Significance of the serum CA-125 level in intrauterine insemination cycles
Article information
Abstract
Objective
There are limited data regarding the significance of elevated serum CA-125 level during IUI cycles, even though it is used widely during the initial evaluation of infertile patients. The aim of this study was to investigate the prognostic value of serum CA-125 levels during IUI cycles.
Methods
Among the patients with controlled ovarian stimulation and IUI cycles at Seoul National University Hospital from Jan 2005 through Dec 2009, 92 cases with no identified endometriotic lesion, ovarian tumor, salpingeal lesion, or uterine myoma were selected. To compare the clinical characteristics between the pregnancy group and the non-pregnancy group, the Mann-Whitney U test and Fisher's exact test were used.
Results
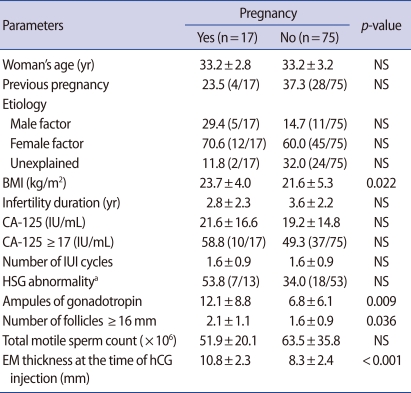
The overall pregnancy rate was 18.5% (17/92). The pregnancy group showed a higher number of follicles 16 mm in diameter (p=0.036), endometrial thickness (p<0.001), ampules of gonadotropin (p=0.009), and higher body mass index (p=0.022) than the non-pregnancy group. No significant difference was observed in the serum CA-125 level or the proportion of patients with CA-125 exceeding 17 IU/mL between the two groups.
Conclusion
The prognostic value of serum CA-125 level among infertile patients with IUI cycles is considered limited.
Introduction
IUI is a common procedure applicable in cases of cervical infertility, male factor infertility, anovulation, endometriosis with a healthy fallopian tube, and unexplained infertility. Endometriosis, uterine myomas, acute and chronic salpingitis, and pelvic inflammatory disease are female factors that are known to have a negative impact on the pregnancy rate during the IUI cycle [1-3]. Still, the mechanisms by which these factors affect the outcome of infertility treatment remain unclear.
CA-125 is a high-molecular-weight glycoprotein that presents on the epithelium of the fallopian tubes, endometrium, endocervix, as well as other tissues of Müllerian origin [4,5]. Measurement and monitoring of CA-125 level may be a simple and inexpensive test that can identify a subgroup of patients who are likely to have pelvic pathologies such as endometriosis, uterine myomas, acute and chronic salpingitis, ovarian tumors, and pelvic inflammatory disease, which are not detectable in the initial examination [5]. Little data is currently available regarding the cutoff values for screening for a uterine myoma or an adnexal mass. Previous studies focused mainly on the detection of endometriosis using the serum CA-125 level and a serum level of 17-35 U/mL has been reported to have sensitivities of 20-94% and specificities of 77-92% in detection of endometriosis grade I/IV [6-9]. Hence we hypothesized that an elevation of the serum CA-125 level may be linked to a lower pregnancy rate in IUI cycles. To our knowledge, few studies have addressed the relationship between the serum CA-125 level and the outcome of IUI. In this study, we aimed to explore whether the serum CA-125 level differs according to the outcomes of IUI cycles.
Methods
Patients who had undergone controlled ovarian stimulation and IUI cycles at Seoul National University Hospital from January 2005 to December 2009 were selected. All couples had a duration of infertility of more than one year and were eligible for superovulation and IUI cycles. Cases with a physically or sonographically identifiable endometriotic lesion, ovarian or salpingeal mass, or uterine myoma were excluded. Other pelvic abnormalities such as unilateral tubal obstruction or uterine anomaly were classified as female causes of infertility. Male factor infertility was defined by the World Health Organization criteria [10]. Serum CA-125 levels were measured on menstrual day 3 with other steroid hormones as an initial evaluation of infertility using radioimmunoassay kit (Fujirebio Diagnostics, Malvern, PA, USA).
Controlled ovarian stimulation (COS) was performed with urinary hMG (IVF-M®, LG Life Sciences, Seoul, Korea) and clomiphene citrate (Clomiphene® Youngpoong Pharma, Seoul, Korea) in a dose of 150 mg/day given from day 3 through day 7 of the menstrual cycle. A recombinant hCG (Ovidrel® Serono, Geneva, Switzerland) in a dose of 250 μg was given when either the leading follicle reached a mean diameter of 18 mm or two or more follicles reached a diameter of 17 mm. IUI was then performed 36 hours later.
The luteal phase was supported by a daily application of 8% progesterone gel (Crinone® Serono, Geneva, Switzerland), initially for 14 days, starting on the day of the IUI, and continuing for another 6-8 weeks in cases with pregnancy. Clinical pregnancy was defined by the presence of an intrauterine gestational sac with fetal heartbeats at 3 to 4 weeks after insemination.
Data were analyzed with SPSS ver. 16.0 (SPSS Inc., Chicago, IL, USA). Fisher's exact test was used to compare proportions, and the Mann-Whitney U test to compare means. A p-values less than 0.05 were considered statistically significant.
Results
The overall pregnancy rate was 18.5% (17/92). The mean age was 33.2±3.1 years; the mean duration of infertility was 3.4±2.2 years, and the mean body mass index (BMI) was 22.0±5.1 kg/m2. More than a third of the women (34.8%) had achieved a prior pregnancy. The infertility factors of the subjects were identified as unexplained (28.3%), female (62.0%), and male (17.4%) factors. The serum level of CA-125 ranged from 1.7 to 77.1 IU/mL in the study population. Among the pregnancy group and the non-pregnancy group, there was no significant difference in patient's age, incidence of nulliparity, cause and duration of infertility. The mean BMI of the pregnancy group was significantly higher than that of the non-pregnancy group (23.7±4.0 vs. 21.6±5.3, p=0.022). The pregnancy group showed a higher number of follicles with a diameter greater than 16 mm (p=0.036), greater endometrial thickness (p<0.001), and a greater number of gonadotropin ampoules (p=0.009). No significant differences were observed in the serum CA-125 levels and the percentage of patients with CA-125 exceeding 17 IU/mL between the two groups (Table 1).
Discussion
The association of the serum CA-125 level with the outcome of in vitro fertilization has been investigated in a number of studies and the results to date have been contradictory [11-15]. To our knowledge, there have been no published studiesregarding serum CA-125 levels before COS with an IUI cycle and pregnancy. Mild elevation of the serum CA-125 level before ovarian stimulation may reflect undetected abnormal pelvic conditions such as endometriosis, uterine myomas, acute/chronic salpingitis, pelvic inflammatory disease, and ovarian tumors, all of which are known to affect fecundity [1,16,17]. The mechanisms of reduced fertility in minimal to mild endometriosis, distal tubal disease, or adnexal lesion are unknown. In endometriosis, for example, some investigators have suggested that cytokines and growth factors secreted by the ectopic endometrial tissue may interfere with ovulation, fertilization, implantation, and embryonic development [18]. However, in the present study, we have found no significant relationship between the success of IUI and the serum CA-125 level, suggesting that the measurement of CA-125 in infertile patients with no history of pelvic pathology would not be valuable in predicting IUI outcome. It is possible that subclinical pelvic pathology combined with a mild elevation of CA-125 does not have a negative effect on the outcome of IUI. Elevation of the serum CA-125 level does not directly correlate to pelvic pathologies since not all of the patients had undertaken laparoscopic examination. Singh and colleagues reported that individuals diagnosed with mild adnexal adhesion through laparoscopic examination did not have a significantly different pregnancy rate following IUI than infertility patients [16].
The pregnancy rate of IUI has been reported to range from 10 to 14% per cycle [3,19,20]. Many factors have been demonstrated to influence the success of IUI, though there is some disagreement about the ranking of the factors [3,19-21]. Although the size of study population was relatively small, we also observed results inconsistent with previous studies. In previous investigations, the woman's age has been shown to affect the outcome of IUI, while we found no significant difference in the patient's age according to outcome of IUI. In addition, the mean BMI of the pregnancy group was significantly higher than that of the non-pregnancy group in the present study, consistent with the result of Wang et al. [22] Since conflicting data on the association between BMI and the success of IUI have been reported, this topic needs further investigation [23-25].
In this investigation, we observed that serum CA-125 level has no prognostic value among infertile patients without gross pelvic pathology undergoing IUI cycles. Careful consideration must be given regarding routine measurement of serum CA-125 level in infertile patients with no pelvic pathology.
Notes
No potential conflict of interest relevant to this article was reported.