|
 |
- Search
| Clin Exp Reprod Med > Volume 39(2); 2012 > Article |
Abstract
Objective
Sterilization (tubal sterilization and vasectomy) is a widely applied contraceptive method worldwide. Although most studies have described sterilization as a safe method, there are reports of tubal ligation (TL) and vasectomy complications. The aim of this study was to evaluate the effects of TL and vasectomy on the serum oxidative stress, specifically prooxidant-antioxidant balance (PAB) and malondialdehyde (MDA) levels, over time.
Methods
Male and female rats were classified into vasectomy, sham-vasectomy, TL, and sham-TL groups, respectively. The PAB and MDA levels were measured on days 15 and 45 and months 3 and 6 after the intervention. For female rats, blood sampling was performed during the diestrous phase and estradiol and progesterone were also measured.
Results
Serum PAB and MDA increased after TL (p<0.05). Vasectomy increased serum MDA remarkably after 45 days, 3 months, and 6 months (p<0.05). After vasectomy, serum PAB also increased although not significantly. Serum estradiol and progesterone decreased remarkably in the TL group compared to the sham group (p<0.05).
Conclusion
Bilateral TL and vasectomy both increase the serum oxidative stress; however the imbalance after TL was very noticeable. As for the TL, the reduction of serum estrogen levels can be involved in this imbalance. Complications followed by TL or vasectomy could be due to increased levels of oxidants. Thus, prescribing antioxidants during and or after surgery may be a solution.
Sterilization (tubal sterilization and vasectomy) is a widely applied contraceptive method all around the world [1]. Although most studies have found sterilization to be a safe method, there are reports of tubal ligation (TL) and vasectomy complications.
According to the literature, TL may cause complications such as menstrual irregularities [2], dysmenorrhea [3], pelvic pain [4], increased risk of osteoporosis [5], early menopause [6], and sexual behavior changes [7]. TL might result in severe hypoxia in the ovaries and uterine tubes and adversely affect ovarian function [8].
Complications following vasectomy include inflammatory reaction [9], formation of granuloma sperm [10], prostate cancer [11], and thickening and dilation of the epididymis [12]. Moreover, several post-vasectomy changes occur in the testis tissue such as tubular dilatation, thickening seminiferous tubules, reduction in germ cells and spermatids [13], incomplete spermatogenesis, phagocyte aggregation [14], and increased occurrence of interstitial fibrosis [13]. It has been suggested that the generation of granuloma sperm [14] and interstitial fibrosis of the testicles [15] are caused by oxidative stress. Studies have shown that all complications caused by TL and vasectomy may change over time [16,17].
Oxidative stress is defined to be an imbalance between prooxidants and antioxidants such that the prooxidant levels are higher [18]. Highly reactive and unsteady reactive oxygen species (ROS) become stable as they obtain electrons from nucleic acids, lipids, proteins, carbohydrates, or any other molecule alongside which brings about a cascade of chain reactions causing cellular harm and diseases [19]. However, the consequences of TL and vasectomy on patterns of oxidative stress are poorly documented. Oxygen-dependent production of ROS and antioxidant activity may be attributable to the pathogenic mechanisms of the complications of sterilization in the reproductive system.
The aim of this study was to evaluate the effects of TL and vasectomy on the serum oxidative stress (prooxidant-antioxidant balance [PAB] and malondialdehyde [MDA]) over time.
The ethical committee of Tehran University of Medical Sciences approved all the procedures involved in the study and care and use of laboratory animals. The rats were caged under controlled lighting (12-hour light/dark cycle) and temperature (24Ōäā) and allowed free access to a standard rat laboratory diet. The surgery was carried out under intraperitoneal anesthesia using ketamine hydrochloride (40 mg/kg) and xylazine hydrochloride (5 mg/kg).
Fifty-six 3-month-old female Sprague-Dawley rats were included in the study. The rats were randomly selected and were divided into 2 groups: sham and TL. Vaginal smears were obtained daily and only rats showing at least three consecutive four-day estrous cycles were used. After shaving and cleaning the abdomen with 10% povidoneiodine solution, laparotomy was performed via a 3 cm midline vertical incision. Then bilateral TL was applied by Pomeroy's technique using 2/0 catgut [8]. The surgical interventions to the fallopian tubes were made 1 cm away from the uterine horns. In the sham group, the uterine horns were exposed and left in the abdominal cavity without ligation. The abdominal wall was then closed with 3/0 polypropylene.
Fifty-six 3-month-old male Sprague-Dawley rats were randomly divided into two groups (Sham and Vasectomy). Under sterile conditions, a 1 cm incision was made on the scrotum, skin, fascia, cremaster muscle, and peritoneal layer. All were cut on one side and then the other to uncover the vas deferens and its blood vessels 2 cm distal to the tail of the epididymis. The ductus deferens was twice ligated and subsequently transected between the two ligatures and this part was taken. Then the cremaster muscle and peritoneal layer were sutured by 4.0 catgut and the skin and fascia were stitched with silk. In the Sham group: the procedure was followed exactly the same as for the vasectomy group, excluding the ligation and truncation of the vas deferens.
Blood samples of all of the animals were obtained via cardiac puncture at 10 to 12 AM at 15 days, 45 days, 3 months, and 6 months after the intervention. They were immediately centrifuged at 2,000├Ś g for 15 minutes at 4Ōäā, and the serum aliquots were separated and stored at -80Ōäā. Serum PAB and MDA were measured. In the female rats, the forgoing process was performed during the diestrous phase. 17 beta-estradiol and progesterone were additionally measured.
Tetramethylbenzidine 3, 3', 5.5' (TMB) and TMB cations were used as oxidation-reduction indicators, due to their optical and electrochemical properties. With this approach, it is possible to measure the oxidant-antioxidant balance simultaneously in one experiment by two different reaction types: one enzymatic reaction where the chromogen TMB is oxidized to a color cation by peroxides and one chemical reaction where the TMB cation is reduced to a colorless compound by antioxidants. Then the photometric absorbency was compared with the given absorbencies of a series of standard solutions that are mixtures of uric acid and various proportions (0% to 100%) of hydrogen peroxide. PAB was arbitrarily expressed in HK units [20].
Serum MDA was measured using the spectrophotometric method introduced by Satoh [21] in 1978. In this method, a thiobarbituric acid solution of sodium sulfate is added to the serum. After heating, the produced chromogen is extracted by n-butyl alcohol solution, and the phase absorbency is recorded at a 530 nm wavelength. The MDA concentration is then calculated in nm/mL via a standard solution [21].
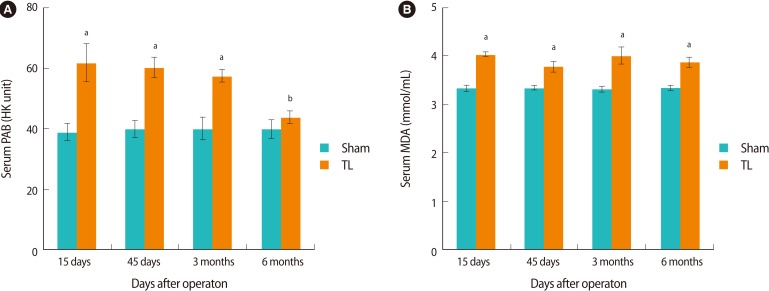
The serum concentration of PAB increased in the TL groups after 15 days, 45 days, and 3 months compared to the sham groups (p<0.05); however, after 6 months, serum PAB decreased compared to the 15 and 45 day groups (p<0.05) (Figure 1A). Moreover, the serum concentration of MDA increased remarkably after 15 days, 45 days, 3 months, and 6 months in the TL groups compared to the sham groups (p<0.05) (Figure 1B).
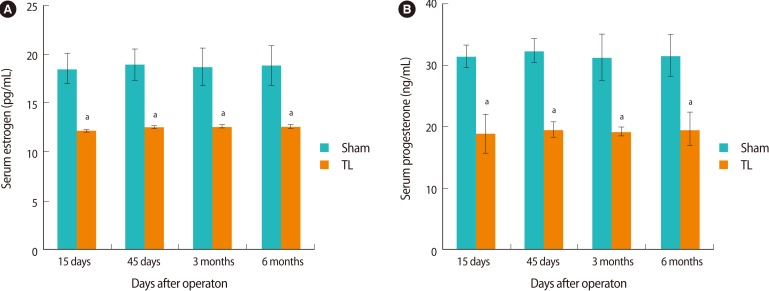
There was a remarkable decrease in the serum concentration of estradiol and progesterone after 15 days, 45 days, 3 months, and 6 months in the TL groups compared to the sham groups (p<0.05) (Figure 2).
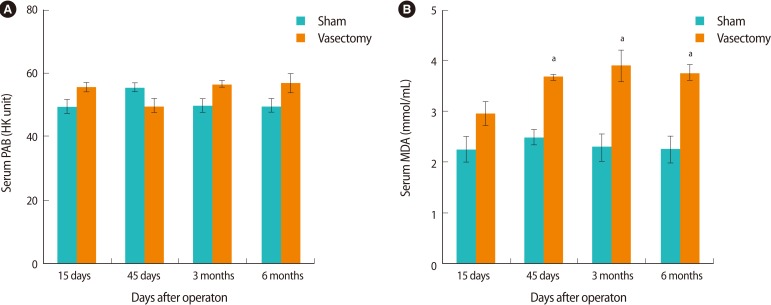
Data from the serum concentration of PAB are shown in Figure 3A. There was no significant difference between the vasectomy and sham groups in serum PAB. However, the serum concentration of MDA significantly increased in the vasectomized group 45 days, 3 months, and 6 months after the operation as compared to the sham groups (p<0.05) (Figure 3B).
The present results demonstrated a significant increase in serum PAB within a short period (15 and 45 days and 3 months) after TL in adult rats; however, the increment of 6 months following the operation was not significant. Thus, the serum concentration of PAB decreased over time. Serum MDA also increased significantly at 15 days, 45 days, 3 months, and 6 months after TL. Our results indicated that vasectomy also increased MDA, which was significant over time.
TL can result in increased hypoxia in the ovaries and tubes and eventually increases ROS generation. Kilic et al. [8] detected that after 15 days, TL in rats led to severe hypoxia in the ovaries and fallopian tubes, which was demonstrated by an increase of hypoxia markers such as vascular endothelial growth factor (VEGF) and inducible nitric oxide (iNOS). In part, hypoxia slows down the electron transmission in the mitochondria; consequently, a large quantity of ROS is generated [22]. Hypoxia-inducible factors (HIF-╬▒) bind to hypoxia-sensitive regions of the VEGF gene, and this will play an important role in regulating VEGF transcription. In case of tissue oxygen reduction, angiogenesis responses occur. iNOS mediates HIF-╬▒ activities and expression of VEGF [8].
MDA is a primary product of polyunsaturated fatty acid peroxidation, which can demonstrate an increase in oxidative stress [23]. In this study, after vasectomy, MDA production was observed to be increasing over time; most likely, this is due to increased formation of abnormal sperm [24], formation of granuloma sperm [10,25], and increased immune response [15]. Aydos et al. [26] indicated an increase in MDA in the testes post-vasectomy. Moreover, it has been reported that after vasectomy in rats, ascorbyl and dienyl radicals and also superoxide production increase significantly (up to four times) and the production of nitric oxide radicals is reduced 50% in the testes [27]. Bilateral vasectomy due to obstruction of the vas deferens adversely affects the maturation and motility of sperm [24,28]. ROS generation increases as the concentration and motility of the sperm decrease [29] and the amount of abnormal sperm increases [30]. Following vasectomy, the number of apoptosis germinal cells in the testes significantly increases [31] and hydrostatic pressure and generated inflammatory reactions increase the number of poly-morphonuclear leukocytes in the seminiferous tubules [32]. Poly-morphonuclear leukocytes play a crucial role in terms of increased generation of ROS [24]. Abrupt obstruction of the vas deferens increases testicular pressure and thus decreases testis blood flow, which in turn increases oxidative stress in the testes.
In the present study, there was a significant increase in serum MDA after vasectomy but the increase in serum PAB was not significant in comparison to the sham groups. This could be due to antioxidant increases in other parts; the oxidant-antioxidant balance in the serum is not affected significantly. Additional studies in this field are required.
In our study, serum estradiol and progesterone significantly decreased after TL. Rico et al. [33] reported that TL decreases the bone mass in the axial bones (spine and femur), which is similar to the bone mass loss in ovariectomy cases. Therefore, it can be concluded that TL may reduce estrogen. In a study of humans, the serum estrogen level declined, but not significantly, six months after TL although the luteal phase progesterone levels significantly declined. The reduced progesterone level during the luteal phase is due to the defective formation of the corpus luteum, which in turn is probably due to vascular changes after TL [34]. TL is assumed to damage the fallopian tubes and some parts of the mesosalpinx which explains the alteration in ovarian blood flow [35].
We did not measure the serum testosterone level after vasectomy. In other studies, it has been shown that bilateral vasectomy does not strongly affect the endocrine function of the testis over time [36] although it may result in damage to spermatogenesis in vasectomized rats [37].
Bilateral TL and vasectomy both increase serum oxidative stress; however, the imbalance after TL was very noticeable. TL probably reduced serum estrogen levels, as a potent antioxidant and could be induced in this imbalance. Therefore, it can be stated that in some ways vasectomy is less likely than tubal sterilization to result in serious complications. Complications following TL or vasectomy could be due to increased levels of oxidants. Therefore, prescribing antioxidants during and or after surgery may be a solution.
References
2. Aygen EM, Ozdamar S, Serin S, Babug M. Ovarian morphology of rats after fallopian tube sterilization. Contraception 2002;66:211-214.PMID: 12384212.


3. Ozerkan K, Aydin G, Koc I, Uncu Y, Uncu G. Menstrual pattern following tubal sterilization. Med Sci Monit 2010;16:CR197-CR201.PMID: 20357719.

4. Mall A, Shirk G, Van Voorhis BJ. Previous tubal ligation is a risk factor for hysterectomy after rollerball endometrial ablation. Obstet Gynecol 2002;100:659-664.PMID: 12383530.


5. Wyshak G. Tubal ligation and the risk of vertebral fractures. Osteoporos Int 2005;16:651-658.PMID: 15455196.


6. Kutlar I, Ozkur A, Balat O, Ugur MG, Genco Y, Aksoy F. Effects of three different sterilization methods on utero-ovarian Doppler blood flow and serum levels of ovarian hormones. Eur J Obstet Gynecol Reprod Biol 2005;122:112-117.PMID: 16154047.


7. Gentile GP, Kaufman SC, Helbig DW. Is there any evidence for a post-tubal sterilization syndrome? Fertil Steril 1998;69:179-186.PMID: 9496325.


8. Kilic S, Tasdemir N, Lortlar N, Yuksel B, Budak G, Batioglu S. Vascular endothelial growth factor (VEGF) and inducible nitric oxide synthase (iNOS) immunoreactivities in rat ovaries and uterine tubes after tubal ligation: a controlled immunohistochemical study. Eur J Contracept Reprod Health Care 2008;13:431-437.PMID: 19117260.


9. Lavers AE, Swanlund DJ, Hunter BA, Tran ML, Pryor JL, Roberts KP. Acute effect of vasectomy on the function of the rat epididymal epithelium and vas deferens. J Androl 2006;27:826-836.PMID: 16837732.


10. Melville C, Bigrigg A. Male and female sterilization. Obstet Gynecol Reprod Med 2008;18:330-334.

11. Rohrmann S, Paltoo DN, Platz EA, Hoffman SC, Comstock GW, Helzlsouer KJ. Association of vasectomy and prostate cancer among men in a Maryland cohort. Cancer Causes Control 2005;16:1189-1194.PMID: 16215869.


12. Reddy NM, Gerscovich EO, Jain KA, Le-Petross HT, Brock JM. Vasectomy-related changes on sonographic examination of the scrotum. J Clin Ultrasound 2004;32:394-398.PMID: 15372447.


13. Shiraishi K, Takihara H, Naito K. Influence of interstitial fibrosis on spermatogenesis after vasectomy and vasovasostomy. Contraception 2002;65:245-249.PMID: 11929647.


14. Chatterjee S, Laloraya M, Kumar GP. Free radical bombing of spermatozoa in spermatic granuloma: an attempt to prevent autoimmune switch-on. Biochem Biophys Res Commun 1994;201:472-477.PMID: 8198611.


15. Nourouzi MR, Keyhani AH, Ayati M. Identification of acrosome as the main antigen of the sperm cells provoking autoantibodies in vasectomized iranian men. Acta Medica Iranica 2008;46:457-460.
16. Gentile GP, Helbig DW, Zacur H, Park T, Lee YJ, Westhoff CL. Hormone levels before and after tubal sterilization. Contraception 2006;73:507-511.PMID: 16627035.


17. Bedford JM. Adaptations of the male reproductive tract and the fate of spermatozoa following vasectomy in the rabbit, rhesus monkey, hamster and rat. Biol Reprod 1976;14:118-142.PMID: 1260083.


18. Halliwell B, Gutteridge JM. Free radicals in biology and medicine. 1985. Oxford: Clarendon Press.
19. Agarwal A, Gupta S, Sharma RK. Role of oxidative stress in female reproduction. Reprod Biol Endocrinol 2005;3:28PMID: 16018814.



20. Alamdari DH, Paletas K, Pegiou T, Sarigianni M, Befani C, Koliakos G. A novel assay for the evaluation of the prooxidant-antioxidant balance, before and after antioxidant vitamin administration in type II diabetes patients. Clin Biochem 2007;40:248-254.PMID: 17196578.


21. Satoh K. Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta 1978;90:37-43.PMID: 719890.


22. Chandel NS, McClintock DS, Feliciano CE, Wood TM, Melendez JA, Rodriguez AM, et al. Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: a mechanism of O2 sensing. J Biol Chem 2000;275:25130-25138.PMID: 10833514.


23. Del Rio D, Stewart AJ, Pellegrini N. A review of recent studies on malondialdehyde as toxic molecule and biological marker of oxidative stress. Nutr Metab Cardiovasc Dis 2005;15:316-328.PMID: 16054557.


24. Ren L, Weng Q, Kishimoto M, Watanabe G, Jaroenporn S, Taya K. Effect of short period vasectomy on FSH, LH, inhibin and testosterone secretions, and sperm motility in adult male rats. Exp Anim 2011;60:47-56.PMID: 21325751.


25. McGinn JS, Sim I, Bennett NK, McDonald SW. Observations on multiple sperm granulomas in the rat epididymis following vasectomy. Clin Anat 2000;13:185-194.PMID: 10797625.


26. Aydos K, Kupeli B, Soygur T, Unsal A, Erden E, Tulunay O, et al. Analysis of the relationship between histologic alterations and the generation of reactive oxygen species in vasectomized rat testes. Urology 1998;51:510-515.PMID: 9510366.


27. Chatterjee S, Rahman MM, Laloraya M, Kumar GP. Sperm disposal system in spermatic granuloma: a link with superoxide radicals. Int J Androl 2001;24:278-283.PMID: 11554985.


28. Pasqualotto FF, Sharma RK, Nelson DR, Thomas AJ, Agarwal A. Relationship between oxidative stress, semen characteristics, and clinical diagnosis in men undergoing infertility investigation. Fertil Steril 2000;73:459-464.PMID: 10688996.


29. Aziz N, Saleh RA, Sharma RK, Lewis-Jones I, Esfandiari N, Thomas AJ Jr, et al. Novel association between sperm reactive oxygen species production, sperm morphological defects, and the sperm deformity index. Fertil Steril 2004;81:349-354.PMID: 14967372.


30. Aitken RJ, Krausz C. Oxidative stress, DNA damage and the Y chromosome. Reproduction 2001;122:497-506.PMID: 11570956.


31. Kubota Y, Sasaki S, Kubota H, Tatsura H, Kohri K. A study on the mechanism of the spermatogenic damage after vasectomy in rats. Nihon Hinyokika Gakkai Zasshi 2001;92:13-22.PMID: 11235138.


32. Singh SK, Chakravarty S. Histologic changes in the mouse testis after bilateral vasectomy. Asian J Androl 2000;2:115-120.PMID: 11232787.

33. Rico H, Cuesta C, Cortes J, Seco C, Monteagudo MD, Hernandez ER. Effect of uterine horn ligation on bone mass: an experimental study in rats. Eur J Obstet Gynecol Reprod Biol 2002;102:48-52.PMID: 12039089.


34. Fagundes ML, Mendes MC, Patta MC, Rodrigues R, Berezowski AT, de Moura MD, et al. Hormonal assessment of women submitted to tubal ligation. Contraception 2005;71:309-314.PMID: 15792652.


35. Cattanach JF, Milne BJ. Post-tubal sterilization problems correlated with ovarian steroidogenesis. Contraception 1988;38:541-550.PMID: 3197418.


36. Batista M, Prats N, Calero P, Gonzalez F, Cabrera F, Medrano A, et al. Semen characteristics and plasma levels of testosterone after bilateral vasectomy in bucks. Reprod Domest Anim 2002;37:375-378.PMID: 12464078.


37. Tanrikut C, Goldstein M, Rosoff JS, Lee RK, Nelson CJ, Mulhall JP. Varicocele as a risk factor for androgen deficiency and effect of repair. BJU Int 2011;108:1480-1484.PMID: 21435152.


Figure┬Ā1
Changes in serum concentrations of (A) prooxidant-antioxidant balance (PAB) and (B) malondialdehyde (MDA) in female rats after tubal ligation (TL) or sham surgery. Each value represents the mean┬▒SE of 7 animals. ap<0.05 compared to sham values. bp<0.05 compared to the TL groups at 15 and 45 days.