Introduction
The mammalian ovary contains follicles at various developmental stages and its function includes the cyclic recruitment, development and regression of the follicles. The pool of primordial follicles is maintained
in dormancy and serves as a reserve for cyclic recruitment of follicles and oocytes.
The primordial follicles are maintained in dormancy or activated to grow into antral follicles. Some of the antral follicles reach ovulation under proper gonadotropin stimulation, however, most of the follicles undergo atresia during follicular development or directly from the dormant state [1]. The maintenance of the quiescence and survival of primordial follicles is important as a reserve to provide oocytes throughout the reproductive life. The mechanisms of the maintenance of such a long dormancy of the primordial follicles have remained unknown, however the gonadotropin-dependent process of follicular development is otherwise well elucidated [2]. In the control of primordial follicle activation, bidirectional communication between the oocytes and surrounding somatic cells is involved. The oocytes are supported by granulosa cells and oocytes also play an active role in the growth of the granulosa cells [3].
Recently, it was reported that phosphatidylinositol 3 kinase (PI3K) pathway is important for the control of follicular activation. In mouse ovaries deficient in forkhead box O3 (Foxo3a), the entire pool of primordial follicles is prematurely activated [4]. Moreover, with the findings from a transgenic mouse model, it has been shown that the PI3K/phosphatase and tensin homolog (PTEN) signaling pathway involves the regulation of dormancy and activation of primordial follicles [5,6].
Maintaining the dormancy of primordial follicles
The pool of primordial follicles is under inhibitory control, therefore, when these inhibitory factors decrease or stimulatory factors increase, the follicles are activated [7]. The activated primordial follicles develop into primary, secondary, and antral follicles, however, most of the antral follicles undergo atresia. Only a few of them grow to the preovulatory stage under the stimulation of gonadotropin [1]. Activation of the primordial follicles of bovine cortical tissue was noted in an in vitro culture in serum-free medium. This implies that the primordial follicles are under inhibitory control to maintain the dormant state [8]. Primordial follicles activate spontaneously in vitro and incubation with exogenous cytokine and growth factors increases the number of activated follicles.
Regulation of activation by multiple activator and repressor pathways
Multiple activator and repressor or signaling pathways are involved in the control of primordial follicle activation. Neurotropin, nerve growth factor (NGF) and its tyrosine receptor kinase NTRK1 are important in follicular activation [9]. Other neurotrophins, such as neurotrophin 4 (NT4), brain-derived neurotrophic factor (BDNF) and their receptor NTRK2 are also involved in regulating oocyte survival [10].
Some ligands play multiple roles in folliculogenesis. transforming growth factor-beta (TGF-β) ligand, growth differentiation factor 9 (GDF9), and bone morphogenic protein 15 (BMP15) play roles in pre-antral follicular development in addition to their facilitation of primordial follicle activation.
1. GDF 9
Treatment of immature rats with recombinant GDF9 leads to an increased rate of follicular activation. The addition of recombinant GDF9 to human ovarian cortical tissue in vitro increases the activation and improves the survival of follicles [11]. In GDF9 null mice, the follicles do not progress beyond the first stage and granulosa cell proliferation is limited [12]. However, oocyte growth is accelerated. These results suggest that GDF9 plays a role in recruiting granulosa cell while limiting the growth of the oocytes.
2. SMADS
SMADS are downstream intracellular signaling molecules of the TGF-β ligands (BMP 4 and 7). SMADS null mice have impaired folliculogenesis showing increased numbers of primordial follicles and decreased numbers of developed follicles in adult life [13].
3. FOXL2
Forkhead boxL2 (Foxl2) is expressed in mammalian eyelids and ovaries. In mice, Foxl2 is expressed in pre-granulosa cells and the expression decreases in the granulosa cells of pre-antral follicles. In humans, mutation of the FOXL2 gene causes blepharophimosis-ptosis-epicanthus-inversus (BPES), an autosomal dominant genetic disease showing eyelid dysplasia and high risk of POF [14]. In Foxl2 inactivated mice, though the primordial follicles form normally, they do not develop into secondary follicles because pre-granulosa cells do not complete the cuboidal transition. Most of the oocytes in primordial follicles are activated prematurely, and undergo atresia, which implies the depletion of the primordial follicle pool [15]. FOXL2 in pregranulosa cells plays an important role in the quiescence of primordial follicles by inhibiting the initiation of the growth of oocytes.
4. Sohlh1
Spermatogenesis- and oogenesis-specific basic helix-loop-helix-containing protein 1 (Sohlh1) is expressed in the germ cell clusters and in new primordial follicles. Sohlh1 null mice show a rapid decrease in the number of oocytes by 7 weeks of age and a defect in the primordial to primary transition [16].
5. AMH
AMH and its receptor AMHRII are expressed in granulosa cells. The role of AMH in primordial follicle was investigated an Amh-/- mice. Amh deficient mice showed premature depletion of primordial follicles, indicating that AMH inhibits the growth of primordial follicles [17]. In mouse ovaries cultured with AMH, growing follicle numbers decreased by 40-50% [18], and human ovarian tissue cultured with recombinant rat AMH showed decreased growth of primordial follicles [19]. Therefore, AMH plays a role in maintaining quiescence by inhibiting the transition from primordial to primary follicles.
Intracellular signaling in oocytes for maintenance of dormancy of primordial follicles
1. PTEN
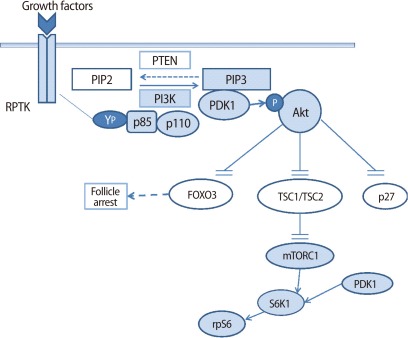
Phosphatase and tensin homolog deleted on chromosome 10 is a negative regulator of PI3K. In the absence of PTEN, 3,4,5-triphosphate (PIP3) increase leads to protein kinase B (Akt) signaling. The role of PTEN in primordial follicles was elucidated by a study using oocyte-specific Pten deleted mice (OoPten-/- mice) [6].
Oocyte-specific Pten was deleted from oocytes of primordial follicles in mice by crossing PtenloxP/loxPmice with transgenic mice expressing Gdf-9 promoter-mediated Cre recombinase (Gcre mice) [20]. In the OoPten-/- mice, the primordial follicles are overactivated and grow to primary and secondary follicles, which results in the early depletion of the primordial follicle pool. Another study with pten-deleted primary oocytes in oocyte-specific Gcre transgenic mice showed similar results of follicular overactivation and early depletion [21]. These results suggest that PTEN is essential for the maintenance of dormancy of primordial follicles [22] (Figure 1).
2. TSC (tuberin/tuberous sclerosis complex)
Mammalian target of rapamycin (mTOR) complex 1 (mTORC1) is negatively controlled by a heterodimeric complex consisting of two molecules: Tsc1 (hamartin) and Tsc2 (tuberin).
TSC1 and TSC2 are products of TSC1 and TSC2 genes, respectively. The TSC1 and TSC2 gene mutations occur in autosomal dominant tumor syndrome, tuberous sclerosis [23]. Tsc 1 is considered to be important for maintenance of the dormancy of primordial follicles. In mice with Tsc1 deleted in oocytes (OoTsc1-/- mice), primordial follicles were prematurely activated, which also occurs in OoTsc2-/- mice [24]. These results imply that Tsc1/Tsc2 complex plays a role in regulating the quiescence and activation of follicles. In Tsc1 or Tsc2 deletion, mTORC1 increases and activates S6K1-rpS6 signaling (p70 S6 kinase-ribosomal protein), which promotes protein translation and ribosome synthesis (Figure 1) [21]. The phenotypes of overactivation of primordial follicles in OoTsc1-/- mice and OoPten-/- mice are similar. Although both Tsc and PTEN regulate follicular activation by inhibiting S6K1-rpS6, Tsc inhibits phosphorylation of S6K1 at threonine 389, and PTEN inhibits the phosphorylation at threonine 229 [21]. Therefore, for the maintenance of the dormancy of primordial follicles, S6K1 should be suppressed by Tsc and PTEN, respectively.
3. Foxo3a
Foxo3a is expressed in the nucleus of oocytes of primordial follicles, and its expression decreases in primary or growing follicles. The decrease of Foxo3a initiates the activation of follicles and growth of oocytes. In Foxo3a-deleted mice (Foxo3a-/-), follicles are depleted, which results in infertility, indicating that Foxo3a is important for maintaining the quiescence of primordial follicles [4]. Foxo3a-/- mice show a phenotype similar to that of Pten-/- mice. In the Pten deleted mice, constant Akt activation causes the suppression of Foxo3a.
Therefore, both PTEN and Foxo3a play important roles in maintaining the dormancy of primordial follicles and PTEN acts upstream of Foxo3a [6]. Akt inactivates Foxo3a through an extracellular signaling pathway such as insulin signaling, so the effect of insulin for follicular activation is by the inhibition of FOXO3 [25,26].
The signals that maintain the survival and activation of primordial follicles
1. PI3K signaling
PI3K activation is maintained by extracellular signals, and it maintains the survival of primordial follicles [27]. The survival of primordial follicles is maintained through the PI3K pathway [28]. The communications between oocytes and the surrounding granulosa cells depend on their interactive signaling, and the main ligand-receptor is the receptor protein tyrosine kinase (RPTK) Kit and its ligand, Kit ligand (KL) [29]. Kit exists on the surface of oocytes, and KL is produced in the granulosa cells. In early stage oocytes and follicles in which FSH receptors are not expressed yet, follicular growth is dependent on Kit-KL signaling [30-32].
PI3K signaling is one of the important pathways activated by Kit-KL [33]. KL activates the PI3K pathway in primary oocytes, followed by phosphorylation of Akt (protein kinase B) and Foxo3a. In mammals, the PI3K pathway is activated by insulin and growth factors and it regulates cell proliferation and survival [33]. PI3K phosphorylates phosphatidylinositol-4,5-bisphosphate (PIP2) to phosphatidylinositol-3,4,5-triphosphate (PIP3), and PIP3 acts as a secondary messenger and provides the docking sites for 3-phosphoinositide dependent kinase-1 (PDK1) and Akt. Akt enhances cell survival, growth, and oocyte activation. Otherwise, PTEN inhibits PI3K by converting PIP3 to PIP2. The FOXO family is a main target of Akt and it suppresses the activation of primordial follicles and maintains dormancy by promoting pro-apoptotic genes (Figure 1) [27]. PI3K signaling is mediated by PDK1, and PDK1 activates Akt signaling. PDK1 phosphorylates Akt at its threonine residue308, and also phosphorylates S6K1, the kinase of rpS6 [34]. In mice with an oocyte-specific Pdk1 deletion, most of the primordial follicles are depleted directly from the dormant state and cause premature ovarian failure. Therefore, it is suggested that the suppression of PDK1-Akt-S6K1-rpS6 signaling leads to the depletion of primordial follicles [35]. In double knock-out mice with deletion of both Pten and Pdk1, the premature activation does not occur, suggesting that the premature activation of follicles in Pten deletion is mediated by PDK1 [36]. PI3K-PDK1 signaling plays an important role in the survival and activation of primordial follicles.
2. mTOR
Mammalian target of rapamycin (mTOR) is a serine/threonine kinase, and regulates protein synthesis and cell growth. mTOR exists in the two forms of mTORC1 and mTORC2. S6K1 is the main substrate of mTORC1 and is phosphorylated by mTORC1 [37]. In Tsc1-deleted (OoTsc1-/-) mice treated with mTORC1 inhibitor, the overactivation of primordial follicles is suppressed, which implies that mTORC1 is involved in the activation of primordial follicles. These results suggest that the mTORC1-S6K1-rpS6 cascade is also important in the survival of primordial follicles [21]. mTOR deletion is expected to accelerate the loss of primordial follicles [21].
In vitro activation of ovarian follicles
In a mouse model, Pten deletion in granulosa cells reduced apoptosis and increased cellular proliferation but not tumors [38]. These results suggest that PTEN inhibitor can activate the primordial follicles. Recently, Li et al. [39] reported that short-term treatment of mouse ovaries with PTEN inhibitor and PI3K-activating peptide increased the activation of primordial follicles with increased nuclear extrusion of Foxo3. The size of the ovarian tissue and the number of follicles increased in PTEN inhibitor-treated ovaries. In addition, in the human ovarian cortical tissues cultured with PTEN inhibitor and PI3K activator, primordial follicles developed to the preovulatory stage, after xeno-transplantation. Thus follicular activation with PTEN inhibitor can be applied to generate more germ cells from cyro-preserved ovarian tissue for fertility preservation in cancer patients.
Conclusion
Primordial follicle activation is regulated by close interactive communication with somatic cells and oocytes. The dormancy of primordial follicles is maintained by multiple factors such as PTEN, Tsc, Foxo3a, Foxl2, and AMH. With the deletion of these factors, follicles are activated prematurely leading to early depletion of the primordial follicles. To maintain the survival and activation, PI3K signaling mediated by PDK1 and mTORC1-S6K1-rpS6 are involved. With this knowledge on the regulatory mechanism for dormancy, survival, and activation of primordial follicles, possible clinical applications could include in vitro maturation of immature oocytes or in vitro activation of follicles by PTEN inhibitor in cryopreserved ovarian cortical tissues for fertility preservation in cancer patients or diminished ovarian reserve.