|
 |
- Search
| Clin Exp Reprod Med > Volume 49(2); 2022 > Article |
|
Abstract
Objective
The aim of this study was to compare the complication rates of oocyte pick-up (OPU) procedures via transvaginal ultrasonography in patients with different levels of ovarian reserve.
Methods
In total, 789 patients who underwent OPU procedures for in vitro fertilization (IVF) were included in the study.
Results
Individuals with normal ovarian reserve had a 2.947-fold higher risk of complications in OPU procedures than individuals with low ovarian reserve, and individuals with high ovarian reserve had a 7.448-fold higher risk of complications than individuals with low ovarian reserve. In addition, a higher number of IVF trials was associated with an increased risk of complications.
Conclusion
The results of this study show that OPU has a higher risk of complications, particularly severe pain, in patients with high ovarian reserve. It is thought that complications can be reduced by preferring mild stimulation in patients with high ovarian reserve. Collecting fewer oocytes is also associated with a lower risk of complications from OPU. Even if a patientŌĆÖs reserve is very good, fewer and higher-quality oocytes should be targeted with the use of the lowest possible dose of drugs.
According to data from the World Health Organization, 48.5 million couples are affected by infertility worldwide [1]. The aim of modern assisted reproductive technologies is to shorten the time until pregnancy, reduce the costs related to infertility, and ultimately, promote the live birth of a healthy baby, which can be achieved with appropriate treatment and measures that strengthen patient safety [2]. In vitro fertilization (IVF), in which fertilization takes place after mature eggs are collected from the ovaries, is a widely used method in the treatment of infertility [3].
Oocyte pick-up (OPU) via transvaginal ultrasonography is a commonly used oocyte collection method that is less invasive than other methods [4]. As this process is easy and effective for collecting eggs, it is frequently preferred as the gold standard approach [5]. Despite these advantages, sometimes damage to the surrounding tissues or complications due to various problems are encountered during or after the procedure. These complications can occur in the early or late period after OPU, and the majority are non-life-threatening conditions, such as bleeding (vaginal or intraabdominal), infection, postprocedural adhesion, and pain [6-8]. More serious complications, such as abscesses, sepsis, fistulas, urethral or/ bladder injuries, and ovarian torsion are rarely encountered [9]. In addition, the risk of complications of OPU is affected by variables such as the experience of the surgeon [4] and the features of the equipment being used [10,11]. In addition to complications arising from the procedure itself, undesirable effects due to anesthesia and sedation can also occur.
It has been reported that 9%ŌĆō24% of patients who undergo ovarian stimulation for IVF have poor ovarian response [12,13]. Possible etiological factors for poor ovarian response include a decrease in the number of follicles due to age, endometriosis, chromosomal and genetic changes, a history of ovarian surgery, pelvic adhesions, and metabolic, autoimmune, and infectious diseases [14-18]. High rates of cycle cancellation and implantation failure have been reported in women with poor ovarian response, despite the implementation of strategies designed to optimize stimulation [19]. Although previous studies have examined the complications that occur after OPU and various factors affecting complications, the number of studies focusing on the relationship between OPU complications and ovarian response level is quite limited. In this context, the current study investigated the relationship between ovarian response and OPU complications in patients who underwent IVF.
A total of 789 patients who underwent oocyte retrieval for IVF were included in the study. The exclusion criteria were a lack of complete clinical records, a history of ovarian surgery, a genetic disease or chromosomal abnormality, a body mass index (BMI) Ōēź30 kg/m2, age over 38 years, and the presence of uterine malformations.
This retrospective cohort study was conducted on patients who underwent OPU procedures between January 2018 and December 2019 at the In Vitro Fertilization Center of BAU Goztepe Medical Park Hospital, Istanbul, Turkey. The Ethics Committee of Beykoz University, Turkey approved the conduct of this study (July 17, 2020; No. 2020/4). During the study period, all procedures and steps of the study were carried out in accordance with the Helsinki Declaration and Good Clinical Practice guidelines. Informed consent was obtained from all individual participants included in the study.
A gonadotropin-releasing hormone (GnRH) antagonist protocol was utilized for ovarian stimulation in all patients. The gonadotropin dose was individualized according to each patientŌĆÖs ovarian reserve and anti-M├╝llerian hormone (AMH) level. Gonadotropin stimulation was started by applying 150ŌĆō450 IU of recombinant follicle-stimulating hormone (Gonal-F, Merck, Istanbul, Turkey), and/or human menopausal gonadotropin (Merional; IBSA Pharmaceutical, Lugano, Switzerland) as the initial dose on the second or third day of the menstrual cycle in all women. Serial vaginal ultrasonography was used to monitor ovarian response. In order to prevent premature luteinization, 0.25 ╬╝g of a GnRH antagonist (Cetrotide 250 ╬╝g, Merck) was added daily when the leading follicle reached a diameter of 14 mm. When the mean diameter of two or three leading follicles reached 17 mm or more, recombinant human chorionic gonadotropin (Ovitrelle 250 ╬╝g, Merck) was used to trigger ovulation. The OPU procedure was carried out after trigger success, between 34 and 36 hours after administration. The equipment used constituted a double-lumen needle (17 gauge) and a pump set at 125 mmHg vacuum (Cook Medical, Bloomington, IN, USA). All follicles sized 14 to 20 mm were obtained without flushing. A glass pipette was used to separate the cumulus-oocyte complexes from the sample, and they were transferred to the laboratory under necessary precautions after washing with G-IVF media (G-IVF Plus, Vitrolife, Kungsbacka,Sweeden). A single dose of cefazolin (1 g) was administered to OPU patients during the procedure. The conscious sedation and analgesia technique was used for pain control during OPU. For sedation: 2 ╬╝g/kg of fentanyl and 0.05 mg/kg of midazolam were administered. Sedation was generally preferred in cases with low follicle development, in cases where a short period of time was predicted. For this purpose, pethidine (0.5ŌĆō1 mg/kg), midazolam (0.01ŌĆō0.03 mg/kg), fentanyl (1ŌĆō2 ╬╝g/kg), lignocaine (10 mL of 1%) and diclofenac (50 mg) were administered. All OPU procedures were performed by a single physician with 15 years of experience (MDG).
The following parameters were recorded: age, BMI, smoking, causes of infertility, AMH levels, day-3 FSH, peak estradiol (E2) level in the cycle, number of previous IVF trials, anesthesia type, duration of OPU, the number of collected oocytes, and complications. The study group was divided according to ovarian response in terms of the number of obtained oocytes: 1ŌĆō5 oocytes: poor responder (PR), 6-13 oocytes: normal responder (NR), and 14-20 oocytes: high responder (HR).
The following complications were recorded: severe pain, bleeding, urinary tract infection, ruptured endometrioma, and pelvic abscess. Severe pain was defined as the presence of pain even after the use of non-opioid analgesics, with a score of 6 or more when patients were asked to score their pain between 1 and 10 on a visual analog scale (VAS). The VAS is a validated, subjective measure for acute and chronic pain. Scores were recorded by a handwritten mark on a 10-cm line that represents a continuum between ŌĆ£no painŌĆØ and ŌĆ£worst pain ever sufferedŌĆØ [20]. Patients with a diagnosis of urinary tract infection and complaints after the procedure (burning while urinating and dysuria) were diagnosed by performing a complete urine test and urine culture, and were given appropriate treatment. During the procedure, we sometimes had to pass through endometriomas while trying to obtain access to the follicle and retrieve oocytes. If we aspirated the endometrioma content, we considered it as a case of endometrioma rupture. The only complication requiring hospitalization was pelvic abscess in 1 patient, in whom laparoscopic bilateral salpingectomy and abscess evacuation were performed.
All analyses were performed using IBM SPSS ver. 21 (IBM Corp., Armonk, NY, USA). Q-Q and histogram plots were used to determine whether variables were normally distributed. Data are given as mean┬▒standard deviation or median (range) for continuous variables according to the normality of distribution, and as frequency (percentage) for categorical variables. Normally distributed variables were analyzed with one-way analysis of variance. Pairwise comparisons of these variables were performed with the Tamhane test. Non-normally-distributed variables were analyzed with the Kruskal Wallis test. Pairwise comparisons of these variables were performed with the Bonferroni correction. Categorical variables were analyzed with the chi-square test. Multiple logistic regression analysis (backward conditional method) was performed to identify significant risk factors for complications. Two-tailed p-values of less than 0.05 were considered to indicate statistical significance.
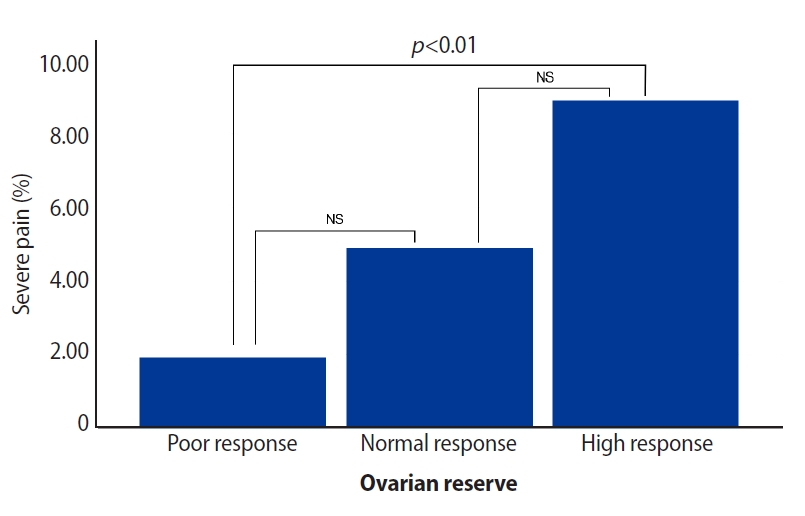
The mean age of the study group was 29.91┬▒5.20 years (range, 21ŌĆō38 years). Patients with low, normal, and high ovarian response were found to be similar in terms of age, BMI and smoking. The frequency of polycystic ovarian syndrome was higher in the HR than in other groups (p<0.001). AMH levels were higher (p<0.001) and day-3 FSH levels were lower (p<0.001) in patients with HR than in patients with PR. In patients with HR, the number of previous trials (p<0.001) was lower, while the operation duration (p<0.001) and the number of retrieved oocytes (p<0.001) were significantly higher than in other patients (Table 1). The frequency of complications (p=0.003) and severe pain (p=0.002) were higher in patients with HR than in patients with PR. The most frequent complication was severe pain (Table 2, Figure 1).
We performed multiple logistic regression analysis to identify significant risk factors for complications. We found that individuals with NR had a 2.947-fold higher risk of complications than individuals with PR (odds ratio [OR], 2.947; 95% confidence interval [CI], 1.123ŌĆō7.736; p=0.028), and individuals with HR had a 7.448-fold higher risk of complications than individuals with PR (OR, 7.448; 95% CI, 2.787ŌĆō19.908; p<0.001). In addition, we found that a higher number of previous IVF trials was associated with an increased risk of complications (p=0.001) (Table 3). Other variables included in the model were found to be non-significant, as follows:, age (p=0.646), BMI (p=0.129), smoking status (p=0.134), cause of infertility (p=0.163), AMH levels (p=0.426), day-3 FSH values (p=0.488), peak E2 levels (p=0.805), type of anesthesia (p=0.629), and procedure duration (p=0.587).
The OPU procedure is the most important stage of complication development in IVF. The majority of complications are minor, but severe complications may occur rarely and can cause life-threatening problems. In this study, the relationship between ovarian response and complications during OPU was investigated. It was found that, as the ovarian reserve increased, AMH and peak E2 levels increased and day 3 FSH levels decreased. The number of oocytes collected and the time spent during the procedure were higher in patients with HR. The most common complication was severe pain, and it was also determined that a higher number of collected oocytes was associated with an increased frequency of complications, especially pain.
Various biochemical markers can be used to estimate ovarian reserve, including FSH, the ratio of FSH to luteinizing hormone, E2, inhibin B, and AMH [21]. Among these, AMH (a hormone secreted from granulosa cells) has been shown to be correlated with the antral follicle count in numerous studies [22-25]. Kim et al. [26] compared AMH and FSH levels with regard to ovarian reserve and reported that the AMH level was high and FSH level was low in patients with diminished ovarian reserve. In our study, in accordance with the literature, it was found that patients with HR had higher AMH and peak cycle E2 levels, but lower day-3 FSH levels.
Although OPU is performed with transvaginal ultrasonography, various complications may occur during and after the procedure. In prior studies, the complications frequently encountered after OPU were bleeding, infection, and severe pain. After OPU, it was reported that 2%ŌĆō3% of patients experienced severe pain that could last up to the second day [27,28]. In our study, the most common complication (detected in 5.7% of cases) was severe pain, which is a higher rate than reported in previous studies. This may be due to the fact that, in our study, patients who expressed their pain as 6 or higher on the VAS scale were defined as having severe pain. Instead, other studies defined severe pain as pain requiring hospitalization. The use of anesthesia during the operation and analgesics after OPU at different doses and at different times may also have affected the level of pain. In most studies, the most frequently reported complication after OPU was vaginal bleeding [9]. The majority of vaginal bleeding events reported after OPU are mild, and severe bleeding is very rare. Furthermore, case reports describing intrabdominal bleeding after OPU indicate that this complication is exceedingly rare [28,29]. In our study, bleeding developed in 0.3% of patientsŌĆöa similar result to those reported in much of the literature [4]. The second most frequently reported complication of OPU is infection [9]. Infections have been reported at different frequencies (range, 0.03%ŌĆō0.6%); however, serious consequences have been demonstrated [30,31]. In our study, occurred in 0.6% of patients. We applied a single dose of cefazolin (1 g) to our patients during the procedure, which may have reduced the frequency of infections. The frequency of complications detected in our study is consistent with the literature, indicating that they are usually predictable.
As the number of retrieved oocytes increases, the time spent during the operation and the risk of complication development due to OPU could also increase. As expected, a higher number of retrieved oocytes translated to an increased frequency of complications, especially severe pain. This may be a direct result of the increased duration of the procedure. Liberty et al. [32] showed that the frequency of complications related to OPU was higher in polycystic ovarian syndrome patients, possibly because more oocytes are retrieved in these patients. Levi-Setti et al. [4] showed that an increased number of oocytes collected and a longer time spent collecting oocytes is associated with the risk of complications. Ludwig et al. [33] reported that after oocyte retrieval, severe to very severe pain occurred in 3.1% of patients, and the pain increased in parallel with the number of oocytes collected. In another study, Singhal et al. [34] showed that the pain scores of patients with an OPU procedure duration exceeding 12 minutes were significantly higher than those with a shorter duration. However, they reported that the pain score was not correlated with the number of oocytes collected. It is therefore important to note that each oocyte retrieval procedure (and its length) could increase the probability of post-procedural adverse effects.
Recent studies reported additional concerns regarding OPU for severely obese women (BMI >40 kg/m2), including anesthetic-related risks and technical difficulties such as an inability to access the ovaries [4,35]. In this article, BMI was not associated with an increased frequency of complications. This may have been due to the exclusion of patients with obesity from our study. BMI may not be associated with OPU complications up to a certain threshold. This issue could be explored in greater detail in studies that include patients with a BMI of 30 kg/m2 and above.
Finally, we found that higher number of previous IVF trials was associated with an increased risk of complications. In patients with less ovarian reserve, more IVF attempts may be required to achieve pregnancy. In other words, patients with more trials may have less ovarian reserve. Indirectly, patients with an increased number of IVF attempts may have an increased risk of complications because they have poor ovarian reserve.
This study has several limitations, such as those associated with the nature of retrospective studies. It has been shown that the experience of the surgeon performing OPU is related to the risk of complications [4]. In our study, OPU was performed by a single doctor with 15 years of experience; thus, the frequency of complications should be assessed in accordance with this fact. In previous studies, the aspiration needle gauge has been shown to affect pain levels after OPU [10,11,36]. In our study, the fact that these conditions facilitate or reduce pain levels may cause misinterpretations in comparison with other studies. Lastly, patients who may have sought treatment at different health institutions due to complications after OPU could not be evaluated in our study. This may have caused the frequency of complications to be underestimated.
In conclusion, it was found that the most common complication in patients who underwent OPU was pain. The risk of complications was higher in patients with normal and high ovarian response than in those with poor ovarian response. We also found that an increased number of previous IVF cycles was associated with a higher risk of complications related to OPU. As pain is common in these patients, it is concluded that pain management may be crucial. Patients with normal or high ovarian response during OPU should be monitored more closely for the development of complications. Comprehensive and prospective studies on OPU complications would be useful.
Table┬Ā1.
Summary of patient characteristics with regard to ovarian reserve
| Variable |
Ovarian reserve |
p-value | ||
|---|---|---|---|---|
| PR (n=209) | NR (n=266) | HR (n=314) | ||
| Age (yr) | 32 (21ŌĆō38) | 32 (21ŌĆō38) | 32 (21ŌĆō38) | 0.894 |
| Body mass index (kg/m2) | 24.46┬▒2.48 | 24.45┬▒2.44 | 24.85┬▒2.77 | 0.106 |
| Smoking | 55 (26.32) | 82 (30.83) | 82 (26.11) | 0.389 |
| Cause of infertility | ||||
| ŌĆāPolycystic ovarian syndrome | 0 | 0 | 97 (30.89) | <0.001 |
| ŌĆāTubal factor | 15 (7.18) | 30 (11.28) | 47 (14.97) | |
| ŌĆāUnexplained infertility | 82 (39.23) | 97 (36.47) | 70 (22.29) | |
| ŌĆāEndometriosis | 28 (13.40) | 57 (21.43) | 35 (11.15) | |
| ŌĆāMale factor | 64 (30.62) | 69 (25.94) | 59 (18.79) | |
| ŌĆāPreimplantation genetic diagnosis | 0 | 9 (3.38) | 6 (1.91) | |
| ŌĆāFertility preservation | 20 (9.57) | 4 (1.50) | 0 | |
| Anti-M├╝llerian hormone (IU/mL) | 0.5 (0.2ŌĆō0.9)a) | 2 (1.1ŌĆō4.22)b) | 5.6 (5.1ŌĆō11)c) | <0.001 |
| Day-3 FSH (IU/mL) | 12.20┬▒1.17a) | 7.20┬▒1.59b) | 6.01┬▒1.83c) | <0.001 |
| Peak E2 level (pg/mL) | 562 (256ŌĆō1,200)a) | 1,652 (869ŌĆō2,625)b) | 3,250 (1,010ŌĆō5,214)c) | <0.001 |
| Previous IVF trial (n) | 3 (1ŌĆō6)a) | 2 (1ŌĆō6)b) | 2 (1ŌĆō4)c) | <0.001 |
| ŌĆā1 | 4 (1.91) | 54 (20.3) | 136 (43.31) | <0.001 |
| ŌĆā2 | 66 (31.58) | 105 (39.47) | 140 (44.59) | |
| ŌĆā3 | 100 (47.85) | 76 (28.57) | 32 (10.19) | |
| ŌĆāŌēź 4 | 39 (18.66) | 31 (11.65) | 6 (1.91) | |
| Anesthesia | ||||
| ŌĆāLocal | 5 (2.39) | 2 (0.75) | 0 | 0.016 |
| ŌĆāGeneral | 204 (97.61) | 264 (99.25) | 314 (100) | |
| Duration of OPU (min) | 12.04┬▒2.66a) | 14.34┬▒2.27b) | 22.54┬▒4.59c) | <0.001 |
| Collected oocytes | 4 (1ŌĆō5)a) | 10 (7ŌĆō12)b) | 17 (14ŌĆō23)c) | <0.001 |
Table┬Ā2.
Complications after transvaginal ultrasoundŌĆōguided oocyte pick-up procedures
Table┬Ā3.
Significant risk factors for complications in multiple logistic regression analysis
References
1. Mascarenhas MN, Flaxman SR, Boerma T, Vanderpoel S, Stevens GA. National, regional, and global trends in infertility prevalence since 1990: a systematic analysis of 277 health surveys. PLoS Med 2012;9:e1001356.



2. Drakopoulos P, Bardhi E, Boudry L, Vaiarelli A, Makrigiannakis A, Esteves SC, et al. Update on the management of poor ovarian response in IVF: the shift from Bologna criteria to the Poseidon concept. Ther Adv Reprod Health 2020;14:2633494120941480.



3. Rani K, Paliwal S. A brief review on in-vitro fertilization (IVF): an advanced and miraculous gateway for infertility treatments. World J Pharm Sci 2014;3:647-58.
4. Levi-Setti PE, Cirillo F, Scolaro V, Morenghi E, Heilbron F, Girardello D, et al. Appraisal of clinical complications after 23,827 oocyte retrievals in a large assisted reproductive technology program. Fertil Steril 2018;109:1038-43.


5. Schulman JD, Dorfmann A, Jones S, Joyce B, Hanser J. Outpatient in vitro fertilization using transvaginal oocyte retrieval and local anesthesia. N Engl J Med 1985;312:1639.

6. Maxwell KN, Cholst IN, Rosenwaks Z. The incidence of both serious and minor complications in young women undergoing oocyte donation. Fertil Steril 2008;90:2165-71.


7. Aragona C, Mohamed MA, Espinola MS, Linari A, Pecorini F, Micara G, et al. Clinical complications after transvaginal oocyte retrieval in 7,098 IVF cycles. Fertil Steril 2011;95:293-4.


8. Muzii L, Marana R, Caruana P, Catalano GF, Mancuso S. Laparoscopic findings after transvaginal ultrasound-guided aspiration of ovarian endometriomas. Hum Reprod 1995;10:2902-3.


9. Pargianas M, Salta S, Fiorentzis S, Kalampoki LG, Beck R, Vizziello D, et al. Complications in oocyte retrieval. In: Malvasi A, Baldini D, editors. Pick up and oocyte management. Cham: Springer; 2020. p. 305-37.
10. Leung AS, Dahan MH, Tan SL. Techniques and technology for human oocyte collection. Expert Rev Med Devices 2016;13:701-3.


11. Rose BI. Approaches to oocyte retrieval for advanced reproductive technology cycles planning to utilize in vitro maturation: a review of the many choices to be made. J Assist Reprod Genet 2014;31:1409-19.



12. Vaiarelli A, Cimadomo D, Ubaldi N, Rienzi L, Ubaldi FM. What is new in the management of poor ovarian response in IVF? Curr Opin Obstet Gynecol 2018;30:155-62.


13. Ubaldi FM, Rienzi L, Ferrero S, Baroni E, Sapienza F, Cobellis L, Greco E. Management of poor responders in IVF. Reprod Biomed Online 2005;10:235-46.

14. Blumenfeld Z. What is the best regimen for ovarian stimulation of poor responders in ART/IVF? Front Endocrinol (Lausanne) 2020;11:192.



15. Blumenfeld Z. Premature ovarian failure: etiology and possible prevention. Expert Rev Endocrinol Metab 2009;4:173-81.


17. Lee HC, Lyndon A, Blumenfeld YJ, Dudley RA, Gould JB. Antenatal steroid administration for premature neonates in California. Obstet Gynecol 2011;117:603-9.



18. Skillern A, Rajkovic A. Recent developments in identifying genetic determinants of premature ovarian failure. Sex Dev 2008;2:228-43.


19. Barrenetxea G, Agirregoikoa JA, Jimenez MR, de Larruzea AL, Ganzabal T, Carbonero K. Ovarian response and pregnancy outcome in poor-responder women: a randomized controlled trial on the effect of luteinizing hormone supplementation on in vitro fertilization cycles. Fertil Steril 2008;89:546-53.


20. Hawker GA, Mian S, Kendzerska T, French M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res (Hoboken) 2011;63 Suppl 11:S240-52.


22. de Vet A, Laven JS, de Jong FH, Themmen AP, Fauser BC. Reprint of: Antim├╝llerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril 2019;112(4 Suppl1): e183-8.


23. Durlinger AL, Visser JA, Themmen AP. Regulation of ovarian function: the role of anti-M├╝llerian hormone. Reproduction 2002;124:601-9.


24. Hansen KR, Hodnett GM, Knowlton N, Craig LB. Correlation of ovarian reserve tests with histologically determined primordial follicle number. Fertil Steril 2011;95:170-5.


25. van Rooij IA, Broekmans FJ, te Velde ER, Fauser BC, Bancsi LF, de Jong FH, et al. Serum anti-Mullerian hormone levels: a novel measure of ovarian reserve. Hum Reprod 2002;17:3065-71.


26. Kim SW, Kim YJ, Shin JH, Kim H, Ku SY, Suh CS, et al. Correlation between ovarian reserve and ─▒ncidence of ectopic pregnancy after ─▒n vitro fertilization and embryo transfer. Yonsei Med J 2019;60:285-90.



27. Ozalt─▒n S, Kumbasar S, Savan K. Evaluation of complications developing during and after transvaginal ultrasound - guided oocyte retrieval. Ginekol Pol 2018;89:1-6.


28. Siristatidis C, Chrelias C, Alexiou A, Kassanos D. Clinical complications after transvaginal oocyte retrieval: a retrospective analysis. J Obstet Gynaecol 2013;33:64-6.


29. Bodri D, Guillen JJ, Polo A, Trullenque M, Esteve C, Coll O. Complications related to ovarian stimulation and oocyte retrieval in 4052 oocyte donor cycles. Reprod Biomed Online 2008;17:237-43.


30. Tsai YC, Lin MY, Chen SH, Chung MT, Loo TC, Huang KF, et al. Vaginal disinfection with povidone iodine immediately before oocyte retrieval is effective in preventing pelvic abscess formation without compromising the outcome of IVF-ET. J Assist Reprod Genet 2005;22:173-5.



31. Weinreb EB, Cholst IN, Ledger WJ, Danis RB, Rosenwaks Z. Should all oocyte donors receive prophylactic antibiotics for retrieval? Fertil Steril 2010;94:2935-7.


32. Liberty G, Hyman JH, Eldar-Geva T, Latinsky B, Gal M, Margalioth EJ. Ovarian hemorrhage after transvaginal ultrasonographically guided oocyte aspiration: a potentially catastrophic and not so rare complication among lean patients with polycystic ovary syndrome. Fertil Steril 2010;93:874-9.


33. Ludwig AK, Glawatz M, Griesinger G, Diedrich K, Ludwig M. Perioperative and post-operative complications of transvaginal ultrasound-guided oocyte retrieval: prospective study of >1000 oocyte retrievals. Hum Reprod 2006;21:3235-40.


34. Singhal H, Premkumar PS, Chandy A, Kunjummen AT, Kamath MS. Patient experience with conscious sedation as a method of pain relief for transvaginal oocyte retrieval: a cross sectional study. J Hum Reprod Sci 2017;10:119-23.



- TOOLS