) signaling pathway in human FT cells in vitro.
Methods
Ten men with a RIF history and high-DFI and 10 healthy donors with low-DFI comprised the high-DFI (>30%) and control (<30%) groups, respectively. After fresh semen preparation, sperm were co-cultured with a human FT epithelial cell line (OE-E6/E7) for 24 hours. RNA was extracted from the cell line and the human innate and adaptive immune responses were tested using an RT2 profiler polymerase chain reaction (PCR) array.
Results
The PCR array data showed significantly higher TLR-1, TLR-2, TLR-3, TLR-6, interleukin 1α (IL-1α), IL-1β, IL-6, IL-12, interferon α (IFN-α), IFN-β, tumor necrosis factor α (TNF-α), CXCL8, GM-CSF, G-CSF, CD14, ELK1, IRAK1, IRAK2, IRAK4, IRF1, IRF3, LY96, MAP2K3, MAP2K4, MAP3K7, MAP4K4, MAPK8, MAPK8IP3, MYD88, NFKB1, NFKB2, REL, TIRAP, and TRAF6 expression in the high-DFI group than in the control group. These factors are all involved in the TLR-MyD88 signaling pathway.
Conclusion
The MyD88-dependent pathway through TLR-1, TLR-2, and TLR-6 activation may be one of the main inflammatory pathways activated by high-DFI sperm from men with RIF. Following activation of this pathway, epithelial cells produce inflammatory cytokines, resulting in neutrophil infiltration, activation, phagocytosis, neutrophil extracellular trap formation, and apoptosis.
The fallopian tube (FT), also known as the uterine tube, is an important part of the female reproductive tract. The FT plays a critical function in human reproduction by providing a specific cellular and molecular microenvironment to support gamete transport, fertilization, and early embryonic development [1]. Therefore, FT obstruction or epithelial dysfunction may result in fertility impairment [2,3]. Interestingly, human FT epithelial cells are recognized as part of the innate immune system, which could mediate inflammatory responses to potential invaders [4]. Studies have investigated Toll-like receptors (TLRs) as key players in inflammation and found that they are highly expressed by FT epithelial cells [5-7]. TLRs play an essential role in immune activation through the recognition of pathogen-associated molecular patterns (PAMPs) and endogenous damage-associated molecular patterns (DAMPs) [5,6,8]. The interaction of sperm with the FT may affect sperm storage, motility, survival, and capacitation [9]. Sperm interaction with FT epithelial cells is a complex process that could induce biochemical and physiological alterations. Moreover, this process leads to the activation of innate immune mechanisms, such as TLR signaling [5,10]. In general, TLRs play a major role in the immune interaction between FT and sperm [11], sperm capacitation, fertilization, and pregnancy [6,11].
Despite advances in technology in the field of infertility treatment, the implantation success rate in embryo transfer cycles ranges from 26% to 45% [12]. Repeated implantation failure (RIF) is defined as implantation failure after the transfer of at least four high-quality embryos in at least three cycles. Since sperm provides half of the genetic material of the embryo, studies have investigated its role in RIF patients [13]. Sperm DNA integrity is an influential factor in embryo development and reproductive outcomes [14,15]. Although the sperm DNA fragmentation index (DFI) may be associated with an increased risk of miscarriage, its relationship with RIF has not been proven, and this issue needs further investigation [16]. Evidence has demonstrated that the sperm DFI is significantly elevated in infertile couples with unexplained or idiopathic infertility [17-19]. In this context, it has been shown that the DNA integrity of spermatozoa that attach to FT cells is higher than spermatozoa that do not attach [20]. Conspicuously, FT epithelial cells are actively able to distinguish normal and abnormal sperm chromatin [21]. Hence, this study aimed to evaluate the effects of sperm with high sperm DNA fragmentation on the expression of TLRs, adaptor molecules, and cytokines in FT epithelial cells through the polymerase chain reaction (PCR) array method.
This study was approved by the Institutional Review Board of the Ethics Committee of Royan Institute (Reference number: IR.ACECR.ROYAN.REC.EC/91/1084). Written informed consent was obtained from all patients.
Ten men with a history of unexplained RIF, a normal standard semen analysis (according to the 2010 World Health Organization [WHO] criteria) and a DFI >30% comprised the high-DFI group, and 10 normozoospermic men with at least one child and DFI <30% comprised the control group. Patients with a medical history of varicocele, testicular atrophy, undescended testis, genitourinary infection, chronic prostatitis, cigarette smoking, and alcohol consumption were excluded from the study.
The OE-E6/E7 cell line was cultured in Dulbecco’s Modified Eagle’s Medium/F12 (Invitrogen, Paisley, UK) with 1% penicillin and streptomycin (Sigma-Aldrich, Poole, UK), L-glutamine (Invitrogen, Waltham, MA, USA) and 10% fetal bovine serum (Invitrogen, Waltham, MA, USA). The cells were incubated in a 37°C incubator in an atmosphere of 5% CO2 in the air. Semen samples were analyzed according to the WHO guidelines and sperm preparation was performed by density gradient centrifugation and the swim-up method.
The sperm chromatin structure assay was used to determine the DFI value. An aliquot of unprocessed semen containing 1-4 million sperm/mL was diluted with Tris-HCl, NaCl, and ethylenediaminetetraacetic acid (EDTA) (TNE) buffer. The cell suspension was treated with an acidic detergent solution (pH 1.2) comprising 0.1% Triton X-100, 0.15 mol/L NaCl, and 0.08 mol/L HCl for 30 seconds. The cells were then stained with 6 mg/L purified acridine orange (AO) (Polysciences, Warrington, PA, USA) in a phosphate citrate buffer (pH 6.0). Then, chromatin damage was determined by measuring the metachromatic change of AO fluorescence from green (associated with double-stranded DNA) to red (associated with single-stranded and denatured DNA). All analysis were performed by a technical expert [22] and a DFI >30% was considered high [23,24].
First, OE-E6/E7 cell culture was performed in 6-well culture plates until confluency. After sperm preparation in both groups (high-DFI and control), 1×103 sperm in each group were added to cultured cells separately [11]. The OE-E6/E7 cells were incubated with sperm for 24 hours. Then, the culture medium was collected. The OE-E6/E7 cells were washed with calcium and magnesium-free phosphate-buffered saline (PBS) and then were harvested with trypsin/EDTA (Invitrogen, Paisley, UK). The harvested cells were centrifuged at 300 ×g for 5 minutes and the pellets were used for RNA extraction.
RNA extraction was performed using an RNeasy mini kit (Cat. No: 73,304, Qiagen, Hilden, Germany), according to the manufacturer’s instructions. The quality and quantity of the extracted RNA were evaluated using a Nanodrop spectrophotometer device. cDNA synthesis was carried out using an RT2 first strand kit (Cat. No: 330,40, Qiagen) [25].
The expression levels of 84 genes involved in the human innate immune response were evaluated by an RT2 profiler PCR array kit (Cat. No: PAHS-052A, Qiagen) and an RT2 SYBR Green ROX qPCR master mix (Cat. No: 330,502, Qiagen). Glyceraldehyde-3-phosphate dehydrogenase was considered as an endogenous control. The experiment was performed with a StepOnePlus Real-Time PCR System (Thermo Fisher, Waltham, MA, USA).
Data were expressed as mean±standard deviation. The normality of the data distribution was examined by the Kolmogorov-Smirnov test. The Mann-Whitney U test and the independent samples t-test were used for non-parametric and parametric analysis, respectively. All data were analyzed using SPSS ver. 22 software (IBM Co., Armonk, NY, USA), and a p<0.05 was considered as the significance level.
After 24 hours of co-culture, up to 90% of sperm were viable and unattached to the OE-E6/EE7 cells. Therefore, sperm were easily isolated from the cell line using PBS washes. A profiler PCR array was used to evaluate alterations in the expression of genes related to the innate and adaptive immune responses in the OE-E6/E7 cell line incubated with sperm from the two study groups (high-DFI and control groups). The genes with significantly upregulated expression in the high-DFI group in comparison with the low-DFI (control) group are shown in Figures 1-3 (p<0.05).
The expression levels of TLR-1, TLR-2, TLR-3, and TLR-6 in the high-DFI group were significantly higher than the control group (p<0.05) (Figure 1). The relative expression levels of proinflammatory cytokine and chemokine receptors, including interleukin 1α (IL-1α), IL-1β, IL-6, IL-12, interferon α (IFN-α), IFN-β, tumor necrosis factor α (TNF-α), C-X-C motif chemokine ligand 8 (CXCL8), granulocyte colony-stimulating factor (G-CSF), and granulocyte-macrophage (GM)-CSF were higher in the high-DFI group than in the control group (p<0.05) (Figure 2). Furthermore, the expression levels of several adaptor molecules, including cluster of differentiation 14 (CD14), ETS transcription factor ELK1 (ELK1), IL-1 receptor-associated kinase (IRAK1), IRAK2, IRAK4, interferon regulatory factor 1 (IRF1), IRF3, lymphocyte antigen 96 (LY96), mitogen-activated protein kinase kinase 3 (MAP2K3), MAP2K4, MAP3K7, MAP4K4, mitogen-activated protein kinase 8 (MAPK8), MAPK8IP3, myeloid differentiation primary response 88 (MYD88), nuclear factor kappa B subunit 1 (NFKB1), NFKB2, REL, Toll/interleukin-1 receptor domain-containing adapter protein (TIRAP), and TNF receptor-associated factor 6 (TRAF6), were significantly greater in the high-DFI group than in the control group (p<0.05) (Figure 3).
The interaction of sperm and the FT is considered an essential event in the fertilization process, and it can activate an immune response. In this study, the expression of TLRs, adaptor molecules, and inflammatory cytokines and chemokines was evaluated in response to the interaction of sperm with high-DFI from RIF patients with a human FT epithelial cell line. The results showed that the interaction of high-DFI sperm from these patients with FT cells stimulated the innate immune system and specific inflammatory pathways.
The innate immune system, as the first line of defense, plays an influential role by recognizing PAMPs and DAMPs through TLRs in FT epithelial cells [26]. Several immune pathways are potentially involved in the interaction of high-DFI sperm with the FT. This study revealed that the expression of TLR-1, TLR-2, and TLR-6 were significantly higher in the high-DFI group than in the low-DFI group (Figure 1). Existing evidence indicates that TLR-2 can form a heterodimer with TLR-1 or TLR-6. These complexes can recognize DAMPs, which are released from damaged cells. Following TLR signaling, two main pathways—the MyD88 and TIR-domain-containing adapter-inducing interferon-β (TRIF)—are activated. In particular, TLR-1, TLR-2, and TLR-6 signaling is mediated by the MyD88-dependent pathway [11]. The intracellular part of these complexes binds to cytosolic adaptor molecules, including MyD88 and TIRAP. Finally, signal transduction occurs through IRAK1. Subsequently, the TLR signaling pathway leads to the activation of activating protein-1 (Ap-1) and nuclear factor kappa light chain enhancer of activated B cell (NF-κB) transcription factors, which regulate the production of inflammatory cytokines and chemokines, including IL-1, IL-6 and, TNF-α, G-CSF, GM-CSF, CXCL10, CXCL8 (IL-8), and C-C motif chemokine ligand 2 (CCL2) (Figure 4) [27,28].
Our study results demonstrated that all transcription factors involved in the MyD88-dependent pathway were significantly increased in the high-DFI group, which may indicate the effect of high-DFI in stimulating this inflammatory pathway (Figures 2 and 3). Although these factors play an important role in fertility, their upregulated expression makes the FT conditions unfavorable for successful fertilization and healthy pregnancy. For instance, IL-6 plays a vital role in sperm capacitation and embryo implantation; however, the upregulation of IL-6 can reduce the ciliary activity of the FT epithelium and lead to defective embryo implantation in the FT [29]. IL-1 also causes destruction of the cilia in pathological conditions [30]. According to the evidence, a high concentration of IL-1 leads to the impairment of fertilization in a sperm penetration assay and zona pellucida assay and inhibits mouse embryo development [31]. Furthermore, the upregulation of TNF-α could reduce sperm motility and prevent sperm/mucus interaction [32].
Many studies have been conducted on the relationship between male infertility and RIF, but a connection has not been securely established. It is possible that high-DFI in sperm may lead to defects in embryo implantation through a negative effect on the FT. In a previous study, we cultured semen samples of 10 healthy men with the OE-E6/E7 cell line and examined the expression of 84 cytokines in the cell line using quantitative PCR. That study showed that sperm exhibited changing expression levels of cytokines, chemokines, and growth factors [33]. Furthermore, in another study, we cultured the sperm samples of 10 men with unexplained infertility and high-DFI compared to semen samples of 10 healthy donor men with OE-E6/E7 cell lines, and quantitative PCR analysis showed that DNA damage-induced activation of the TLR signaling pathway in human FT led to the upregulation of inflammatory cytokines and chemokines [11].
Oxidative stress, apoptosis, and chromatin remodeling are three major causes of sperm DNA fragmentation. In patients with high sperm DNA fragmentation, DAMPs are released from damaged spermatozoa, which can activate the immune response [34]. The FT epithelial cells may respond to these molecules through TLRs, leading to inflammatory cytokine secretion, which may orchestrate the infiltration of granulocytes, especially neutrophils [27,28].
As Figure 2 shows, IL-1 and TNF-α expression was increased in the high-DFI group; these molecules promote the expression of CXCL8 by endothelial cells [35]. However, CXCL8, also known as IL-8, regulates immune cell migration, especially the infiltration of neutrophils [27,36]. Neutrophils, the major granulocytes in the FT, respond to foreign molecular patterns through signaling pathways, antimicrobial agents, cytotoxic components, and proinflammatory cytokine production [37]. Interestingly, neutrophils can be found in the lumen of the FT through transepithelial migration. In general, neutrophils are capable of producing high amounts of reactive oxygen species, which exert a negative effect on sperm motility and fertility [38]. The activation of neutrophils can also lead to NETosis and apoptosis (Figure 5). NETosis is considered as a cell death model that is different from necrosis and apoptosis. Neutrophil extracellular traps (NETs) are structures consisting of decondensed chromatin and expanded nuclear material that combine with cytoplasmic components after the nuclear membrane decomposes. They are then released following the rupture of the plasma membrane, leading to the trapping and killing of microorganisms [39-41]. It has been shown that the incubation of peripheral neutrophils with equine sperm resulted in severe NET formation. It also has been observed that the repeated deposition of sperm in the presence of neutrophils could reduce fertility potential [42]. These plausible immuno-inflammatory mechanisms may be viewed as a potential factor in male infertility. Since the pathophysiological identification of patients with RIF can guide the treatment of patients and increase the success rate, more research on the effects of sperm with high-DNA fragmentation on the female reproductive system and a better understanding of the immune-inflammatory mechanism and neutrophil function in these patients are needed.
In conclusion, this study provides novel insights into the role of the innate immune responses and TLR signaling pathways in men with history of unexplained RIF. The MyD88-dependent pathways of TLR-1, TLR-2, and TLR-6, as well as neutrophil activation, are inflammatory pathways activated by high-DFI sperm that could be considered as a potential immunopathogenesis of male infertility.
Acknowledgments
The authors would like to thank the Royan Institute for Reproductive Biomedicine, Tehran, Iran for their cooperation throughout the period of the study.
Figure 1.
The relative mRNA levels of Toll-like receptors (TLRs) 1-6 in the high-DNA fragmentation index (DFI) and control groups. a)p≤0.05; b)p≤0.001.
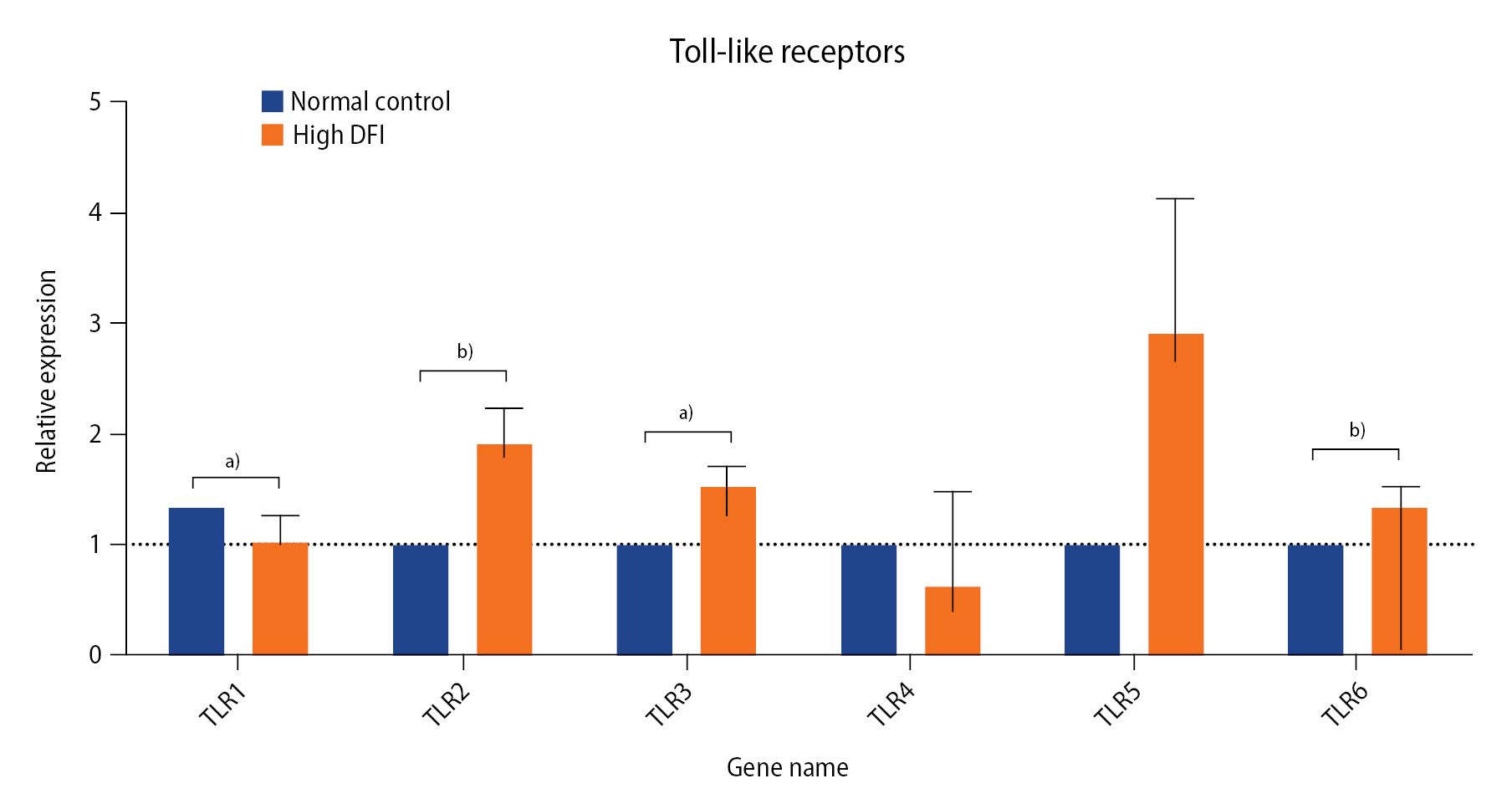
Figure 2.
The relative mRNA levels of cytokines and chemokines in the high-DNA fragmentation index (DFI) and control groups. G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; CCL2, C-C motif chemokine ligand 2; CXCL8, C-X-C motif chemokine ligand 8; IL, interleukin; IFN, interferon; TNF, tumor necrosis factor. a)p≤0.001.
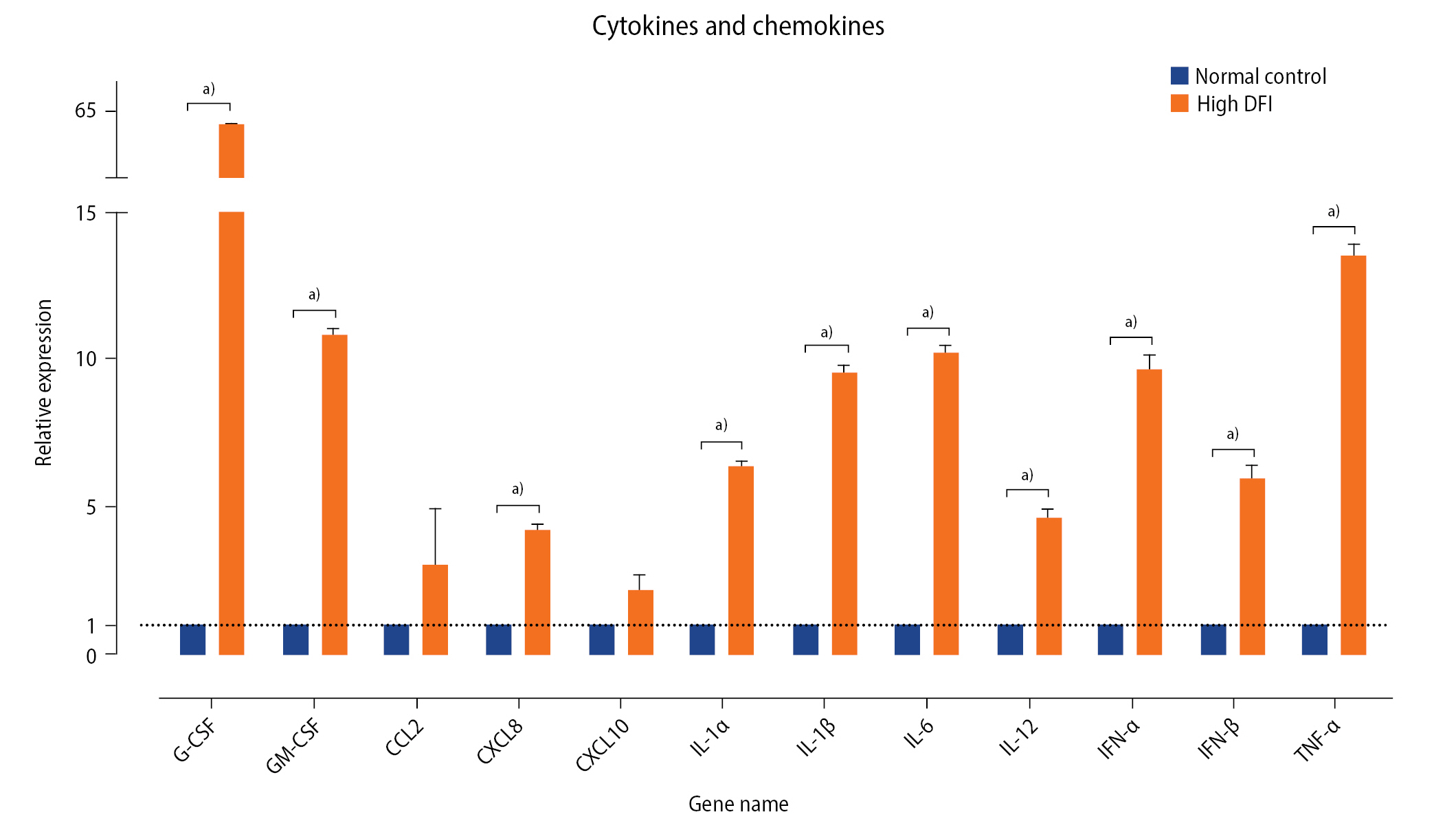
Figure 3.
The relative mRNA levels of adaptor molecules in the high-DNA fragmentation index (DFI) and control groups. CD14, cluster of differentiation 14; ELK1, ETS transcription factor ELK1; IRAK1, IL-1 receptor-associated kinase; IRF1, interferon regulatory factor 1; LY96, lymphocyte antigen 96; MAP3K7, mitogen-activated protein kinase kinase kinase 7; MAPK, mitogen-activated protein kinase; MYD88, myeloid differentiation primary response 88; NFKB1, nuclear factor kappa B subunit 1; TIRAP, Toll/interleukin-1 receptor domain-containing adapter protein; TRAF6, TNF receptor-associated factor 6. a)p≤0.05; b)p≤0.001.
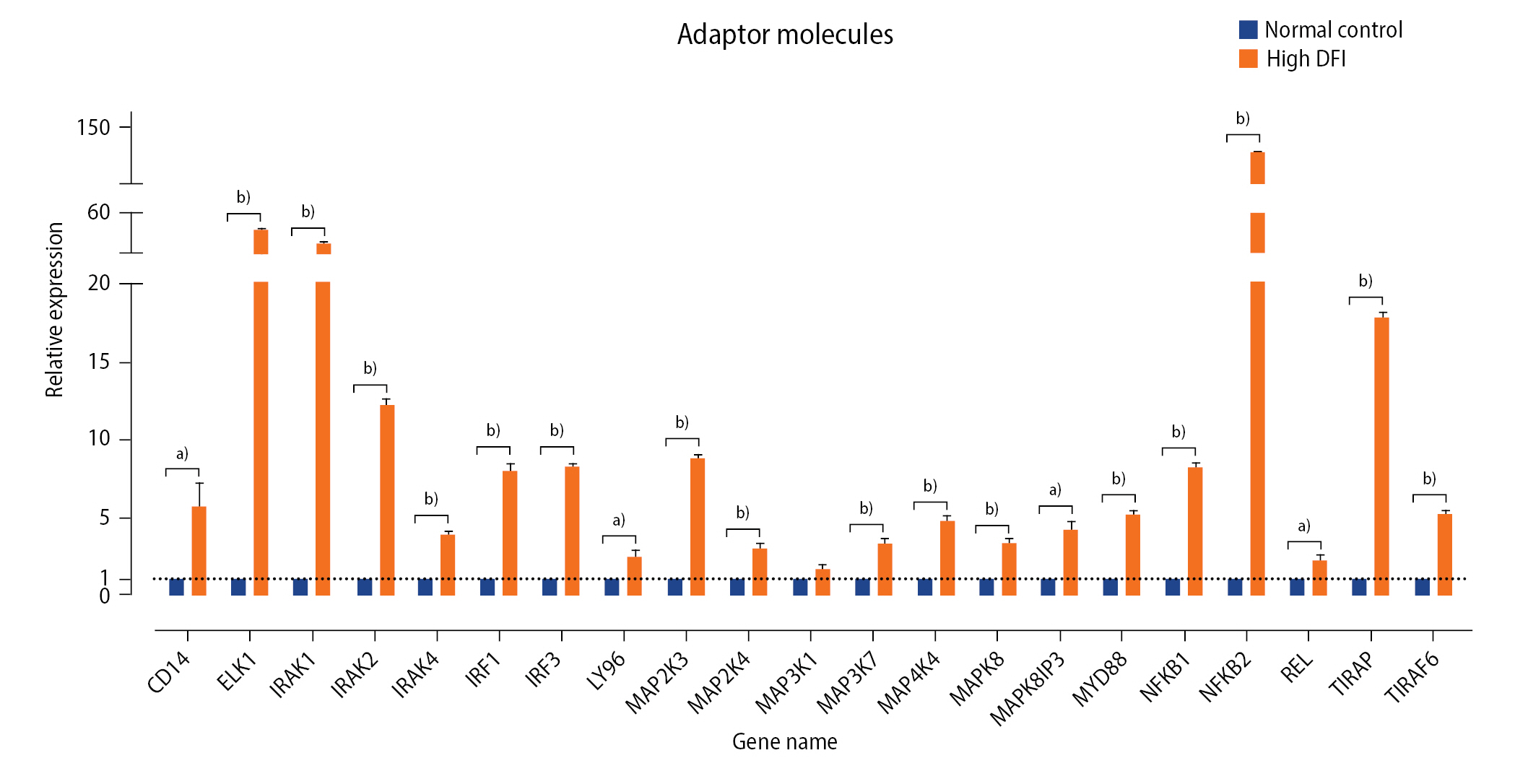
Figure 4.
Toll-like receptor (TLR) signaling pathway. TLR-2 can form a heterodimer with TLR-1 or TLR-6. These complexes can recognize damage-associated molecular patterns, which are released through damaged cells. Activation of the TLR pathway leads to inflammatory cytokines, which can orchestrate neutrophil activation and NETosis. MD2, myeloid differentiation protein 2 (also known as lymphocyte antigen 96); MyD88, myeloid differentiation primary response 88; TIRAP, Toll/interleukin-1 receptor domain-containing adapter protein; IRAK, IL-1 receptor-associated kinase; TRAF6, TNF receptor-associated factor 6; NF-κB, nuclear factor kappa light chain enhancer of activated B cell; MAPK, mitogen-activated protein kinase; AP-1, activating protein-1; G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; CXCL8, C-X-C motif chemokine ligand 8; CCL2, C-C motif chemokine ligand 2; IL, interleukin; TNF-α, tumor necrosis factor α; NET, neutrophil extracellular trap.
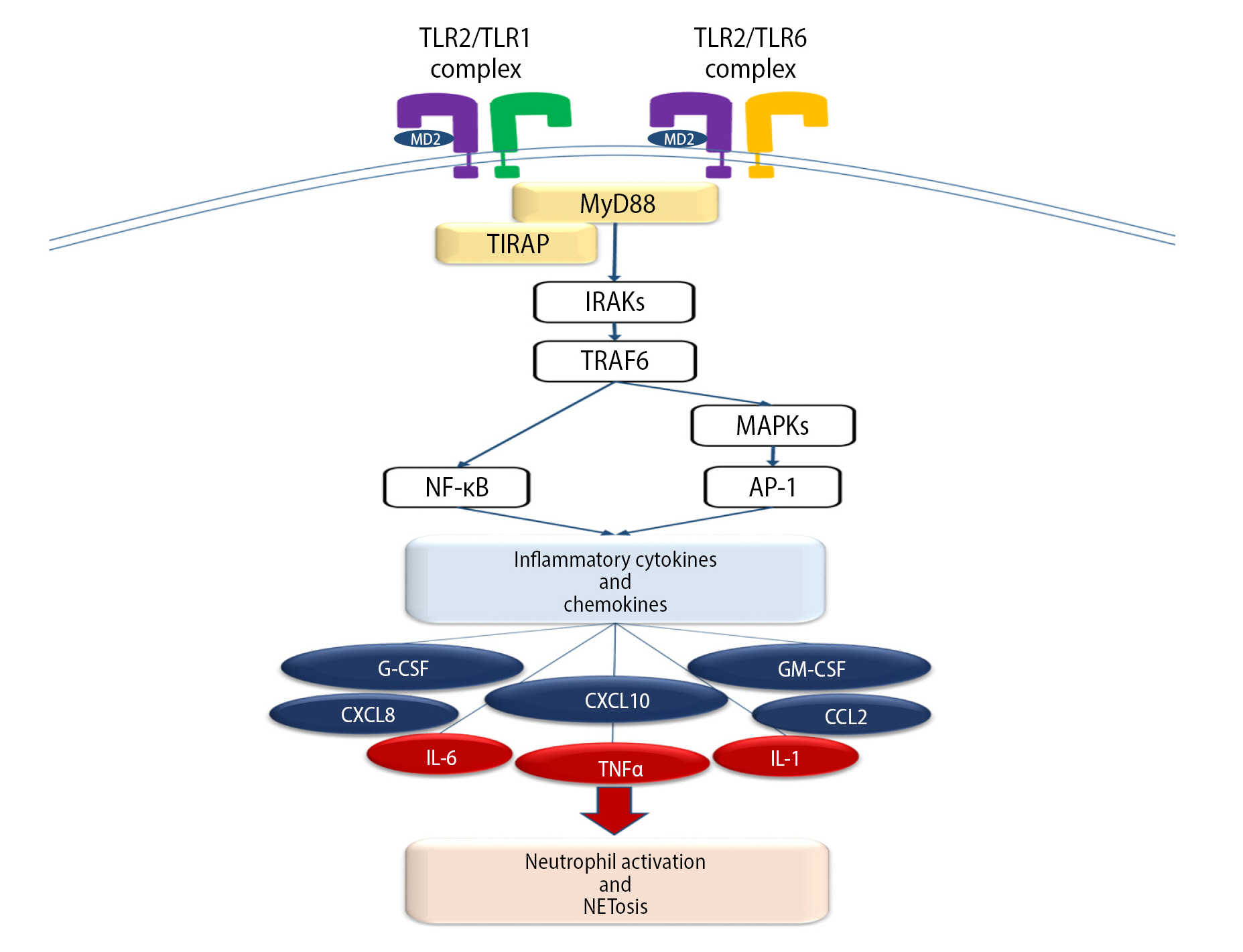
Figure 5.
Immuno-inflammatory mechanism of sperm loss and infertility of men with high sperm DNA fragmentation. The released damage-associated molecular patterns (DAMPs) through damaged sperm are recognized by Toll-like receptors (TLRs) on the epithelial cells. Following TLR signaling, epithelial cells produce inflammatory cytokines, which result in the infiltration of neutrophils. In the lumen of the fallopian tube (FT), neutrophils are activated via TLR-1/TLR-2 and TLR-2/TLR-6 complexes, leading to neutrophil activation, phagocytosis, neutrophil extracellular trap (NET) formation, and apoptosis. DC, dendritic cell; TC, T cell; NEU, neutrophil; MQ, macrophage; MC, mast cell; BC, B cell; EOS, eosinophil; G-CSF, granulocyte colony-stimulating factor; GM-CSF, granulocyte-macrophage colony-stimulating factor; CCL2, C-C motif chemokine ligand 2; CXCL8, C-X-C motif chemokine ligand 8; TNF-α, tumor necrosis factor α; IL, interleukin; TCR, t-cell receptor; CD3, cluster of differentiation 3.

References
1. Li S, Winuthayanon W. Oviduct: roles in fertilization and early embryo development. J Endocrinol 2017;232:R1-26.


2. Barriere P, Thibault E, Jean M. Role of Fallopian tube in fertilization. Rev Prat 2002;52:1757-61.

3. Lyons RA, Saridogan E, Djahanbakhch O. The reproductive significance of human Fallopian tube cilia. Hum Reprod Update 2006;12:363-72.


4. Ochiel DO, Fahey JV, Ghosh M, Haddad SN, Wira CR. Innate immunity in the female reproductive tract: role of sex hormones in regulating uterine epithelial cell protection against pathogens. Curr Womens Health Rev 2008;4:102-17.



5. Nasu K, Narahara H. Pattern recognition via the toll-like receptor system in the human female genital tract. Mediators Inflamm 2010;2010:976024.




6. Kannaki TR, Shanmugam M, Verma PC. Toll-like receptors and their role in animal reproduction. Anim Reprod Sci 2011;125:1-12.


7. Amjadi F, Zandieh Z, Salehi E, Jafari R, Ghasemi N, Aflatoonian A, et al. Variable localization of Toll-like receptors in human fallopian tube epithelial cells. Clin Exp Reprod Med 2018;45:1-9.




8. Horne AW, Stock SJ, King AE. Innate immunity and disorders of the female reproductive tract. Reproduction 2008;135:739-49.


9. Holt WV, Fazeli A. Sperm selection in the female mammalian reproductive tract. Focus on the oviduct: hypotheses, mechanisms, and new opportunities. Theriogenology 2016;85:105-12.


10. Zandieh Z, Ashrafi M, Jameie B, Amanpour S, Mosaffa N, Salman Yazdi R, et al. Evaluation of immunological interaction between spermatozoa and fallopian tube epithelial cells. Andrologia 2015;47:1120-30.


11. Zandieh Z, Ashrafi M, Aflatoonian K, Aflatoonian R. Human sperm DNA damage has an effect on immunological interaction between spermatozoa and fallopian tube. Andrology 2019;7:228-34.



12. Timeva T, Shterev A, Kyurkchiev S. Recurrent implantation failure: the role of the endometrium. J Reprod Infertil 2014;15:173-83.


13. Coughlan C, Clarke H, Cutting R, Saxton J, Waite S, Ledger W, et al. Sperm DNA fragmentation, recurrent implantation failure and recurrent miscarriage. Asian J Androl 2015;17:681-5.



14. Rybar R, Markova P, Veznik Z, Faldikova L, Kunetkova M, Zajicova A, et al. Sperm chromatin integrity in young men with no experiences of infertility and men from idiopathic infertility couples. Andrologia 2009;41:141-9.


15. Govahi A, Amjadi F, Nasr-Esfahani MH, Raoufi E, Mehdizadeh M. Accompaniment of time-lapse parameters and cumulus cell RNA-sequencing in embryo evaluation. Reprod Sci 2022;29:395-409.



16. Coughlan C, Ledger W, Wang Q, Liu F, Demirol A, Gurgan T, et al. Recurrent implantation failure: definition and management. Reprod Biomed Online 2014;28:14-38.


17. Oleszczuk K, Augustinsson L, Bayat N, Giwercman A, Bungum M. Prevalence of high DNA fragmentation index in male partners of unexplained infertile couples. Andrology 2013;1:357-60.


18. Zandieh Z, Vatannejad A, Doosti M, Zabihzadeh S, Haddadi M, Bajelan L, et al. Comparing reactive oxygen species and DNA fragmentation in semen samples of unexplained infertile and healthy fertile men. Ir J Med Sci 2018;187:657-62.



19. Asgari F, Gavahi A, Karimi M, Vatannejad A, Amjadi F, Aflatoonian R, et al. Risk of embryo aneuploidy is affected by the increase in sperm DNA damage in recurrent implantation failure patients under ICSI-CGH array cycles. Hum Fertil (Camb) 2022;25:872-80.


20. Zini A, Kamal K, Phang D, Willis J, Jarvi K. Biologic variability of sperm DNA denaturation in infertile men. Urology 2001;58:258-61.


21. Bianchi PG, De Agostini A, Fournier J, Guidetti C, Tarozzi N, Bizzaro D, et al. Human cervical mucus can act in vitro as a selective barrier against spermatozoa carrying fragmented DNA and chromatin structural abnormalities. J Assist Reprod Genet 2004;21:97-102.



22. Erenpreiss J, Elzanaty S, Giwercman A. Sperm DNA damage in men from infertile couples. Asian J Androl 2008;10:786-90.


23. Oleszczuk K, Giwercman A, Bungum M. Intra-individual variation of the sperm chromatin structure assay DNA fragmentation index in men from infertile couples. Hum Reprod 2011;26:3244-8.


24. Gardner DK, Weissman A, Howles CM, Shoham Z. Textbook of assisted reproductive technologies: laboratory and clinical perspectives. CRC press; 2008.
25. Ajdary M, Keyhanfar F, Aflatoonian R, Amani A, Amjadi F, Zandieh Z, et al. Design and evaluation of a novel nanodrug delivery system for reducing the side effects of clomiphene citrate on endometrium. Daru 2020;28:423-32.




26. Marshall JS, Warrington R, Watson W, Kim HL. An introduction to immunology and immunopathology. Allergy Asthma Clin Immunol 2018;14(Suppl 2):49.


27. Fukui A, Ohta K, Nishi H, Shigeishi H, Tobiume K, Takechi M, et al. Interleukin-8 and CXCL10 expression in oral keratinocytes and fibroblasts via Toll-like receptors. Microbiol Immunol 2013;57:198-206.


28. Cheng K, Gao M, Godfroy JI, Brown PN, Kastelowitz N, Yin H. Specific activation of the TLR1-TLR2 heterodimer by small-molecule agonists. Sci Adv 2015;1:e1400139.



29. Papathanasiou A, Djahanbakhch O, Saridogan E, Lyons RA. The effect of interleukin-6 on ciliary beat frequency in the human fallopian tube. Fertil Steril 2008;90:391-4.


30. Hvid M, Baczynska A, Deleuran B, Fedder J, Knudsen HJ, Christiansen G, et al. Interleukin-1 is the initiator of Fallopian tube destruction during Chlamydia trachomatis infection. Cell Microbiol 2007;9:2795-803.


31. Sueldo CE, Kelly E, Montoro L, Subias E, Baccaro M, Swanson JA, et al. Effect of interleukin-1 on gamete interaction and mouse embryo development. J Reprod Med 1990;35:868-72.

32. Vassiliadis S, Relakis K, Papageorgiou A, Athanassakis I. Endometriosis and infertility: a multi-cytokine imbalance versus ovulation, fertilization and early embryo development. Clin Dev Immunol 2005;12:125-9.




33. Mousavi SO, Mohammadi R, Amjadi F, Zandieh Z, Aghajanpour S, Aflatoonian K, et al. Immunological response of fallopian tube epithelial cells to spermatozoa through modulating cytokines and chemokines. J Reprod Immunol 2021;146:103327.


34. Tang D, Kang R, Coyne CB, Zeh HJ, Lotze MT. PAMPs and DAMPs: signal 0s that spur autophagy and immunity. Immunol Rev 2012;249:158-75.



35. Young RS, Wiles BM, McGee DW. IL-22 enhances TNF-α- and IL-1-induced CXCL8 responses by intestinal epithelial cell lines. Inflammation 2017;40:1726-34.



36. Adolfsson E, Andershed AN. Morphology vs morphokinetics: a retrospective comparison of inter-observer and intra-observer agreement between embryologists on blastocysts with known implantation outcome. JBRA Assist Reprod 2018;22:228-37.



37. Lee SK, Kim CJ, Kim DJ, Kang JH. Immune cells in the female reproductive tract. Immune Netw 2015;15:16-26.




38. Plante M, de Lamirande E, Gagnon C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil Steril 1994;62:387-93.


39. Gupta S, Kaplan MJ. The role of neutrophils and NETosis in autoimmune and renal diseases. Nat Rev Nephrol 2016;12:402-13.




40. Desai J, Mulay SR, Nakazawa D, Anders HJ. Matters of life and death. How neutrophils die or survive along NET release and is "NETosis" = necroptosis? Cell Mol Life Sci 2016;73:2211-9.




- TOOLS







