|
 |
- Search
| Clin Exp Reprod Med > Volume 42(3); 2015 > Article |
Abstract
Essure (Bayer) received approval from the U.S. Food and Drugs Administration as a permanent non-hormonal contraceptive implant in November 2002. While the use of Essure in the management of hydrosalpinx prior to in vitro fertilization (IVF) remains off-label, it has been used specifically for this purpose since at least 2007. Although most published reports on Essure placement before IVF have been reassuring, clinical experience remains limited, and no randomized studies have demonstrated the safety or efficacy of Essure in this context. In fact, no published guidelines deal with patient selection or counseling regarding the Essure procedure specifically in the context of IVF. Although Essure is an irreversible birth control option, some patients request the surgical removal of the implants for various reasons. While these patients could eventually undergo hysterectomy, at present no standardized technique exists for simple Essure removal with conservation of the uterus. This article emphasizes new aspects of the Essure procedure, as we describe the first known association between the placement of Essure implants and the subsequent development of fluid within the uterine cavity, which resolved after the surgical removal of both devices.
The standard recommendation for the surgical correction of hydrosalpinx before in vitro fertilization (IVF) assumes that the patient is a suitable candidate for laparoscopy. This may not always be the case. For example, extensive pelvic adhesions and an increased risk for bowel injury may make abdominal access unsafe or difficult for some patients. In contrast, the hysteroscopic management of hydrosalpinges has important advantages over laparoscopy, such as obviating the need for abdominal access, reducing the overall cost, and minimizing anesthesia requirements. These features have led the office hysteroscopic Essure (Bayer, Whippany, NJ, USA) procedure to be considered an attractive non-incisional alternative to traditional laparoscopic tubal surgery for women with hydrosalpinx before IVF. Most published descriptions of the use of Essure to treat hydrosalpinx before embryo transfer have been favorable. Indeed, the reported complications following Essure placement have mainly occurred in the context of its Food and Drugs Administration-approved birth control application [1,2], not in a pre-IVF setting.
Data are unavailable regarding how often Essure is placed before IVF, or what proportion of Essure patients ultimately seek IVF after undergoing a tubal occlusion procedure. This is the first known report to describe a case of abnormal endometrial fluid collection after Essure placement one year before IVF was planned. Once the devices were surgically excised, the intrauterine fluid disappeared. We present some novel counseling considerations regarding Essure when used for this purpose and also outline a successful surgical technique for the intact removal of the Essure device.
A 42-year-old unmarried nulligravida presented for a reproductive endocrinology consultation; the patient was in good general health, had regular menses, and had never smoked. Her body mass index was 22 kg/m2. At the time of her initial appointment, she had undergone Essure placement 1 year previously and stated her wish to undergo IVF with anonymous donor gametes. Many years earlier, she experienced 'chronic belly pain' for which multiple specialists were seen, and eventually she underwent a total colectomy at age 28. Pelvic adhesions were later suspected, based on a hysterosalpingogram (HSG) that showed distal occlusion in both fallopian tubes. Twelve years after partial bowel resection, the patient underwent bilateral Essure placement for the management of hydrosalpinges. She remained asymptomatic after the Essure procedure, but a confirmatory HSG was not performed.
At our center, an intrauterine fluid collection measuring approximately 5.7 mm (in the anterior-posterior dimension) was identified and confirmed on three separate transvaginal sonograms obtained at various phases of the patient's menstrual cycle. No evidence of tubal pathology was found on the ultrasound images. A representative sample of this fluid was aspirated on the final sonogram visit and was submitted for analysis; no organisms or abnormal cytological findings were identified. The potential negative impact of the uterine fluid on the outcome of IVF was carefully discussed with the patient.
All of the patient's screening tests, cervical cytology, and pre-IVF laboratory testing were normal. However, her serum anti-M├╝llerian hormone level was <0.16 ng/mL, suggesting poor ovarian reserve. The patient reviewed anonymous oocyte donor profiles, contacted an anonymous sperm bank, and written informed consent was obtained for IVF using dual anonymous donor gametes. The patient completed psychological counseling, and a favorable report was returned. However, her treatment could not begin until the problem of endometrial compartment fluid was resolved. While laparoscopic and hysteroscopic routes for Essure removal were considered, the patient was advised that hysteroscopic removal would probably fail in her case since far more than 12 weeks had elapsed since the implants were placed [3]. The patient was carefully counseled about the risks associated with laparoscopy.
Informed consent was obtained for hysteroscopy, laparoscopy, laparotomy (in case of emergency), the removal of foreign objects, and partial bilateral salpingectomy. A bowel preparation was ordered prior to surgery, and a colorectal surgeon and gynecologic oncologist were on stand-by to assist with the abdominal portion of the operation if needed. Under general anesthesia, the uterine cavity was evaluated via a 5-mm video hysteroscope. The endometrial cavity was normal and the uterotubal junctions were easily visualized. Full device engraftment was noted bilaterally and no exposed coils were seen. Attention was next turned to the abdomen, which was safely entered on the first attempt and explored laparoscopically. Two 5-mm accessory ports were placed under direct video laparoscopic guidance. Moderate adhesions were noted at the proximal left fallopian tube, although the uterine exterior and both ovaries appeared unremarkable.
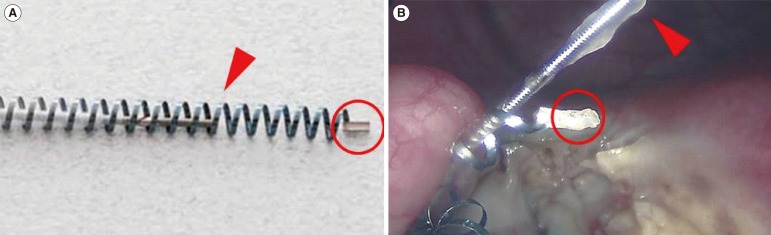
Bipolar cautery set to 40 W of power was used sparingly to desiccate the outer tissue layers of the right fallopian tube near the right uterine junction. The tissue was carefully divided in a circumferential fashion down to the level of the implant. The Essure implant and the associated tubal tissue were isolated carefully from the cornual placement site, and any exterior uterine bleeding was easily managed with electrocautery. In order to avoid traction on the device, resistance to lateral movement was met with more extensive uterine dissection to facilitate lifting the implant away from the cornu, rather than pulling the device out of it. Once the tube and its attached Essure implant were freed from the uterotubal junction, the outer coil's terminal marker (Figure 1) was closely inspected to ensure that the device had been removed intact. With the medial portion of the Fallopian tube now unattached, the lateral extent of the implant was observed through serial palpation with an atraumatic grasper. Cautery was applied to the fallopian tube approximately 1 cm lateral to the distal margin of the Essure implant. This freed the device and its associated tubal tissue for en bloc removal; no linear salpingotomy was performed. Next, the contralateral tube was examined and treated with the bipolar cautery tool. Tissue desiccation was again achieved with 40 W of power in a similar fashion to remove an equivalent tubal segment contralaterally. Both adnexal sites were carefully examined and excellent hemostasis was noted. No vasoconstrictive solution was injected at either operative site. The patient tolerated the procedure well and no complications occurred; she was discharged home in less than 4 hours. Her immediate postoperative course was uneventful. Three weeks after surgery, pelvic ultrasound confirmed that the uterine fluid had resolved. Six months after the laparoscopic removal of the Essure implants, pelvic MRI was performed and was normal, with no signs of contour defect, no retained Essure fragments, or residual fluid collection. The patient continues to do well, and her planned embryo transfer was postponed for personal reasons.
In IVF patients, fluid collections within either the fallopian tube(s) or endometrial compartment are well-known negative prognostic factors for reproductive outcomes. Although the exact mechanism remains unknown, the harmful effect of hydrosalpinx fluid may be due to the retrograde flow of embryotoxic substances into the endometrial compartment, the possibility that the fluid could mechanically flush or dislodge embryos after transfer, or both. Even in the absence of tubal pathology, fluid within the endometrial compartment is still detrimental to embryo implantation [4]. Although the standard management approach for the problem of hydrosalpinges before embryo transfer is laparoscopic surgery, a non-incisional hysteroscopy-based technique (Essure) has gained attention in recent years. As experience increases with Essure-and most published reports have been favorable-important questions regarding patient counseling, informed consent, post-Essure monitoring, and even techniques for implant removal require attention.
Some studies have indicated that it can be useful for selected IVF patients with hydrosalpinges to undergo the Essure procedure as a non-incisional alternative to laparoscopy [5]. Most research on Essure used in this way has focused on comparing the efficacy of Essure with the standard laparoscopic management of tubal disease. A recent Spanish study reported on 15 IVF patients for whom laparoscopy was contraindicated, and their outcomes were compared with patients undergoing laparoscopy [5]. The IVF pregnancy rates after Essure were slightly-but not significantly-lower than in the laparoscopic salpingectomy group, although this may not have been due to the Essure device per se, but rather because the ovarian reserve was lower among Essure patients [5].
The persistent endometrial fluid observed in the present case led to a recommendation for Essure removal before planned embryo transfer, although exactly how the Essure device may have caused this buildup of uterine fluid remains unclear. Considering the mechanism of action of Essure when it functions as a tubal contraceptive implant, it is possible that the inflammation is not limited to the immediate vicinity of the tubal ostia. This would seem more likely to be a problem when the proximal portion of the implant remains within the uterine cavity, as recommended by the manufacturer. Of course, if IVF were anticipated, this issue could be attenuated by ensuring that no implant component protruded medially past the tubal ostium (e.g., by positioning the Essure device completely within the fallopian tube). While this technique has not been standardized, a 'deep Essure placement' if IVF is planned has been proposed [6], in contrast to how Essure is inserted when used for conventional tubal sterilization [7].
According to the manufacturer's guidelines, bilateral tubal occlusion must be confirmed 3 months after Essure device placement by a HSG. By this time, fibrosis of the proximal Fallopian tubes has occurred, which leads to tubal occlusion and yields the desired contraceptive effect. Thus, patients must use a back-up method of birth control for at least 3 months after the Essure procedure. If at that point bilateral tubal occlusion is not confirmed on a HSG, the patient should continue to use an alternative method of contraception for three more months. Pregnancies that have occurred after hysteroscopic sterilization with Essure have been explained by lack of follow-up HSG, the improper use of alternative contraception, the failure of alternative contraception, or the misinterpretation of HSG results. Of note, a recent investigation of unplanned pregnancies with Essure revealed that 66% of contraceptive failures occurred among women who were compliant with their post-procedure HSG [8].
As of 2013, fewer than 15 cases of Essure removal have appeared in the global medical literature. While most removals are performed within 12 weeks of the initial insertion [3], no consensus exists regarding the preferred surgical approach to "late" Essure removal. For our patient, we used a modified Albright technique [3], in which laparoscopic partial bilateral salpingectomy was performed without salpingotomy. We agree that careful assessment of the tubal anatomy should take into account the exact alignment of the Essure device with respect to the surrounding tubal tissue. The length of the Essure device is 4 cm, and its distal end should therefore reside at the tubal isthmus, assuming proper insert placement and no migration.
Effecting an en bloc resection (i.e., removing the device while still within its associated tubal tissue), eliminates the need to expose the full Essure implant to the extreme temperatures from electrocautery. Given the polyethylene terephthalate (PET) component of Essure, we chose to use cautery sparingly during laparoscopy. The production of PET involves the catalytic use of antimony, which can still be found on product surfaces post-production. While this residue can be removed by irrigation, antimony remains in the Essure device itself and may dissipate under certain conditions. Extrapolating from experimental work in anon-surgical context, drinking water sold in plastic bottles containing PET can, when heated, release worrisome levels of leached antimony [9]. Accordingly, subjecting the Essure device to prolonged heat could increase antimony exposure [10], thus potentially dispersing this hazardous vapor throughout the abdominopelvic cavity. Our experience suggests that the Essure device can remain intact as long as the bipolar energy does not exceed 40 W of power. Since this approach had never before been attempted, even though the implants appeared grossly intact at surgery, a pelvic magnetic resonance image obtained within 30 days of removal indicated that complete extraction was achieved and no Essure fragments were retained. While magnetic resonance imaging up to a magnetic field strength of 1.5 T is not contraindicated for patients with Essure, a postoperative HSG or ultrasound may provide similar reassurance with comparable sensitivity.
While Essure may be considered for sterilization when laparoscopy is contraindicated, little clinical experience has been reported of the use of Essure as a treatment for hydrosalpinges before IVF. To be sure, most Essure placements before IVF reported to date have been successful; but no consensus exists about how to evaluate, counsel, and obtain informed consent from such patients. If the Essure procedure becomes more common for hydrosalpinges in the future, a standardized approach to informed consent should be developed. Unfortunately for our patient, after undergoing the Essure procedure, the problem of hydrosalpinx fluid was replaced with a different problem-endometrial compartment fluid. The development of Essure-associated intracavitary fluid that was corrected after Essure removal suggests additional points for consideration regarding the use of this device before planned IVF.
References
1. Mahmoud MS, Fridman D, Merhi ZO. Subserosal misplacement of Essure device manifested by late-onset acute pelvic pain. Fertil Steril 2009;92:2038.e1-2038.e3.PMID: 19819445.


2. Belotte J, Shavell VI, Awonuga AO, Diamond MP, Berman JM, Yancy AF. Small bowel obstruction subsequent to Essure microinsert sterilization: a case report. Fertil Steril 2011;96:e4-e6.PMID: 21565339.


3. Albright CM, Frishman GN, Bhagavath B. Surgical aspects of removal of Essure microinsert. Contraception 2013;88:334-336.PMID: 23290426.


4. He RH, Gao HJ, Li YQ, Zhu XM. The associated factors to endometrial cavity fluid and the relevant impact on the IVF-ET outcome. Reprod Biol Endocrinol 2010;8:46PMID: 20465847.



5. Matorras R, Rabanal A, Prieto B, Diez S, Brouard I, Mendoza R, et al. Hysteroscopic hydrosalpinx occlusion with Essure device in IVF patients when salpingectomy or laparoscopy is contraindicated. Eur J Obstet Gynecol Reprod Biol 2013;169:54-59.PMID: 23561020.


6. Thebault N, Broux PL, Moy L, Vialard J. Utilization of Essure® micro-insert for hydrosalpinx occlusion in infertile women. J Gynecol Obstet Biol Reprod (Paris) 2012;41:145-150.PMID: 22226634.


7. Mijatovic V, Veersema S, Emanuel MH, Schats R, Hompes PG. Essure hysteroscopic tubal occlusion device for the treatment of hydrosalpinx prior to in vitro fertilization-embryo transfer in patients with a contraindication for laparoscopy. Fertil Steril 2010;93:1338-1342.PMID: 19147140.


8. Sills ES, Li X, Jones CA, Wood SH. Contraceptive failure after hysteroscopic sterilization: analysis of clinical and demographic data from 103 unplanned pregnancies. Obstet Gynecol Sci 2015;In press.

9. Westerhoff P, Prapaipong P, Shock E, Hillaireau A. Antimony leaching from polyethylene terephthalate (PET) plastic used for bottled drinking water. Water Res 2008;42:551-556.PMID: 17707454.


10. Cheng X, Shi H, Adams CD, Ma Y. Assessment of metal contaminations leaching out from recycling plastic bottles upon treatments. Environ Sci Pollut Res Int 2010;17:1323-1330.PMID: 20309737.


Figure┬Ā1
(A) Pre-placement view of an Essure contraceptive implant, depicting the inner rod (arrow) and the terminal marker for the outer Nitinol coil (circle). (B) An intraoperative photograph showing intact surgical excision of the device, including the inner rod (arrow) and the outer coil terminal marker (circle).