Nanotechnology in reproductive medicine: Opportunities for clinical translation
Article information
Abstract
In recent years, nanotechnology has revolutionized global healthcare and has been predicted to exert a remarkable effect on clinical medicine. In this context, the clinical use of nanomaterials for cancer diagnosis, fertility preservation, and the management of infertility and other pathologies linked to pubertal development, menopause, sexually transmitted infections, and HIV (human immunodeficiency virus) has substantial promise to fill the existing lacunae in reproductive healthcare. Of late, a number of clinical trials involving the use of nanoparticles for the early detection of reproductive tract infections and cancers, targeted drug delivery, and cellular therapeutics have been conducted. However, most of these trials of nanoengineering are still at a nascent stage, and better synergy between pharmaceutics, chemistry, and cutting-edge molecular sciences is needed for effective translation of these interventions from bench to bedside. To bridge the gap between translational outcome and product development, strategic partnerships with the insight and ability to anticipate challenges, as well as an in-depth understanding of the molecular pathways involved, are highly essential. Such amalgamations would overcome the regulatory gauntlet and technical hurdles, thereby facilitating the effective clinical translation of these nano-based tools and technologies. The present review comprehensively focuses on emerging applications of nanotechnology, which holds enormous promise for improved therapeutics and early diagnosis of various human reproductive tract diseases and conditions.
Introduction
Reproductive medicine is an emerging field that focuses on male and female reproductive tract function and associated clinical issues such as fertility preservation, infertility, pubertal development, menopause, and sexually transmitted infections (STIs), including AIDS (acquired immunodeficiency syndrome) and cancers [1]. Over the years, reproductive medicine has come a long way, as remarkable technological advancements have given the gift of parenthood to infertile couples by empowering them to manage their fertility [2]. However, the objectives of reproductive medicine are not confined to only conception and delivery-related issues, but also include fertility-sparing treatments, selection and micromanipulation of gametes and embryos, in vitro culture, preimplantation genetic testing, and reproductive cryopreservation and associated diseases and conditions [3,4]. Despite recent advances in this particular field of clinical medicine, the existing challenges are still enormous [5]. To confront such challenges, nanoengineering-based tools and technologies that offer minimally invasive detection and treatment of reproductive tract–associated pathologies are indispensable and warrant an innovative plan of action for the development of therapeutic interventions depending upon the stage and type of diseases and conditions [6]. In order to put forward any such strategy, it is essential to design and develop novel applications using functionalized nanomaterial-based methodologies that are rapid, selective, and sensitive in nature. Of course, the effective implementation of such interventions will require careful validation in control, experimental, and field settings.
The concept of nanotechnology dates back to 1959, when an American theoretical physicist, Professor Richard P. Feynman, at the Annual Physical Society Meeting at Caltech emphasized a conceptual framework that involves direct manipulation of individual atoms using synthetic chemistry to develop denser computer circuitry-based imaging platforms that could visualize tinier objects with higher resolution [7]. Although he never used the term “nanoscience,” his pioneering views laid the foundation of the newly emerging field. In later years, Professor Norio Taniguichi, a MIT-based American engineer, drew significant attention from a wider audience to the nanotechnological field. During the 1990s, many nanotechnological applications were derived from a specialized form of materials science [8]. However, these materials are currently used for a number of applications in various fields such as energy, electronics, food and agriculture, cosmetics, and healthcare. With the availability of enormous opportunities and resources at public-funded institutions and in the industrial research and development sector, the field of nanotechnology is growing at an exponential scale. The trends in the development of improved nanotechnological techniques over conventional research and therapeutic approaches will inexorably encourage the rapid broadcasting of the nanobiotechnological “vision” to a growing number of research and clinical disciplines [9,10].
Over the last decade, the advent of new applications and technologies linked to biomedical nanotechnology have revolutionized existing preventive, diagnostic and treatment approaches. This innovative approach has paved the way for development of improved and very sensitive tools for the investigation of the mechanisms underlying the biology of various diseases. This has been the major driving force leading to the expansion of global health sector [11-13]. The global market of healthcare nanotechnology (including nanomedicine) accounted for about 78.54 billion USD in 2012, which was expected to rise to 177.60 billion USD in 2020 with a compound annual growth rate of 12.3% from 2012 to 2020 [14]. A plethora of work is currently under progress in the healthcare sector. This includes a spectrum of systems that target specific cells or proteins from the systemic circulation to frameworks that incorporate multiple drugs and diagnostic agents, all the way to developing novel formulations that can deliver a diverse set of agents through the oral route. Some of these nanoproducts developed by the biotechnological and pharmaceutical industry have demonstrated remarkable improvements in drug delivery systems, medical imaging, theranostics, biodegradable implantable materials, tissue regeneration strategies, and diagnostic platforms [15,16].
Among the potential clinical applications of nanomaterial-based approaches, the use of investigational nanobiotechnological tools in reproductive medicine has already resulted in encouraging outcomes in the treatment of several high-impact conditions, opening significant opportunities for alternative non-invasive or minimally-invasive treatments for several traditionally “surgical” pathologies. The use of such tools has immensely contributed towards improved technologies for fertility preservation and the diagnosis and treatment of infertility and other clinical problems associated with puberty, menopause, STIs (including HIV [human immunodeficiency virus]), and cancers of the reproductive tract [17]. In reproductive biology, the availability of versatile delivery vehicles with a large loading capacity and spontaneous internalization into target cells has created unprecedented possibilities to explore and manipulate the fine mechanisms underlying reproduction and early embryo development for research purposes. Of late, a few experimental investigations using nanodrug vectors for endometriosis, uterine fibroids, ectopic pregnancy, and trophoblastic diseases have also been reported. Most of the trials involving nanoengineering methods for fertility regulation and treatment of reproductive cancers are still at the initial stage [18].
The present review focuses on all emerging aspects of nanomaterial-based approaches for the precise identification, therapy, and monitoring of reproductive tract-associated disorders, which are at the center of attention in reproductive medicine, along with their bench-to-bedside translational potential to the point-of-care setting in a comprehensive fashion.
Assisted reproductive technologies in clinical practice
Reproductive medicine is an emerging discipline that not only deals with the application of novel approaches to address issues related to successful pregnancy and preservation of fertility, but also helps in the diagnosis and treatment of disorders or ailments that disrupt the normal functioning of the reproductive tract. In light of advances in the field of reproductive medicine, assisted reproductive technology (ART) has emerged as a successful and widely performed treatment paradigm throughout the world that involves a set of procedures that assist with infertility-related issues in males and females [6,19,20]. ART includes the use of in vitro fertilization (IVF), zygote intrafallopian transfer (ZIFT), gamete intrafallopian transfer (GIFT) and intracytoplasmic sperm injection (ICSI). IVF is generally applicable for women with low ova production or men with a low sperm count. In IVF, women are treated with multiple drugs that lead to the production of numerous eggs, which are removed and fertilized in vitro and the healthy embryos are finally implanted in the uterus. ZIFT involves a similar procedure to IVF with the striking difference that the embryos are transferred in the fallopian tube. GIFT involves the transfer of eggs and sperm in the fallopian tube where the fertilization takes place. ICSI is often appropriate for couples affected by severe inadequacy of the sperm count, wherein a single sperm is injected into a mature egg followed by implantation to the uterus or transfer to the fallopian tube [1,21].
The past few decades have witnessed a substantial augmentation in the frequency of ART procedures, and ART has made it possible for countless infertile couples to experience parenthood. However, excessive usage is generally accompanied by elevated perinatal risk and problems related to multifetal gestations. Cases of multiple gestations are often found to be associated with risks of maternal and fetal morbidity and mortality due to factors such as pre-term birth, pre-eclampsia, and pregnancy-related complications. However, evidence supporting the generation of risk associated with multi-fetal gestations arising due to the application of excessive ART is limited. Apart from the aforementioned problems, the increased use of ART may also lead to the potential danger of monozygotic twinning, which may contribute to growth abnormalities and twin-to-twin transfusion [22,23]. Such complications could be overcome by appropriate consideration of the maternal condition as well as a thorough assessment of the patient’s obstetric history prior to ART procedures. In addition to these, determination of pre-existing disorders of the cardiopulmonary system and common ailments such as diabetes, hypertension, epilepsy, obesity, maternal medical status, treatment regimen, and other aspects of care may have productive effects on pregnancy outcomes and significantly reduce risk to the woman’s life and health. In men, detection of oligospermia and azoospermia, which are indicative of male infertility, by testing for cystic fibrosis, Y micro-deletion and karyotyping are essential to prevent any further complications in ART procedures such as IVF and ICSI. In addition to tests to identify any conditions that may cause complications prior to commencement of the ART procedures, couples must be counselled about the risks associated with the procedures [24,25].
Biomarkers indicative of reproductive impairment
Biomarkers indicative of reproductive disorders might aid in the real-time assessment of exposure, early detection, identification of patients who require immediate treatment, prediction of outcomes, monitoring of disease progress, and stratification of the population on the basis of the etiology and severity of diseases. Despite the aforementioned benefits of biomarkers, the field of reproductive medicine has received remarkably little attention in the development of novel molecular signatures. This may be due to the limited number of novel molecular entities entering clinical investigations. With the increase in the number of molecular markers that have been identified in reproductive science, it is highly imperative to understand the pathway from discovery to translational impact of a disease-associated signature, as most markers may not be clinically relevant due to a variety of complexities [26,27]. Therefore, extensive evaluation, standardization, and validation are required before establishing a molecular signature as clinical biomarker for disease assessment and monitoring. In the most recent decade, biomarker research has witnessed a significant resurgence in interest due to advancements in “omics” methodologies, which involve generating voluminous amounts of data by high-throughput systems. These advances have made it possible to establish fascinating association of novel biomarkers and disease mechanisms in a relatively easy way for early screening [24,28]. The challenge is to validate such associations and to move them into the clinical setting. Reproductive medicine is still in its infancy in terms of integrating novel biomarkers into precision medicine. However, concerted efforts and speedy advancements in the research in reproductive medicine might narrow the gap of the translational process.
Pursuit of nanotechnology in reproductive medicine: accomplishments and challenges
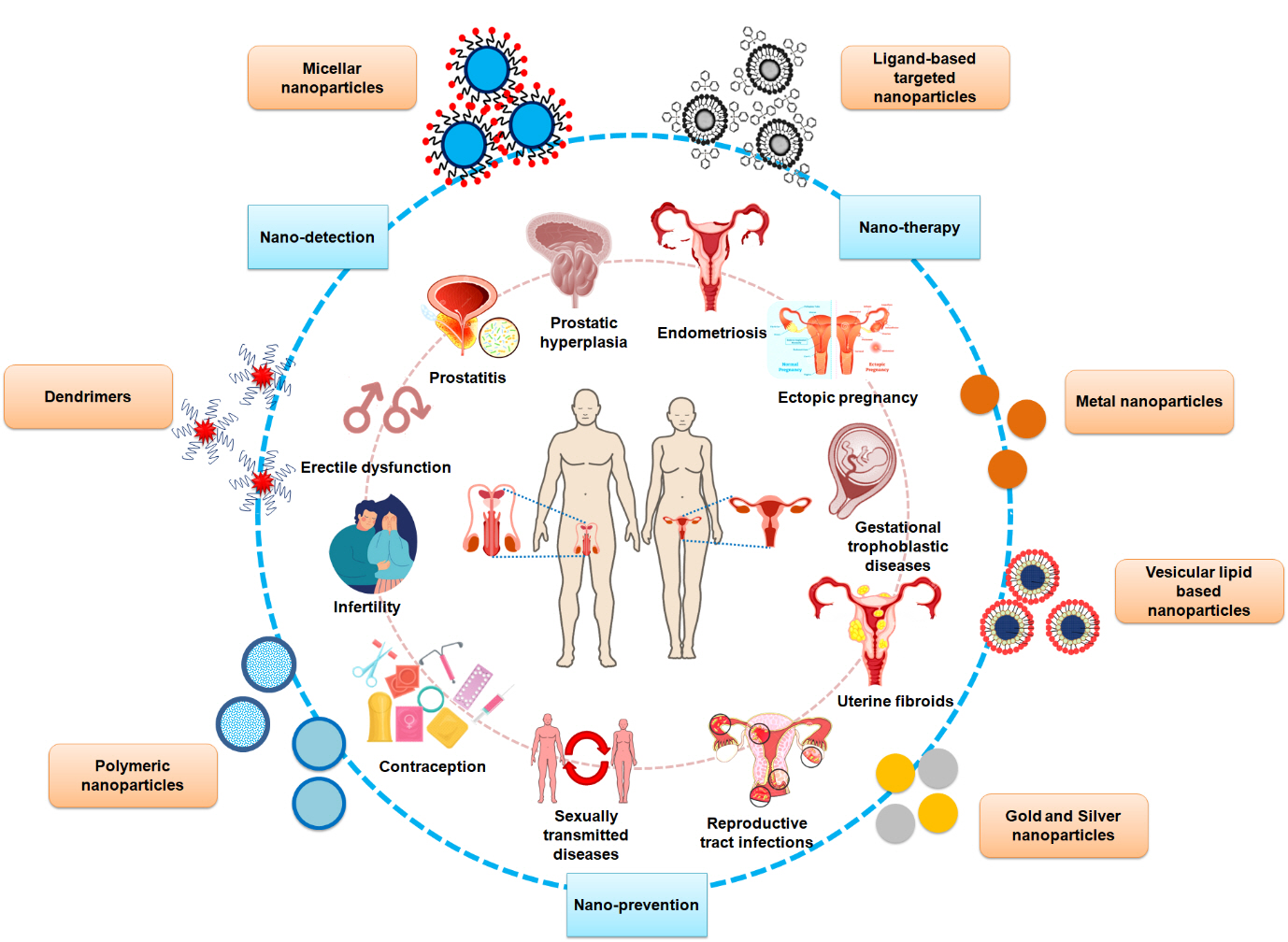
Increasingly many studies have affirmed that impotence and reproductive disorders, such as endometriosis, adenomyosis, polycystic ovary syndrome, and uterine fibroids, negatively affect pregnancy. Repeated surgical interventions may reduce the likelihood of natural and assisted conception, which may result in an increasing dependence on third-party reproductive methodologies. The combination of nanotechnology-based approaches with reproductive medicine has led to the development of safer strategies for improving diagnosis and increasing precision and responsiveness [29]. Several studies have demonstrated the potential of nanomaterial-based methodologies that offer efficient noninvasive detection, treatment and monitoring of common disorders that may affect women of reproductive age (Figure 1).
1. Endometriosis
Endometriosis and fibroids represent the most common female reproductive disorders; endometriosis is present in approximately 10%–15% of reproductive-age women and in 70% of women with chronic pelvic pain. Endometriosis represents the commonest form of pregnancy-related complication and affects both the endometrial glands and the stroma outside of the endometrial cavity. The increase in the percentage of women affected by endometriosis is due to the late diagnosis of the disease, which results in significant reduction in the quality of life due to symptoms including non-menstrual pelvic pain, dyspareunia, and infertility [30,31]. Since the majority of women report the symptoms of endometriosis in adolescence, early detection and timely treatment may significantly alleviate pain and avert progression of the disease, thereby preserving fertility. The treatment approach has recently been gradually shifting from surgical ablation to medication control, and it is therefore highly desirable to develop a non-surgical mode of diagnosis. Several investigative studies have reported the application of nanomaterial-based approaches for the treatment of endometriosis and uterine fibroids. For instance, a novel mitigation approach using nanoceria (cerium oxide nanoparticles) was demonstrated in an endometriosis-induced mouse model by Chaudhury et al. [32], who reported a considerable reduction of endometrial glands, microvessel density in the peritoneum, and endometrial lesions upon treatment with nanoceria, highlighting the antioxidant and anti-angiogenic potential of the developed system, both in vitro and in vivo. The system also exhibited a promising reduction in the adverse effects related to endometriosis on the quality of oocytes, which is a critical factor for successful pregnancy. A novel herbal nanoformulation comprising a Copaifera langsdorffii oil-resin nanocomposite showed evidence of reduction of cell viability, alterations of cellular morphology, and induction of necrosis and apoptosis in proliferative primary endometrial and endometriotic stromal cells [33,34]. Singh et al. [35] reported a combinatorial approach for the treatment of endometriosis that consisted of dual drug-loaded poly(lactic-co-glycolic acid) (PLGA) nanoparticles combining the anti-angiogenic and antioxidant properties of epigallocatechin gallate and the targeted matrix metalloproteinase inhibitory activity of doxycycline. A novel laser-mediated photothermal ablation therapy of endometriosis was reported to show enhanced permeability and retention and targeted delivery of gold nanoparticles to endometriotic sites using the TNYL peptide, which has a strong affinity for overexpressed EphB4 receptors in endometriosis lesions [36].
The increase in the number of women experiencing discomfort due to endometriosis is attributable to late diagnosis; this problem is exacerbated if the ovaries are involved, resulting in the formation of cysts, often termed as endometriomas. The discomfort and complications associated with endometriosis may be avoided if it is diagnosed at an early stage, providing better possibilities for pain and infertility management. Conventional diagnostic procedures such as operative laparoscopy and biopsy are often invasive in nature, which is a major cause of distress among patients that hinders early screening [37]. Such situations could be avoided by employing nanomaterial-based sensing approaches, which offer real-time assessment of the disease. Several nano-sensing strategies have been proposed by investigators for effective detection of endometriosis, enabling timely commencement of therapy. The presence of endometriosis, owing to its inflammatory characteristics, is often characterized by disturbances in the immunological environment, which is strongly reflected by alterations in the expression levels of various cytokines, angiogenic factors, matrix metalloproteases, tumor suppressor genes, and circulating nucleic acids [38,39]. Cathepsins (in particular cathepsins B, D, and G), which belong to the class of proteases, have been quantitatively evaluated in patients with proliferative eutopic endometrium using a surface plasmon resonance (SPR) imaging technique, since cathepsins have been found to have a positive impact on the establishment of endometriotic lesions. The results of the study demonstrated a significantly higher expression of cathepsin G (CatG) in eutopic endometriotic patients than in the control group [40,41]. In view of this, an SPR-chip-based biosensor was developed by Grzywa et al. [42] for the selective determination of CatG in endometrium samples of patients and healthy controls. The gold-chip was surface-tethered with a phosphonic-type inhibitor that offered selective determination of active CatG in the sample at a picomolar concentration. For the in vivo detection of endometriotic lesions, hyaluronic acid (HA)-attached magnetic iron oxide nanoparticles (HA-Fe3O4 NPs) were developed as novel contrast agents for magnetic resonance imaging (MRI) in rodents. The results of the study highlighted the potential of the developed systems for hyperthermal treatment of endometriosis in the future [43].
2. Uterine fibroids
Uterine fibroids, often known as uterine leiomyomas, are non-cancerous benign tumors that often appear during pregnancy. After endometriosis, uterine fibroids are among the most commonly occurring solid tumors in women of reproductive age [31]. These neoplasms occur in about 77% of women, including 25% of those above the age of 45 years. The presence of fibroids in the uterus is often accompanied by symptoms with a major impact on quality of life, such as irregular and prolonged menstrual bleeding, anemia, pelvic discomfort, pelvic masses, bowel and bladder dysfunction, and obstetric complications. The presence of uterine fibroids has been strongly correlated with infertility and abortion, which significantly affect women’s health and quality of life and may necessitate the use of ART [44,45]. Early treatment of fibroids, which hamper fertility, might increase the likelihood of future pregnancies. Shalaby et al. [46] demonstrated an effective noninvasive adenovirus-based alternative for the treatment of uterine fibroids using a combination of viral-based gene delivery with nanotechnology. A targeted magnetic nanoparticle-based approach was adopted for the efficient transduction of adenovirus under an external magnetic field. The novel combinatorial method offered a paradigm shift in therapeutic interventions for uterine fibroids, as it may significantly eliminate tumor-forming fibroid stem cells, which currently pose a major challenge to treatment.
3. Pregnancy-related complications: ectopic pregnancy and gestational trophoblastic diseases
Ectopic pregnancy is the most common cause of death among women during the early stages of pregnancy. An ectopic pregnancy occurs when the fertilized egg/embryo is implanted outside of the uterine cavity. The overwhelming majority of ectopic pregnancies (97%) involve implantation in the fallopian tube, while in the remaining 3% of cases, implantation occurs in the cervix, ovary, peritoneal cavity, or uterine scars. This pregnancy-related complication can result in rupture of the tube, leading to life-threatening internal bleeding. Ectopic pregnancies account for about 4% to 10% of pregnancy-related deaths and are characterized by a high incidence of ectopic site gestations in subsequent pregnancies [47]. Although several advances have been made in the detection and treatment of ectopic pregnancy, not all can be considered highly effective. Therefore, there is an urgent need for the development of diagnostic and therapeutic interventions that focus on precisely identifying and addressing the causes of the disease without compromising the other reproductive organs. Nanomedicine comprises an amalgamation of medicine and nanotechnology for the development of methodologies or strategies to improve the safety and efficacy of the conventional drugs by achieving targeted delivery to the site of action. Such precise targetability is urgently required for the development of novel methods of drug transport in women with pregnancy-related complications or diseases [48,49]. Fertility preservation-based surgical interventions are a major challenge in initial-stage ectopic pregnancy cases, especially among women with rare locations of extra-uterine pregnancies. This challenge has led medical practitioners to seek alternative methods for treatment. For instance, doxorubicin-loaded EnGeneIC delivery vehicles were prepared for targeted delivery to the epidermal growth factor receptor expressed on placental cells. In vitro, ex vivo, and in vivo studies demonstrated significant inhibition of the trophoblastic tumor cell proliferation mediated by an increase in apoptosis, which offers a novel alternative to ectopic pregnancy treatments [50]. Complications could be avoided if ectopic pregnancy is diagnosed at the earliest possible stage. In recent years, significant advances in the field of nanotechnology have sparked research interest in the development of early detection approaches using nano-biosensing. Studies have reported the detection of human chorionic gonadotropin as an important protein marker indicative of pregnancy-related complications such as ectopic pregnancy, miscarriage, fetal abnormalities, and testicular tumors using ultrasensitive and highly selective carboxyl-graphene oxide–based SPR aptasensor and electrical double layer-gated field effect transistor-based chip sensor [51].
Another major pregnancy-related complication is gestational trophoblastic diseases (GTDs) which comprise a spectrum of tumors with a wide range of biological behavior and potential for distant metastases that arise from placental trophoblastic tissue after normal or abnormal fertilization [52]. Gestational choriocarcinoma is the most prevalent GTD. Despite progress in the field of modern reproductive medicine, gestational trophoblastic tumors continue to pose a significant challenges both for diagnosis and treatment owing to their irregularity, broad range of differences in diagnoses, and the uncertainty of detection of the precursor lesions [53]. These factors have fueled the need for extensive research on the development of precise and selective strategies for identifying and treating these tumors through nanotechnology. A glycosaminoglycan, placental chondroitin sulphate A (plCSA), is highly expressed in a wide range of cancer cells and placental trophoblasts, highlighting its potential as a target in the treatment of GTDs [54]. Recently, a study investigated the efficient delivery of methotrexate to placental cells using synthetic plCSA binding peptide (plCSA-BP)–tethered lipid-based nanoparticles as a novel targeted approach for the treatment of pregnancy-related complications such as ectopic pregnancy and choriocarcinoma. The results of the research illustrated efficient binding of the plCSA-BP attached to the methotrexate-loaded nanoparticles to human placental syncytiotrophoblasts and mouse trophoblasts throughout gestation after targeted delivery of the drug to the placenta of the mouse model without any fatal effects on the fetus [55]. Likewise, a similar study illustrated the development of plCSA-BP-decorated polymeric core-lipid shell nanoparticles for the targeted delivery of doxorubicin to the placental choriocarcinoma (JEG3) cells [54]. Therapeutic interventions in patients with gestational trophoblastic choriocarcinoma often involve the application of systemic anti-neoplastic agents at high doses, which results in arbitrary distribution of the drugs to other organs and consequent systemic toxicity. To avoid such adverse effects, in light of the targeting potential of plCSA-BP, Zhang and co-investigators demonstrated the efficient delivery of doxorubicin by nanoparticles to choriocarcinoma cells as a novel targeted cancer therapeutic approach. The results of the investigation reported competent internalization in the lysosomes resulting in increased in vitro anti-cancer action, rapid localization in the tumors, inhibition of primary tumor growth, and suppression of metastasis when observed in vivo using a rodent model [56].
4. Sexually transmitted infections and HIV
Reproductive tract infections (RTIs) are a concealed epidemic affecting the quality of life of both women and men, as well as compromising health and economic conditions throughout the world by means of their severe consequences, which include pelvic inflammatory disease, infertility (in both women and men), ectopic pregnancy, and adverse pregnancy outcomes such as miscarriage, stillbirth, preterm birth, and congenital infections. According to World Health Organization estimates, about 200 million cases of RTIs among women due to sexual transmission are reported every year in developing countries [5,57]. The reproductive-age population is highly susceptible to developing RTIs, especially women. RTIs include endogenously occurring infections such as bacterial vaginosis; infections transmitted due to sexual contact such as gonorrhoea, chlamydia, syphilis, chancroid, trichomoniasis, genital herpes, genital warts, and HIV; and iatrogenic infections such as pelvic inflammatory disease. Prompt diagnosis and treatment would reduce the transmission of these diseases [5,58]. Several studies have reported the use of nanomaterial-based treatment and monitoring approaches for safeguarding reproductive health and improving their quality of life. There is ample evidence of the usage of nanoparticles for the prevention, treatment, and early detection of RTIs, including STIs. For instance, oxygen vacancies comprising zinc oxide tetrapod nanoparticles have been fabricated for the nano-immunotherapy of genital herpes in females. This nano-system offered a great potential as an intravaginal microbicide/vaccine, leading to a significant reduction in vaginal infections and animal deaths, as well as an increase in the T cell-mediated and antibody-mediated responses, which subsequently suppressed re-infections [59]. Another group reported the treatment of genital herpes using acyclovir as a model drug encapsulated in polyvinyl pyrrolidone–Eudragit RSPO hybrid polymeric nanoparticles in the form of an in situ gel system. The developed formulation demonstrated a controlled release of the drug with improved permeability and viability for the vaginal epithelial cell lines, as well as a two-fold increase in the bioavailability of the drug in rat models in comparison to the pure drug, highlighting its potential for clinical therapy [60]. Several polyphenolic compounds exhibit anti-viral properties, among which tannic acid was used by Orlowski et al. [61], for the development of a synergistically acting formulation against herpes simplex virus 2 (HSV-2). For this, silver nanoparticles, with inherent anti-microbial activity, were synthesized using green chemistry followed by modification with tannic acid, which in mice models demonstrated an increment in levels of interferon-gamma-positive CD8+ T cells, activation of B cells and plasma cells, and reduction of viral titers in vaginal tissues shortly after the treatment. Another therapeutic approach for HSV-2-induced genital infections using exclusively customized zinc oxide tetrapod nanoparticles was described by Agelidis et al. [62], who reported a significant inhibitory effect on viral vaginal infections in female Balb/c mice. The proposed microbivac system is a promising platform for the development of live virus vaccines. A novel polymeric nano-carrier system developed by Ganda et al. exhibited excellent potential for the delivery of peptide vaccines. The system comprised fourth-generation hydroxyl-terminated polyamidoamine (PAMAM) dendrimers (G4OH) conjugated with an ester bond to a chlamydial glycolipid antigen mimicking peptide (peptide 4; Pep4). The G4OH and Pep4 bond, upon dissociation in the intracellular environment, led to antigen presentation, which induced chlamydia-specific serum antibody recruitment after subcutaneous immunization. The action of the formulation demonstrated an increased anti-chlamydia antibody response in mice owing to enhanced and sustained Pep4 immunogenicity as a consequence of ester bond dissociation by phagolysosomes [63]. Several reports have described examples of nanoparticle-based delivery to the reproductive organs, which offer bioavailability and biodistribution to a higher degree, as well as prolonged release/action and retention of the drug in comparison to the drug alone. In light of this, Park et al. [64], demonstrated the potential of an anti-retroviral drug, elvitegravir, loaded surface-tailored bio-adhesive poly(lactic acid)-hyperbranched polyglycerols (PLA-HPG) nanoparticle formulation for prolonged intravaginal delivery. The developed formulation exhibited a remarkable improvement (roughly five-fold) in the sustained delivery of the drug in comparison to the non-adhesive alternative formulation. The results of the research highlighted the potential of the adhesive PLA-HPG nano-formulation for intravaginal therapeutics to treat and prevent STIs [65]. Aside from STIs associated with HSV, Wagner et al. [66] developed a nanoparticle-based targeted approach that comprised microbicidal drugs encapsulated in mucous-penetrating PLGA-PEG nanoparticles for intravaginal inoculation against STIs. Soler et al. [67] illustrated the development of a rapid nano-plasmonic biosensor that offered simultaneous detection of Chlamydia trachomatis and Neisseria gonorrhoeae. The nano-biosensor comprised an array of gold nano-hole sensors that allowed precise detection and quantification of the levels of the two aforementioned bacterial strains in an amplification-free fashion.
STIs are among the most prevalent diseases around the world. Despite extensive research, STIs such as HIV and HSV still haunt a large population due to their incurability. Considering the present situation, the global health initiative programs have primarily focused on reducing the incidence of STIs, especially in highly susceptible female populations [68]. Several nanotechnology-based therapeutic, preventive, and early diagnostic approaches have emerged as potential alternatives to conventional orally administered medications. For instance, Mandal et al. [69] reported that a conventional anti-retroviral drug, emtricitabine, was encapsulated in PLGA polymeric nanoparticles to circumvent the limitations associated with the large volume of distribution, short plasma life, low bioavailability, and cytotoxic nature of the drug to improve its efficacy for the therapeutic treatment of HIV infection. Similar studies have demonstrated the encapsulation of conventional anti-retroviral drugs such as zidovudine, a combination of three agents (zidovudine, efavirenz, and lamivudine); and stavudine in PF-68-coated alginate nanoparticles, and lactoferrin nanoparticles and gelatine liposomal nano-formulations have been reported as strategies for enhanced anti-HIV therapy [70-72]. Efficient microbicide action was displayed by a combination of a HIV reverse transcriptase–inhibiting drug (efavirenz) and an integrase inhibitor (elvitegravir) encapsulated in a graft copolymer of methoxypolyethylene glycol-polylysine with a fatty hydrophobic core. The developed nano-formulation exhibited a significant reduction of cytotoxicity and in vivo bio-distribution upon topical intravaginal administration [73]. A similar effect was demonstrated by Mirani et al. [74], wherein a nano-formulation comprising a lipidic nano-microbicide gel was loaded with tetrahydrocurcumin. The formulation exhibited significant stability in the release profile and quick time-independent intracellular uptake, highlighting its potential for anti-retroviral therapy. A recent study illustrated a potential multiplexed in vivo treatment approach for HIV and HSV infections using a composite system consisting of methoxypoly(ethylene glycol)-b-poly(lactide-co-glycolide) Griffithsin nanoparticles incorporated in polycaprolactone fibers adjoined with polyethylene oxide fibers [75].
With advancements in the field of nanomaterials for diagnostic purposes, a surge of studies have been reported that offer rapid, and minimally-invasive methods for the early detection of STIs such as HIV and HSV. For the detection of human immunodeficiency virus type 1 (HIV-1) DNA, as a potential molecular signature of retroviral disease, a novel, user-friendly lateral flow assay was developed by Fu et al. [76] that utilizes surface-enhanced Raman scattering for precise identification and quantification. Another group reported the application of fluorescence resonance energy transfer for the detection of viral DNA using a combination of single-strand DNA (ssDNA)-tethered silver nanocrystals as donor and carbon nanoparticles as acceptor species. The combination exhibited desorption of the fluorescence in the presence of the target analyte, while restoration of the fluorescence in the absence of the same led to a highly sensitive diagnosis [77]. Fluorescence quenching as a mode of detection of HIV-1 DNA was employed by Deng et al. [78] and Fang et al. [79], who used fluorescent quantum dot–based lateral flow strips and surface DNA-decorated fluorescent silver nanocrystals, respectively. The HIV-related p24 antigen has emerged as another highly specific marker for the prompt detection of HIV in the recent years. Nano-detection approaches, such as a sandwich immunoassay amalgamating optoplasmonics and a micro-cantilever-based nanoplatform, have been developed for the precise detection of the HIV-1 capsid antigen p24 directly from human serum samples [80]. A similar sandwiched immunoassay developed by Chunduri et al. [81], involving the application of surface streptavidin-decorated europium-doped fluorescent silica nanoparticles, demonstrated a 100-fold improvement in detection of the HIV-1 p24 antigen in clinical samples compared to conventional enzyme-linked immunosorbent assay.
5. Gene therapy for reproductive disorders
Efficient gene therapy for the treatment of endometriosis was demonstrated by Zhao et al. [82], through the application of pigment epithelium-derived factor plasmid encapsulated in lipid-grafted chitosan micelles. An in vivo study in a rat model showed a significant diminishment of endometriotic lesions, as well as atrophy and degeneration of ectopic endometrium with no cytotoxic effect on the reproductive organs. A similar gene therapy for endometriosis was reported by Wang et al. [83], using endostatin-loaded PAMAM (PAMAM-Es) dendrimers in a non-invasive animal model. The results demonstrated a significant reduction in endometriotic lesions owing to the anti-angiogenic mechanisms of the PAMAM-Es in comparison to the traditional gene carrier, Lipofectamine, both in in vitro and in vivo settings. A novel polymeric gene delivery system, comprising HA-tethered polyethylenimine-grafted chitosan oligosaccharide nanoparticles encapsulating small interfering RNA (siRNA) was reported to considerably reduce the size of endometriotic lesions, with atrophy and degeneration of the ectopic endometrium. Endometriotic rat models exhibited a noteworthy decline in the expression of CD44 expression in the treated group with respect to the control endometriotic population [84]. Gene therapy using cyclic arginine-glycine-aspartic acid (cRGD)-attached fifth-generation PAMAM dendrimers to efficiently deliver siRNAs to spermatogonial stem cells (SSCs) demonstrated great potential for promoting auto-transplantation of SSCs with genetically modified cells as a curative approach for male infertility caused by genetic disorders [85].
6. Treatment of male associated disorders: erectile dysfunction
Reproductive disorders in men include benign prostatic hyperplasia (BPH) or prostate enlargement in general, prostatitis, and male infertility issues such as erectile dysfunction, testosterone deficiency, undescended testicle, varicocele or dilated veins around the testicle, and hydrocele or fluid around the testicle. BPH is often characterized by the increased growth of connective tissue, smooth muscle, and glandular epithelium upon histological visualization. As it progresses, BPH results in compression of the urethral pathways, leading to bladder outlet obstruction, which may subsequently result in lower urinary tract infections, retention, and other undesirable effects. Evidence supports the potential role of diet, lifestyle, and genetic factors, as well as metabolic syndrome and erectile dysfunction, on the incidence of hyperplasia of the prostate [86,87]. Although many choices of treatment are available, ranging from laser ablation methods medications (e.g., α-blockers, 5-α reductase inhibitors), and combination therapy to surgical procedures and medical devices (e.g., Prolieve and Urolift), the treatment options are often accompanied by unwanted effects like retrograde ejaculation, complications during urination, urinary tract infections, erectile dysfunction, and even incontinence (very rarely). In order to circumvent these undesirable events, nanomaterial-based therapies have been developed in recent years. For instance, de Sousa et al. [88] reported that PLGA nanoparticles and clay nano-systems encapsulating babassu oil were prepared for targeted delivery to hyperplastic tissue. Both the developed systems demonstrated an efficient encapsulation of about 90% with excellent bioavailability, highlighting the potential of the system for BPH therapeutics. Another study corroborated the ability of gold nanoparticles to target inflammation and angiogenesis for the development of novel BPH therapies. Adult male rats with experimentally induced BPH that received treatment with gold nanoparticles demonstrated a significant reduction of BPH in a size-dependent fashion with inhibition of inflammation, angiogenesis, and prostatic cell proliferation [89]. This could be a remarkable milestone for the progression of nanomaterial-based therapies towards clinical investigation for bench-to-bedside translation.
Prostatitis is a common urinary tract condition following prostate cancer and BPH that involves therapeutic challenges. Prostatitis accounts for about 8.2% of men, with an estimated expenditure of $84 million on its diagnosis and treatment excluding pharmaceutical aids. Chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) occurs in 90% to 95% of men with prostatitis, of whom 10% to 14% are severely affected. The symptoms of CP/CPPS range from a combination of acute and chronic bacterial prostatitis, CPPS, and none whatsoever (in asymptomatic patients). This medical condition is often diagnosed based on the patient’s history, a physical assessment, urine specimen culture and testing, and pre- and post-prostatic massage. After diagnosis, CP/CPPS is treated with medical agents such as antimicrobials, alpha blockers, and anti-inflammatory drugs. However, the conventional diagnostic and therapeutic approaches are often questioned due to limited availability of evidence supporting their effectiveness [90,91]. The effectiveness of these treatments could be improved by exploiting the targeting potential of the nanoparticles to prostatic tissue. With the targeting potential of nano-systems in mind, disease-relevant antigenic T2 peptide-conjugated biodegradable PLGA nanoparticles modified with poly ethylene-alt-maleic anhydride were prepared by Cao et al. [92] and were demonstrated to be an effective strategy for the treatment of prostatitis in a CP/CPPS-induced mouse model. A similar study by Cheng et al. [93] illustrated the preparation of PLGA nanoparticles coupled with auto-antigen peptide T2 for targeted action, initiation of immune tolerance, and amelioration of disease symptoms in a mouse model with induced CP and CPPS. Both studies presented a potential mitigation approach and an economically feasible tool for treatment of CP/CPPS.
Erectile dysfunction is a common problem impacting millions of men and is defined as the incapability of a man to achieve or maintain sufficient erection for satisfactory sexual performance. Despite the availability of various treatment options, ranging from first-line treatments with oral agents such as sildenafil, tadalafil, and vardenafil to second-line approaches such as intracavernosal injections and intra-urethral therapy, a significant proportion of men discontinue treatment. The discontinuation is directly attributed to the expensive nature of these therapies, as well as the fact that they involve discomfort, lack of spontaneity, prolonged erections, and priapism. However, with the advancements in the pharmaceutical industry, the search for alternatives for the delivery of vasoactive and vasodilatory medications is continuing [94,95]. Topical delivery has proven to be highly effective for the treatment of erectile dysfunction because local action avoids systemic effects and is easy to use. However, the applicability of topical methods is often hindered by the capability of the treatments to cross the barrier due to the penile skin and tunica albuginea, which inhibits the therapeutic efficacy of the drug. This limitation could be surmounted if efficient delivery of the drug is achieved, which is the reason for the success of nanoparticles in this field. A noteworthy example involved treatment and monitoring of erectile dysfunction by human mesenchymal stem cell- labelled super-paramagnetic iron oxide nanoparticles (SPION-MSCs) using MRI. The SPION-MSCs transplanted in the cavernosa of rats exhibited retention for about 4 weeks after cavernous nerve injury and enabled an efficient recovery of erectile dysfunction that could be monitored by MRI in an in vivo setting [96]. A formulation (NanoShuttle) comprising a complex of adipose-derived stem cells and magnetic nanoparticles demonstrated effective in vivo cell tracking in the corpus cavernosum under the effects of a magnetic field for up to 3 days and was shown to be an efficient stem cell therapy for erectile dysfunction in an animal model [97]. The dendrimer-based delivery of vardenafil hydrochloride was demonstrated by Tawfik et al. [98], and sustained release of the drug was found, with a 3.7-fold improvement in bioavailability. Among the pharmaceutical nano-formulations, lipid-based systems have attracted significant attention in the field of reproductive medicine owing to their capability to improve the solubility, bioavailability, and biocompatibility of poorly water-soluble medications. Fahmy’s work illustrated the development of a biocompatible nanoethosome formulation of vardenafil for enhanced permeation via the transdermal route and improved bioavailability. The developed system, which exhibited a 3.05-fold improvement in permeation, consisted of a vardenafil powder-containing film. A two-fold increment in the transdermal bioavailability was found when the topical nanoethosome film was used in comparison to an aqueous suspension of vardenafil. This transdermal delivery system provides a potential method of curing erectile dysfunction and managing impotence [99]. A similar study using nano-transfersomal transdermal films as a mode of delivery of sildenafil citrate reported a significant enhancement in the ex vivo permeation characteristics, controlled release, bioavailability, and absorption of the drug [100]. Another study demonstrated the transdermal delivery of papaverine to the penis using an ultra-flexible liposomal formulation (nano-transferosomes) for the effective diagnosis and treatment of erectile dysfunction. The prepared formulation exhibited remarkable potential as a therapy for male impotency [101]. A considerable improvement in the bioavailability and transdermal delivery of avanafil, a first-line drug for the treatment of erectile dysfunction, was observed when it was delivered in the form of solid lipid nanoparticle-based hydrogel film [102].
7. Nano-based approaches for treatment and diagnosis of male infertility
Infertility is a condition wherein a couple is unable to become pregnant despite being sexually active and avoiding contraception. Male factor infertility can result from a low sperm count, poor sperm quality, or both. According to global statistics, about 15% of couples suffer from infertility-related issues, and in 50% of these couples, the male partner is affected by aberrations in sperm properties, count, vitality, and morphology [103,104]. This fact underscores the need to develop novel methodologies for the early identification of the causes of infertility and approaches for the personalized treatment of infertility. In this aspect, nanotechnology has come to the forefront in recent years, offering better solutions for infertility related issues. One such solution was described in the work of Moridi et al. [105], who found that the deleterious effect of malathion, a common organophosphorus pesticide, on the male reproductive system was remarkably reduced by the application of cerium dioxide nanoparticles (CeNPs). Restoration of testicular changes was observed upon treatment with CeNPs in malathion-exposed male rats, and the nanoparticles exerted a protective effect on sperm count, motility, and viability. Another study utilized the magnetic properties of surface charged Fe3O4 nanoparticles for the development of a method of controlling sperm motility as a novel and simplified approach for the improvement of fertility in infertile males [106]. Rapid and precise detection of semen abnormalities is crucial for diagnosing male infertility and arranging customized care. The size and abundance of samples, however, impose a number of detection constraints [104]. One such solution was proposed by Vidya and Saji [107], wherein a quick screening approach utilizing an environment-friendly, minimally-invasive, and label-free heparin gold nanoparticle-based colorimetric biosensor was developed for the detection of semen protamines as effective biomarkers of male infertility. The nano-biosensor demonstrated noteworthy changes in plasmon absorption spectra upon specific detection of protamines in real semen and serum samples. A similar colorimetric nano-biosensing approach for human semen analysis was proposed by Sun et al. [108]. The nano-biosensor comprised zirconium metal−organic frameworks (Zr-MOFs) coupled with single-stranded DNA-decorated gold nanoparticles (ssDNA-AuNPs), and precise detection was enabled by the possibility that the target proteins in the test sample would hamper the co-precipitate formation of Zr-MOFs and ssDNA-AuNPs. This change manifests as an alteration in the color of the supernatant, offering the potential to accurately identify possible cases of male infertility with utmost simplicity and high sensitivity.
8. Nanomaterials in contraception
Contraception has been a major topic of debate for decades, as promoting birth control and family planning not only benefits individuals’ health and well-being, but also helps facilitate economic growth. The current methods of birth control usually involve administration of hormonal contraceptive medications through the oral, transdermal, intravaginal, and intrauterine routes. Several other approaches such as female sterilization, male condoms, and intrauterine devices (IUDs) have attracted significant attention as potential alternatives to conventional drugs. However, these approaches are faced by challenges such as an increased risk of blood clots or breast cancer associated with the long-term usage of contraceptive pills, the risk of failure of condoms, and the irreversible nature of vasectomies [109,110]. This situation clearly indicates the increasing need for new developments in both male and female contraceptive methods [110]. The past decades have witnessed remarkable improvements in contraceptive methods owing to nanotechnological advancements. Chitosan nanoparticle-based immuno-contraceptive vaccines have exhibited efficient peptide and protein delivery with increased uptake by dendritic cells and retention in the lymph nodes. The vaccines target luteinizing hormone-releasing hormone, thereby increasing the generation of antibodies that hamper reproductive capabilities; thus, this framework offers a potential system of contraception development [112]. Another study used biodegradable polymer-based polyethylene sebacate particulates as effective carriers for the delivery of an HSA peptide-1 vaccine [113]. The improved immunogenic activity of an mCRISP1 DNA contraceptive vaccine was demonstrated by chitosan-DNA nanoparticles that exhibited high effectiveness and safety [114]. Apart from vaccine delivery, photo-thermal therapy using in situ testicular injection of methoxypoly (ethylene glycol)-modified gold nano-rods under near-infrared light in male mice led to male infertility. The high temperature induced by plasmonic nanomaterials resulted in complete destruction of the morphological characteristics of the testes or seminiferous tubules, leading to loss of fertility [115]. Remarkable achievements have been made in combining IUDs with nanomaterials to develop effective and long-term contraceptives. For instance, nano-copper and polymeric substances such as polydimethylsiloxane, silicone rubber, and low-density polyethylene-based nanocomposite have been developed for potential use in IUDs as novel, safe and pragmatic alternatives for contraceptive applications [116-118]. A cocktail-inspired medium-term, reversible male contraceptive strategy was proposed by Bao et al. [119], wherein four layers of nanomaterials were injected sequentially into the vas deferens. The first layer comprised a hydrogel for the formation of a physical barrier to sperm, the second layer contained gold nanoparticles for heating on irradiation, the third layer was made up of ethylenediaminetetraacetic acid (EDTA), which functioned as a system for breaking the hydrogel and exterminating the sperm, and the fourth layer comprised gold nanoparticles. An in vivo study of this approach in a male rat model exhibited inhibition of impregnation of females for more than 2 months. The reversible nature of the approach was confirmed, as near-infrared irradiation led to dissolution of all the layers, allowing the rats to conceive [119]. A biodegradable polymer-based point-of-care micro-needle patch encapsulating contraceptive hormones was developed by Li et al. [120] and demonstrated sustained release for more than a month.
Conclusion
In the modern world, maintaining sustainable health of both men and women is a major challenge due to rapidly evolving lifestyle changes and environmental impact. Several maternal and fetal factors exhibit a strong correlation with adverse outcomes of children and mothers. Increasingly many studies have reported that infertility and reproductive disorders such as endometriosis, polycystic ovary syndrome, and uterine fibroids have a harmful effect on conception, starting from the first events of implantation and lasting until term delivery. These risks have led to the increasing use of ART; however, the use of ART is often accompanied by other deleterious effects that directly or indirectly hamper fertility or pregnancy-related outcomes. In addition to this, the tendency for women to become pregnant at older ages has contributed to an increased risk of gestational complications. Therefore, it has become more important to develop novel strategies for the management of reproductive health. Therapeutic, preventive, and diagnostic approaches in combination with nanotechnology have provided remarkable opportunities for the development of superior strategies in reproductive medicine, with the projected aim of improving the quality of life of both men and women (Table 1). Despite enormous advances, nanotechnological approaches in reproductive health face several shortcomings, such as the limited availability of validation studies of both diagnostic and therapeutic interventions and toxicity issues related to the use of nanomaterials. Most trials involving nanoengineering methods for reproductive health management are still at the nascent stage. There is a sharp dearth of evidence supporting the effective clinical translation of these approaches; therefore, it is highly essential to conduct meticulous research in the future to establish the potential applicability of nanotechnological methodologies for the real-time prevention, treatment, and monitoring of disease. In order to bridge the gap between translational research and product development, a careful choice of qualified partners will be highly essential. Strategic partnerships with the insight and ability to anticipate challenges early on, as well as in-depth understanding of the impact of early processes on later-stage development, will have considerable advantages in overcome the regulatory gauntlet and technical hurdles, thereby facilitating effective clinical translation of these engineered nano-particles for use in reproductive medicine. Nonetheless, several serious concerns linked to the potential risks of the systemic and local toxicity of engineered nanomaterials, the disruption of metabolic function, and potential immune pathophysiological consequences of long-term usage must be addressed. Therefore, the utilization of nanomaterials to treat any form of reproductive ailment must be done after careful compliance with regulatory pharmacological and toxicological guidelines. There is not an iota of doubt that nanotechnology will help to bring about specific, sensible, and cost-effective clinical interventions that are suitable, acceptable, and more personalized in the future.
Acknowledgements
The authors are thankful to the Indian Council of Medical Research (ICMR), Department of Health Research, Ministry of Health and Family Welfare, Government of India, New Delhi, India for funding support to the laboratory of Professor Pradyumna Kumar Mishra.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Author contributions
Conceptualization: PKM. Data curation: RS and NP. Formal analysis: RS, NP, PKM. Funding acquisition: PKM. Methodology: RS, NP, PKM. Project administration: PKM. Visualization: NKL, RSS. Writing--original draft: RS, NP, PKM. Writing--review & editing: NKL, RSS, PKM.