Comparison between intracytoplasmic sperm injection and intracytoplasmic morphologically selected sperm injection in oligo-astheno-teratozoospermia patients
Article information
Abstract
Objective
The aim of this study was to evaluate the efficiency of the intracytoplasmic morphologically selected sperm injection (IMSI) technique compared with conventional ICSI and previous ICSI attempts in oligo-astheno-teratozoospermia (OAT) patients.
Methods
The sperms were selected under high magnification (6,600×) and used to induce fertilization in previous ICSI patients by IMSI. These results were compared with previous conventional ICSI cycles in patients with OAT infertility.
Results
These results demonstrated no significant difference in the fertilization rate between IMSI and previous ICSI cycles (67.7% vs. 65.0%). However, the pregnancy and implantation rates with IMSI were significantly higher than those of the ICSI cycles (33.3% vs. 12.5% and 14.6% vs. 5.4%, respectively; p<0.05). The miscarriage rate among pregnant patients (18.2% vs. 37.5%) showed no statistically significant difference between groups.
Conclusion
Compared to conventional ICSI, this study found that IMSI increased the IVF-ET success rates in patients with OAT.
Introduction
ICSI has been utilized as an ordinary fertilization method in artificial reproductive technological cycles to overcome severe male infertility, since its introduction in 1992 [1].
Injected spermatozoa are typically randomly selected, and the selection by an embryologist is based on only the sperms' morphological characteristics and motility in the ICSI technique. However, many studies have reported that the presence of various defects in the spermatozoa is associated with reduced fertilization, and pregnancy rates, and with an increased risk of miscarriage [2,3,4].
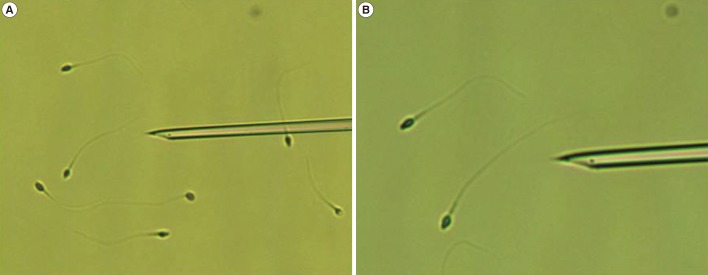
Sperm selection is usually performed under an optical magnification of approximately 200× or 400× in ICSI techniques. However, the low magnification used in routine microscopy results in limitations in identifying sperm organellar malformations, particularly vacuoles in the sperm head, where major defects, such as abnormal sperm head size proportions and midpiece abnormalities were observed [5]. Evaluation of the normal morphology of the nucleus in sperm should be performed accurately using a high magnification and resolution [6,7,8,9].
Bartoov et al. [10,11] have developed a new method known as motile sperm organellar morphology examination (MSOME), which evaluates the real-time movement of sperm. They made it possible to obtain a perfect sperm with a normal nucleus by combining MSOME and micromanipulation technology. This process is known as the modified IVF process, or intracytoplasmic morphologically selected sperm injection (IMSI), because only one moving sperm is selected when performing ICSI using MSOME.
We adopted IMSI for patients who underwent ICSI due to oligo-astheno-teratozoospermia (OAT) in previous cycles since February 2011.
The aim of the present prospective study was to assess the clinical advantages of the IMSI procedure in the treatment of patients with male factors, particularly OAT, by comparing the outcomes of the previous ICSI cycles.
Methods
1. Patients and design
This study was performed in 66 cycles that had undergone ICSI treatment in the previous cycle for OAT infertility from May 2010 to June 2012 at Seoul Maria Fertility Hospital. The patients provided informed consent for IMSI between December 2011 and October 2012, and the study was approved by the Institutional Review Board of the Maria Fertility Hospital.
2. Semen evaluation and preparation
Analysis of OAT, was confirmed by the World Health Organization (2010) criteria: sperm concentration <15×106/mL, motility <40%, and strict morphology <4%. The semen was prepared by centrifugation on a density gradient (Sperm Filter, Cryos, Aarhus, Denmark) using MOPSE/MRC (Biosupply, Seoul, Korea) supplemented with 10% serum substitute supplement (Irvine Scientific, Irvine, CA, USA).
3. Ovarian stimulation and oocyte retrieval
Women were treated with GnRH agonist (Superfact, Sanofi-Aventis, Frankfurt, Germany) using the long protocol or GnRH antagonist protocol and recombinant FSH (Gonal F, Merk Serono, Darmstadt, Germany). When two or more follicles reached 18 mm in diameter, a dose of 10,000 IU of hCG (Ovidrel, Merk Serono) was administered. Oocyte retrieval was performed 36 hours after hCG injection.
4. Conventional ICSI
After removing the cumulus cell attached to the egg with MRC#ICSI (Biosupply) containing 0.1% hyaluronidase (Biosupply), the collected eggs were cultured in Sydney IVF fertilization medium (Cook, Brisbane, Australia). ICSI was performed using an inverted Nikon microscope (TE 2000 U, Nikon, Kawasaki, Japan) equipped with a Narishige manipulator (Narishige, Tokyo, Japan). Sperm selection was performed under 200× to 400× magnification (Figure 1), and normal-looking spermatozoa were injected into the oocyte under 200× magnification.
5. IMSI
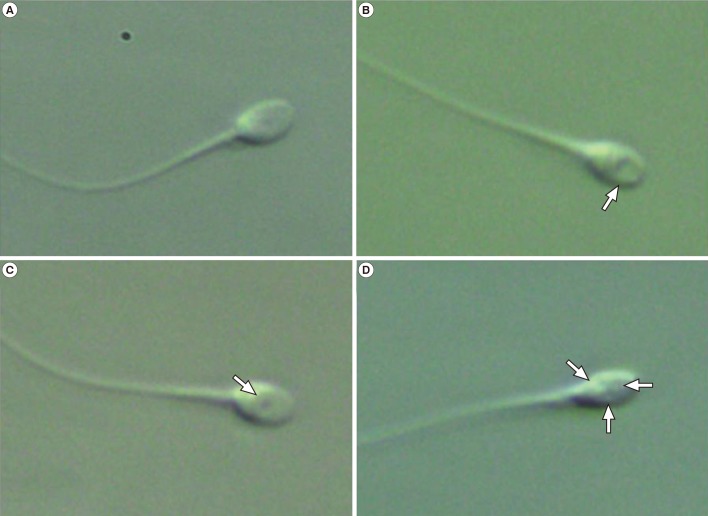
All motile spermatozoa were analyzed under high-magnification (6,600×) (Figure 2) microscopy (TE 2000 U, Nikon) incorporating 60× air objectives with modulation contrast illumination (RI IMSI, RI, Cornwall, UK). The images were captured using a DC2 IMSI camera (Nikon) with DICOM compliant medical imaging monitor (EIZO, Ishikawa, Japan). Sperm selection was performed in drops of PVP (Irvine Scientific), and the drops were covered with sterile mineral oil (Biosupply). Normal sperm morphology was defined as smooth, symmetric, having an oval configuration, with average length, and width limits (4.75±0.28 µm and 3.28±0.20 µm, respectively), with a homogeneous nuclear chromatin mass, with no regional nuclear disorders, and containing no more than one small vacuole with a borderline diameter of 0.78±0.18 µm [10].
6. Embryo culture and transfer
Fertilization was assessed 16 to 18 hours after insemination by the appearance of two distinct pronuclei and two polar bodies. The zygotes were cultured in Sydney IVF cleavage medium (Cook) for 37℃ in 6% CO2, 5% O2, and 89% N2. The embryos were then transferred three days after oocyte retrieval.
7. Confirmation of pregnancy
If the β-hCG concentration in the blood was positive 14 days after egg collection and the trace test after one week showed a continuous increase, and if the result of vaginal ultrasonography performed at 6 to 7 weeks of pregnancy showed a gestational sac, then clinical pregnancy was verified. The implantation rate was shown by the ratio of the number of implanted embryos to the number of embryonic sacs implanted in the uterus. Miscarriage was defined as a fetal loss prior to 12 weeks gestation despite the presence of a gestational sac in the first trimester ultrasound. Ongoing pregnancy was judged by fetal cardiac activity after 12 weeks.
8. Statistical analysis
Statistical analysis was performed with SPSS ver. 12.0 (SPSS Inc., Chicago, IL, USA). A Student's t-test was used to compare continuous variables, whereas a chi-squared test was applied to discrete variables. A p-value <0.05 was accepted as significant.
Results
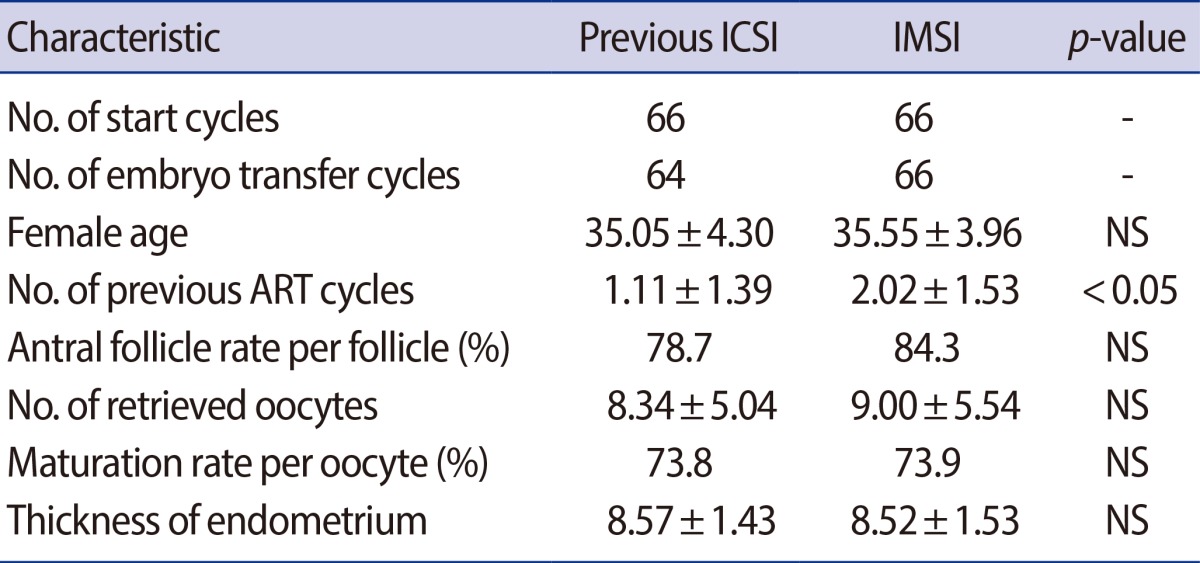
A total of 66 IMSI cycles were compared with the previous ICSI cycles in this study. When the characteristic features of previous ICSI cycles were compared with IMSI cycles, there was no significant difference in female age, retrieved oocytes, or the maturation rate per oocyte (Table 1).
However, the number of IVF-ET procedures, performed in advance, was statistically significantly high in IMSI compared to previous ICSI cycles (2.02±1.53 vs. 1.11±1.39, p<0.05) (Table 1).
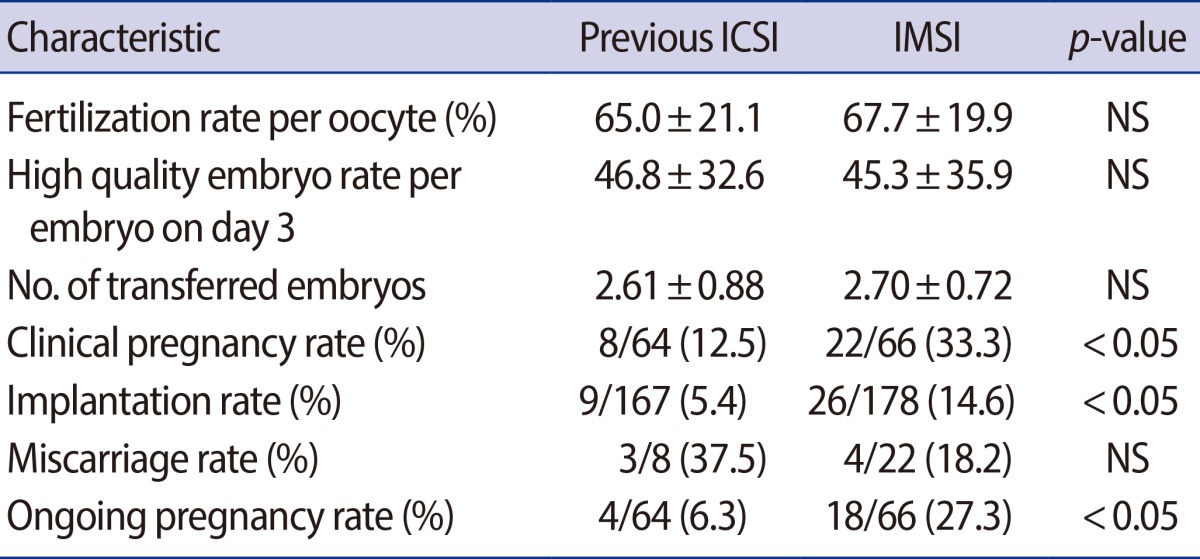
Comparisons of the clinical outcomes between the two groups are presented in Table 2. No significant differences were observed between the two groups with regard to the fertilization rate, high quality embryo rate per embryo on day 3, and transferred embryos. However, the clinical pregnancy rate and implantation rate were significantly higher in IMSI than in previous ICSI cycles (33.3% and 14.6% vs. 12.5% and 5.4%, respectively, p<0.05). There was also a significant difference in the ongoing pregnancy rate (27.3% vs. 6.3%, p<0.05). The miscarriage rate among pregnant patients (18.2% vs. 37.5%) showed no significant difference between the two groups.
Discussion
Introduction of a new method based on fine motile sperm organelle morphology examination in real-time, called MSOME, was developed to examine and select sperm cells. By the use of MSOME criteria with the application of high magnification (>6,600×), subtle morphological features, such as abnormal sperm head proportions, midpiece abnormalities, and the presence of vacuoles in the sperm head, can be characterized.
Since the development of IMSI, these results have reported that the new technology could enhance the clinical IVF results in patients for whom ICSI treatment failed. It increased the pregnancy rate of patients who failed on ICSI more than twice and of female infertility patients with unknown reasons for failure after injection of sperm with no nuclear alteration. These results also demonstrated a 50% decrease in the rate of miscarriage compared to that with general ICSI [12,13,14,15].
We applied the IMSI procedure on patients who were fertilized by ICSI due to OAT in the previous IVF/ET cycle and investigated these effects. This study demonstrated that there were no differences in the fertilization rate or high quality embryo rate between current IMSI and previous ICSI cycles (Table 2).
Mauri et al. [16] concluded that the IMSI procedure did not improve embryo quality on day 2. Many studies have found no correlation between high magnification sperm selection and early embryo development in terms of fertilization, or the rate of top quality embryos on day 3 and day 5 [12,14,17]. Similarly, Vanderzwalmen et al. [3] found no significant difference in the fertilization when sperm selection was performed at a high magnification prior to ICSI compared with conventional ICSI in a sibling oocyte study. Moreover, IMSI provided no significant difference in embryo quality up to day 3, but blastocyst formation and quality were closed associated with the different grades of sperm. The oocytes injected with sperm with no vacuoles or ≤2 small vacuoles in their heads produced significantly higher rates of blastocyst formation (56.3% and 61.4%, respectively) than sperm with either <2 small vacuoles or at least one large vacuole, or a large vacuole and abnormal head shape or other abnormalities (5.1% and 0%, respectively).
When IMSI began, it had been applied to patients who had experienced repeated implantation failures [6,10,11] or displayed high DNA fragmentation [14]. However, a recently published study demonstrated that application of IMSI technology produced satisfactory results in patients with a high level of vacuoles when observed with high definition [18]. Vacuoles of various sizes are known to have a very close relationship with the integrity, accurate denaturation, and chromatin package of sperm DNA [8,9,12,14].
Garolla et al. [9] demonstrated that sperm with vacuoles were related to the integrity of sperm DNA, fragmentation, and abnormal chromosomes. Wilding et al. [17] showed that DNA fragmentation was related to vacuoles in sperm with morphological abnormalities. Recently, Hammoud et al. [19] noted that the presence of large or multiple vacuoles was associated with a higher amount of DNA fragmentation in sperm than the lack of vacuoles. In these research studies, vacuoles might be associated with chromatin condensation failure.
Previous reports have shown that when ICSI was performed with abnormal sperm, especially the presence of vacuoles in injected sperm, it was related to late embryo development. Slow embryonic development with poor development to cleavage is generally due to DNA damage on sperm or aneuploidy of the embryo [7].
Knez et al. [20] observed that IMSI improved embryo development and the clinical outcomes of sperm microinjection in the same infertile couples with male infertility and poor embryo development in previous ICSI attempts. Our results also showed that IMSI improved clinical pregnancy (33.3% vs. 12.5%, p<0.05), implantation (14.6% vs. 5.4%, p<0.05), and the ongoing pregnancy rate (27.3% vs. 6.3%, p<0.05) compared to previous ICSI cycles in the same couples.
In conclusion, IMSI performed at high magnification such as 6,600x has the advantage of enabling the sorting of sperm with vacuoles in their heads or other defects that cannot be observed during conventional ICSI (200×-400×). Clinical IMSI is an effective technique for IVF-ET with positive clinical outcomes such as better implantation and pregnancy rates than conventional ICSI.
Notes
No potential conflict of interest relevant to this article was reported.