|
 |
- Search
| Clin Exp Reprod Med > Volume 41(1); 2014 > Article |
Abstract
Methods
Medical records from 1,212 women with amenorrhea who visited the Department of Obstetrics and Gynecology, Asan Medical Center, between January 1989 and December 2011 were retrospectively reviewed. Amenorrhea was categorized as either primary or secondary.
Results
Primary amenorrhea was identified in 132 of the patients (10.9%) and secondary amenorrhea in 1,080 (89.1%). The most frequent causes of primary amenorrhea were gonadal dysgenesis (28.0%, 37/132); Mayer-Rokitansky-Küster-Hauser syndrome (20.0%, 27/132); and constitutional delay and androgen insensitivity syndrome (8.3%, 11/132; 8.3%, 11/132, respectively). Secondary amenorrhea was due to polycystic ovary syndrome (48.4%, 523/1,080); premature ovarian insufficiency (14.0%, 151/1,080); and nutrition-related hypogonadotropic hypogonadism (8.3%, 90/1,080).
Amenorrhea may result from a number of different conditions. Through knowledge of the physiology of menstruation is essential to understand the various causes of amenorrhea. The normal menstrual cycle involves complex interactions between the hypothalamic-pituitary axis, the ovaries and the outflow tract. In this complex and highly regulated sequence of events, any disruption or functional abnormality in the hypothalamic-pituitary-ovarian axis can result in abnormal menstruation or amenorrhea. Endocrinological disease and structural abnormalities of the outflow tract can cause amenorrhea [1,2]. For the treatment of these amenorrheic patients, there is a need for data about the causes of amenorrhea.
Our previous study of 158 patients reported in 1999 showed that the most common cause of primary amenorrhea was Turner syndrome and the most common cause of secondary amenorrhea was polycystic ovary syndrome (PCOS) [3]. The present study was an extension of our previous study to evaluate the cause of amenorrhea in a large case series at our center. Several studies on the cause of primary amenorrhea have been published in Asia [4,5,6]. However, few studies including both primary and secondary amenorrhea have been reported. Most large case series have been reported on from western countries. These studies have been used as a reference regarding the causes of amenorrhea.
Since only a few studies have focused on the causes of amenorrhea among women in Asian countries, the aim of the present study was to determine the most common causes of primary and secondary amenorrhea in Korean women on a large scale.
A retrospective study was conducted in the Gynecologic Endocrinology Clinic, Department of Obstetrics and Gynecology, Asan Medical Center, Korea. The study protocol was in accordance with the Helsinki Declaration, and the Institutional Review Board of our center approved the study.
The medical records of 1,212 patients with amenorrhea, excluding cases due to pregnancy, who were registered in our center from January 1989 to December 2011 were reviewed. Amenorrhea was divided into two categories: primary and secondary.
Women with primary amenorrhea had normal secondary sexual characteristics but no menarche by 15 years of age, or had no secondary sexual characteristics and no menarche by 13 years of age. In secondary amenorrhea, menstruation was absent for 3 cycles in a woman with previously regular menstruation, or for 6 months in a woman with a history of oligomenorrhea [7,8].
Because primary amenorrhea is defined as no menarche and no secondary sexual characteristics over 13 years and premature ovarian insufficiency (POI) occurs in women under the age of 40 [9,10], the patients aged 13 to 40 years (mean±SD, 24.0±6.3 years) were included in this study.
The initial work-up was based on a comprehensive patient history and physical examination. Short stature, under or overweight, and secondary sexual development were evaluated. A history of prescription drug use, previous chemotherapy and radiation (brain, pelvis), weight loss, psychosocial stress, exercise, chronic illness, or eating disorders (anorexia or bulimia nervosa) was determined. A family history (mother and sisters) and specific menstrual history took note of gravida, excessive blood loss during delivery, surgical history, and history of pelvic organ disease [1,7].
Laboratory tests were performed to identify systemic disease. In women with secondary sexual characteristics, pregnancy was ruled out. In patients that were confirmed as not pregnant but with amenorrhea, prolactin and TSH levels were assayed. If the levels of both of these hormones were normal, a progesterone challenge test and estrogen/progesterone challenge test were performed to detect endogenous estrogen, which affects the endometrium, and to evaluate the patency of the outflow tract. In case of a positive estrogen/progesterone challenge test, FSH and LH levels were measured. An elevation in the levels of these hormones is consistent with an ovarian abnormality (gonadal failure), whereas normal or low levels points to a pituitary or hypothalamic abnormality (of either pituitary or hypothalamic origin) [7].
Based on these work-ups, patients with amenorrhea were classified with respect to the organ compartment involved and a diagnosis was subsequently further specified. Anatomic defects in the outflow tract, primary dysfunction of the ovary, disruption of pituitary or hypothalamic function and endocrine disease were included. In all cases, the initial evaluation procedure was similar; however, in patients with primary amenorrhea, the major difference was including a procedure to determine the presence of the uterus.
Among the 1,212 patients, 132 (10.9%) had primary amenorrhea and 1,080 (89.1%) had secondary amenorrhea. At the time of registration at our clinic, patients with primary and secondary amenorrhea were 19.1±4.1 and 24.6±6.2 years of age, respectively (Table 1). Most of the patients with primary amenorrhea were within ages 11 to 20 (67.4%, 89/132), and a slight majority of those with secondary amenorrhea were ages 21 to 30 (56.0%, 604/1,080).
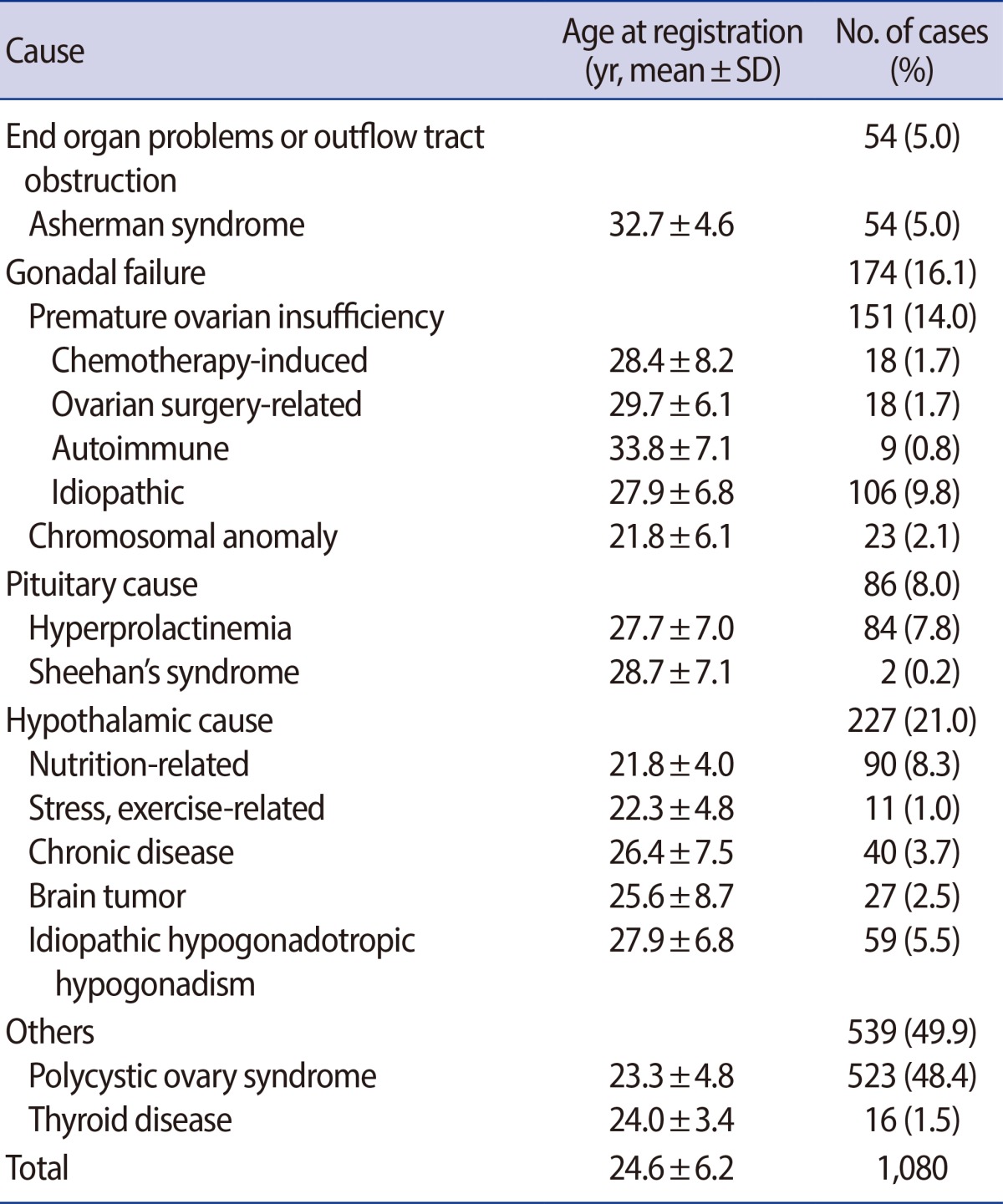
The common causes of primary amenorrhea were gonadal dysgenesis (28.0%, 37/132), the Mayer-Rokitansky-Küster-Hauser (MRKH) syndrome (20.0%, 27/132), constitutional delay (8.3%, 11/132), and androgen insensitivity syndrome (8.3%, 11/132) (Table 2). In patients with secondary amenorrhea, PCOS (48.4%, 523/1,080), POI (14.0%, 151/1,080), and nutrition-related hypogonadotropic hypogonadism (8.3%, 90/1,080) were the three most common causes (Table 3).
The treatment of amenorrhea requires first determining its cause, so a thorough history and physical examination, accompanied by imaging studies and measurements of hormone levels are important to narrow the differential diagnosis. Treatment goals include the prevention of complications such as osteoporosis and endometrial hyperplasia from the associated abnormal hormone levels, and the preservation of fertility [1,9]. In many cases of primary amenorrhea, treatment should be started immediately following the diagnosis, with the primary therapeutic goal being to facilitate normal secondary sexual development. In general, the treatment of amenorrhea must be patient-tailored according to the causative factor.
In this study, gonadal dysgenesis was the most commonly diagnosed condition in primary amenorrhea. Turner syndrome is the most frequent cause of female gonadal dysgenesis. Among 37 cases of gonadal dysgenesis, 28 cases underwent karyotype analysis. Twenty cases had an abnormal karyotype including 45,X (n=8), mosaicism (n=11), and 46,XY (n=1). Detection of a Y chromosome mandates the surgical removal of the streak gonad as soon as possible because of their high likelihood of being the source of a malignancy after puberty [10,11]. In the case of 46,XY (Swyer syndrome) case, gonadectomy was performed immediately after diagnosis. Eight cases had a normal karyotype; however in 9 cases a karyotype analysis was not performed. A number of the patients with primary amenorrhea were young fertile women under the age 20. Where appropriate, either the correction of any anatomic abnormality or a gonadectomy, performed based on the results of karyotyping, should be considered in such cases. Karyotype analysis should be performed if there are grounds for suspecting that the patient is not genetically female.
The findings of the present study are quite consistent with Reindollar et al. [12], which showed that the most common cause of amenorrhea in the American population was gonadal dysgenesis (48.5%). Furthermore, the recent study by Klein and Poth [8] reported that primary amenorrhea is the result of primary ovarian insufficiency (e.g., Turner syndrome) or anatomic abnormalities (e.g., Müllerian agenesis). However, Tanmahasamut et al. [4] noted that Müllerian agenesis was the most prevalent cause in primary amenorrhea in Thailand. They reported that the three most common causes of primary amenorrhea were Müllerian agenesis (39.7%), gonadal dysgenesis (35.3%), and hypogonadotropic hypogonadism (9.2%). There was some limitation in that this study was conducted in a single tertiary care hospital, which would have been affected by a referral pattern, but it is the largest case series of primary amenorrhea published yet in Asia.
As reported by the American Society of Reproductive Medicine Practice Committee in 2008 [7], the four most common causes of amenorrhea were polycystic ovary syndrome, followed by hypothalamic amenorrhea, ovarian failure, and hyperprolactinemia.
Compatible with western studies, secondary amenorrhea was most often due to PCOS in our center. According to the revised Rotterdam 2003 criteria [13], PCOS is diagnosed if two of the following are present: clinical and biochemical signs of hyperandrogenism, oligo- or anovulation, and polycystic ovaries on ultrasonography. Other etiologies, such as congenital adrenal hyperplasia, androgen-secreting tumor, and Cushing syndrome, must be excluded. Before the Rotterdam 2003 criteria, despite the lack of consensus, we diagnosed PCOS according to the following criteria; chronic anovulation and clinical and/or biochemical signs of hyperandrogenism, and exclusion of other etiologies.
The prevalence of an abnormal karyotype in POI of secondary amenorrhea appeared low compared with cases of gonadal failure causing primary amenorrhea. Most of the causes related to POI were idiopathic, and they were attributed to chemotherapeutic agents, ovarian operations and autoimmune disease. The patients with autoimmune disease including systemic lupus erythematosus or autoimmune thyroiditis were referred from rheumatology. These patients did not have other causes of amenorrhea besides their imunologic problems. Immunologic tests were not performed on all of the patients with POI.
Many reproductive disorders appear to be related to nutritional status. In Table 3, nutrition-related hypothalamic causes of amenorrhea include severe weight loss, dieting, and eating disorders, such as anorexia nervosa and bulimia nervosa, in which the starved conditions leads to menstrual and fertility abnormalities. Women with functional hypothalamic amenorrhea have reduced central GnRH, resulting in low FSH and LH levels and thus amenorrhea. Recently, an increase in the prevalence of nutrition-related amenorrhea was reported, perhaps due to socio-cultural influences that focus on body image and weight in young women [14,15].
The present study was a retrospective chart review and it therefore had some limitations. For example, information regarding the cause of the amenorrhea could, in some cases, only be partially analyzed due to an insufficient patient work-up. In addition, some patients did not undergo karyotyping. Furher well designed prospective studies are needed to compensate for these problems.
Furthermore, this study was conducted in a tertiary-care hospital, which implies differences in referral patterns compared to other centers. Thus, patients with the MRKH syndrome or Müllerian agenesis were referred to our center for surgery, perhaps explaining why a former comprised the frequent cause of primary amenorrhea in our series.
In conclusion, the present study is the largest case series in Asia to examine women with amenorrhea. The proportions of the various causes of primary and secondary amenorrhea determined in our study were often different from those reported in the largest western studies and other Asian studies. Thus, ethnic, environmental, and genetic factors appear to play a role in the development of amenorrhea.
References
2. Master-Hunter T, Heiman DL. Amenorrhea: evaluation and treatment. Am Fam Physician 2006;73:1374-1382.PMID: 16669559.

3. Chae HD, Kang EH, Chu HS, Kim JH, Kang BM, Chang YS. Clinical characteristics of amenorrhea according to the etiological classification. Korean J Obstet Gynecol 1999;42:975-980.
4. Tanmahasamut P, Rattanachaiyanont M, Dangrat C, Indhavivadhana S, Angsuwattana S, Techatraisak K. Causes of primary amenorrhea: a report of 295 cases in Thailand. J Obstet Gynaecol Res 2012;38:297-301.PMID: 22070792.


5. Rattanachaiyanont M, Kunathikom S, Angsuwattana S, Techatraisak K, Mekmahan O, Karavagul C, et al. Primary amenorrhoea: a retrospective study at Siriraj Hospital. J Med Assoc Thai 1997;80:619-625.PMID: 10904563.

6. Choi YM, Ku SY, Chae HJ, Jeong HJ, Kim KD, Kim H, et al. The causes of primary amenorrhea. Korean J Obstet Gynecol 2001;44:1834-1837.
7. Practice Committee of American Society for Reproductive Medicine. Current evaluation of amenorrhea. Fertil Steril 2008;90:S219-S225.PMID: 19007635.


8. Klein DA, Poth MA. Amenorrhea: an approach to diagnosis and management. Am Fam Physician 2013;87:781-788.PMID: 23939500.

9. Reichman DE, Laufer MR. Congenital uterine anomalies affecting reproduction. Best Pract Res Clin Obstet Gynaecol 2010;24:193-208.PMID: 19897423.


10. Simpson JL, Rajkovic A. Ovarian differentiation and gonadal failure. Am J Med Genet 1999;89:186-200.PMID: 10727994.


12. Reindollar RH, Tho SP, McDonough PG. Delayed puberty: an update study of 326 patients. Trans Am Gynecol Obstet Soc 1989;8:146-162.
13. Rotterdam ESHRE/ASRM-Sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod 2004;19:41-47.PMID: 14688154.


14. Gordon CM. Clinical practice. Functional hypothalamic amenorrhea. N Engl J Med 2010;363:365-371.PMID: 20660404.


15. Skalba P, Guz M. Hypogonadotropic hypogonadism in women. Endokrynol Pol 2011;62:560-567.PMID: 22144224.

-
METRICS

- Related articles in Clin Exp Reprod Med
-
Marriage in Korea I. Evidence of Changing Attitudes and Practice1975 ;2(2)