Advantages of the outgrowth model for evaluating the implantation competence of blastocysts
Article information
Abstract
The implantation process is highly complex and difficult to mimic in vitro, and a reliable experimental model of implantation has yet to be established. Many researchers have used embryo transfer (ET) to assess implantation potential; however, ET with pseudopregnant mice requires expert surgical skills and numerous sacrificial animals. To overcome those economic and ethical problems, several researchers have tried to use outgrowth models to evaluate the implantation potential of embryos. Many previous studies, as well as our experiments, have found significant correlations between blastocyst outgrowth in vitro and implantation in utero by ET. This review proposes the blastocyst outgrowth model as a possible alternative to animal experimentation involving ET in utero. In particular, the outgrowth model might be a cost- and time-effective alternative method to ET for evaluating the effectiveness of culture conditions or treatments. An advanced outgrowth model and further culture of outgrowth embryos could provide a subtle research model of peri- and postimplantation development, excluding maternal effects, and thereby could facilitate progress in assisted reproductive technologies. Recently, we found that outgrowth embryos secreted extracellular vesicles containing specific microRNAs. The function of microRNAs from outgrowth embryos should be elucidated in further researches.
Introduction
During mammalian preimplantation development, the fertilized zygote undergoes a continuous series of cleavage steps and a series of morphological changes as it becomes a blastocyst. Prior to attachment and implantation into the maternal uterine endometrium, the blastocyst should hatch from the zona pellucida. Hatched blastocysts are also able to attach onto a culture dish as a form of in vitro implantation through outgrowth [1]. The morphological features of the periimplantation development of mouse embryos to the outgrowth stage are presented in Figure 1. The implantation process is highly complex and difficult to mimic in vitro, and a reliable research model has yet to be established. Many researchers have used embryo transfer (ET) to assess implantation potential, but ET into pseudopregnant mice requires expert experimental skills and numerous sacrificial animals [2,3]. To overcome these economic and ethical problems, several researchers have tried to use outgrowth models to evaluate the implantation potential of embryos. Mammalian embryos, including mouse and human embryos, can attach to an extracellular matrix (ECM) protein-coated culture dish, and develop to peri- and postimplantation stages [4-6].
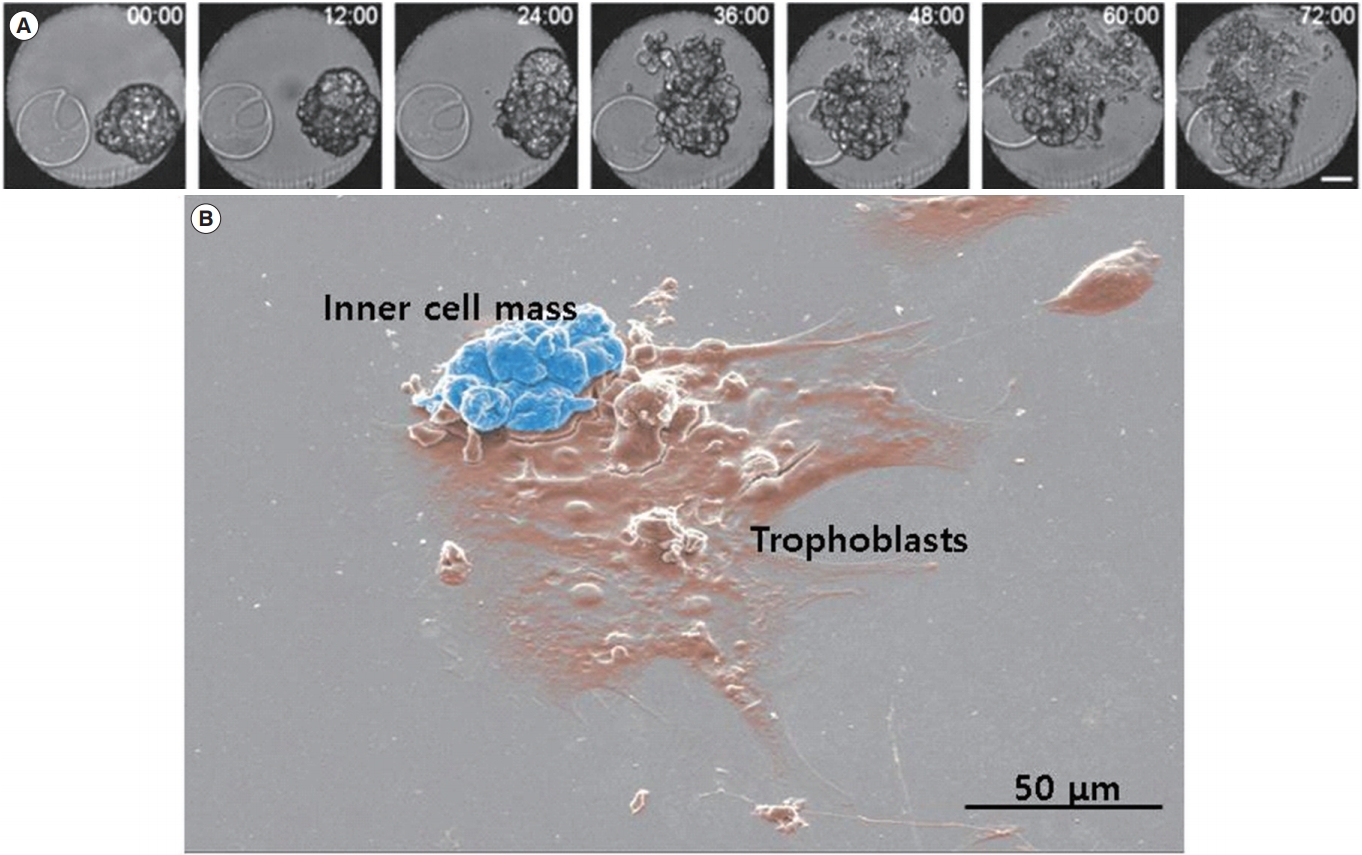
Morphology of hatched blastocyst and the outgrowth stage. (A) Serial images of hatched blastocysts were captured by a time-lapse monitoring system for 3 days. Scale bar, 50 μm. (B) Scanning electron microscopic image of outgrowth embryos. The cluster of the inner cell mass is presented in blue and the spreading-out trophoblasts are presented in brown.
The initial stages of implantation are poorly understood in humans due to ethical limitations on experimentation. Most of our understanding of blastocyst development and early trophoblast differentiation derives from work with animal models, including non-human primates, livestock, and rodents [7]. The location of the implantation site is difficult to find when in utero biochemical approaches are used due to its small diameter within the relatively large uterine lumen. To explore the embryonic side of implantation, in vitro culture of blastocysts has provided a useful experimental model.
The outgrowth model provides an in vitro implantation assay that can be used to characterize the effects of various factors on invasion and proliferation in peri-implantation embryos [8,9]. Culturing beyond the blastocyst stage and assessing outgrowth development provide a more sensitive assay for toxins present in in vitro fertilization (IVF) laboratories than traditional quality control assays such human sperm motility and the mouse two-cell embryo assay [10]. However, a more refined approach than the blastocyst outgrowth model is required to investigate the regulation of signaling and adhesion molecules at the apical surface of mural trophoblast cells before they dissociate and migrate outward [11].
The fibronectin-binding assay is ideal for investigating the onset of adhesion at the apical surface of trophoblast cells, since it is performed rapidly and detection does not require any cellular activity beyond ligand binding. Image analysis is effectively used to quantify the spreading area of trophoblasts and the inner cell mass (ICM). The fibronectin-binding assay provides a useful model for assessing the adhesive activity of integrins on the blastocyst surface during periimplantation development. This model is applicable for a comparative analysis of adhesion throughout development if embryos are treated with various biological molecules and reagents. Some agonists capable of accelerating the rate of blastocyst outgrowth concomitantly shift the onset of fibronectin-binding activity, providing evidence for the physiological relationship between this activity and the adhesion competence of blastocysts [12-15].
In this review, we provide an overview of significant reports regarding historical and functional blastocyst outgrowth models, and suggest that the outgrowth model might be an effective alternative method to ET for analyzing experimental culture conditions or treatments. Furthermore, advanced researches with the blastocyst outgrowth model is proposed to investigate extracellular vesicles (EVs) and microRNAs (miRNAs) as bioactive molecules and biomarkers.
History of research using the outgrowth model of mammalian blastocysts
We searched for the keywords “blastocyst” and “outgrowth” in the PubMed database, and found that 472 papers had been published. The oldest paper was “Chromosomes of the mouse blastocyst following its attachment and outgrowth in vitro” published by Gwatkin and Meckley in 1966 [16]. This outgrowth model of in vitro implantation was developed by culturing blastocysts under various conditions. After breakdown of the spherical morphology of the blastocyst, trophoblast cells can spread out as a monolayer of cells around the base of the embryo concomitantly with ICM (Figure 1). The outgrowth rate is calculated by the identified number of blastocysts. The grading of outgrowth embryos is determined by the size of the ICM, the number of cells, and the area of trophoblasts, which are spreading out. This in vitro experimental model was developed to overcome the complications of in vivo and ex vivo implantation models, and has been used by several laboratories to evaluate the effects of morphological and biochemical factors on the peri-implantation development of blastocysts [17-19].
Significant papers related to blastocysts and outgrowth published through 2000 are summarized in Table 1. Most of the early papers focused on the outgrowth of mouse blastocysts, but they also encompassed such as rat, bovine, swine, field vole, guinea pig, sheep, hamster, and monkey models, and some were even conducted in humans. In addition, studies reported the morphological changes experienced by blastocysts during outgrowth, the effects of serum and serum-derived components on various conditions, and the effects of inhibiting outgrowth using antibiotic components. In particular, the functional role of the Arg-Gly-Asp (RGD) peptide in the process of implantation and outgrowth was thoroughly studied by the Armant group. The RGD sequence of various ECM proteins intervene in their binding to integrins during the cell adhesion and peri-implantation process [20,21]. The antagonistic peptide Gly-Arg-Gly-Asp-Ser-Pro (GRGDSP) inhibits blastocyst outgrowth on various ECM proteins such as fibronectin, vitronectin, type II collagen, and entactin/nidogen [4,22,23]. In the adhesion and invasion of trophoblast cells to the maternal endometrium, the diverse array of adhesive ECM proteins and integrins could provide functional redundancy to increase the likelihood of successful implantation.
Since then, molecular biological methods have been introduced to study the genes that are specifically expressed during outgrowth and their mechanisms of action. Major outgrowth studies from 2000 to the present have focused on evaluating the effects of various amino acids, growth factors, cytokines, and biological and chemical components, and on identifying the mechanisms of action [24-28]. The outgrowth model has also been used in studies to effectively separate and to efficiently produce embryonic stem cells from blastocysts [29-31]. Studies have been conducted to identify and characterize miRNAs and EVs derived from blastocysts and outgrowth embryos in mouse and bovine models, as well as humans. It was shown that the miRNAs and EVs had significant effects on pre- and peri-implantation embryonic development and regulation of the implantation process.
Recent papers have used the mouse blastocyst outgrowth model as an efficient method for assessing the developmental competence of in vitro cultured embryos and peri-implantation viability. Kelley and Gardner [32] published a paper entitled “Individual culture and atmospheric oxygen during culture affect mouse preimplantation embryo metabolism and post-implantation development.” They found that peri-implantation development was not affected by individual culture under 5% oxygen, but under 20% oxygen, individual culture resulted in smaller outgrowths than embryos that had been cultured in groups, indicating they were less viable. We similarly showed that a dynamic oxygen concentration (decreasing from 5% to 2%) had beneficial effects on mouse pre- and peri-implantation development using an outgrowth model [33].
In a knock-out model in mice, an outgrowth assay was applied to evaluate early embryonic lethality and impaired trophoblast function. The blastocysts from dUTPase knock-out mice could not advance to the outgrowth stage [34]. It was also shown that the med20 gene is essential for early embryogenesis and regulates nanog expression in mouse blastocysts and outgrowth embryos [35]. Mcrs1 mutant preimplantation embryos exhibited normal morphology at the blastocyst stage, but did not progress to the gastrulation stage, resulting in embryonic loss. Outgrowth assays revealed that the mutant blastocysts did not form a typical ICM colony, the source of embryonic progeny [36].
Developmental competence of mouse blastocysts in outgrowth in vitro and implantation in utero
The in vitro blastocyst outgrowth model mimics implantation in the uterus in vivo and enables experimental studies on implantation events and mechanisms. This model has also revealed the relationship between metabolism—based on morphokinetic findings of preimplantation embryos—and implantation potential, and has been used as an alternative tool to study trophoblastic invasion and motility [37-40].
Lane and Gardner [41] reported quantitative findings on various parameters of embryo development in vitro, which were correlated with fetal development after the transfer of cultured blastocysts. Morphology, as assessed by blastocyst formation and hatching, was not correlated with subsequent developmental competence. In contrast, significant positive correlations were found between the number of blastocyst cells and the number of ICM cells and subsequent fetal development. Similarly, the attachment ability of blastocysts and ICM outgrowth were also positively correlated with fetal development. Glycolytic activity of blastocysts appeared to be negatively correlated with fetal development after transfer.
In our previous studies, we confirmed a correlation in the developmental competence of preimplantation embryos between blastocyst outgrowth in vitro and implantation in utero [42,43]. We established a novel coculture system with outgrowth and preimplantation embryos, and investigated how this coculture system improved preimplantation and peri-implantation embryonic development both in vitro and in utero. In the coculture system, it was observed that outgrowth embryos secreted EVs by time-lapse monitoring and scanning electron microscopy. Coculture with outgrowth embryos significantly increased the percentage of outgrowth in vitro, which was correlated with implantation rates in utero after ET [42].
A time-lapse monitoring system has been applied to select transferrable embryos and to predict the developmental competence of preimplantation embryos in human IVF-ET programs. We studied blastocyst development and implantation potential in utero based on the third cleavage and compaction times using a mouse model [43]. Our results provided evidence that analyzing morphokinetics by a time-lapse monitoring system may improve the efficacy of selection of transferrable embryos with high implantation potential in human IVF-ET programs. In that study, we found that the times of the third cleavage to the four-cell stage and compaction to the morula stage were useful morphokinetic parameters for predicting the potential of mouse preimplantation embryos to develop into outgrowth in vitro and implantation in utero [43].
Future research using the outgrowth model
Recently, many studies have investigated the role of EVs in reproductive events, including oogenesis, embryo development and death, oviduct–embryo crosstalk, IVF and others [44-47]. Saadeldin et al. [48] demonstrated that porcine embryos secreted EVs in culture medium that contained mRNA sequences of pluripotency genes. They suggested that EVs carrying embryotrophic signals could act as mediators to improve preimplantation development.
It was proposed that EVs from pre- and peri-implantation embryos might also communicate with maternal immunological factors by presenting and processing antigens [49,50]. EVs were found to contain major histocompatibility complex molecules, cytokines, and miRNAs. Of particular note, HLA-G-positive EVs from healthy term pregnant women’s plasma have been found to bind with T lymphocytes and regulate peripheral T lymphocyte STAT3 phosphorylation and activation [51]. As a way to induce a favorable immune system response, EVs from embryos bind to CD8+ and increase the number of interleukin-10+ cells among peripheral CD8+ cells. By producing interleukin 10, an anti-inflammatory cytokine, CD8+ T lymphocytes might alleviate the antigen-induced inflammatory responses. Using immunoelectron microscopy, it was observed that progesterone-induced blocking factor containing EVs from embryos communicated with immune cells [52].
We also isolated and identified EVs and miRNAs from blastocysts and outgrowth embryos [53]. The EVs from outgrowth embryo-conditioned media have rounded membrane structures that range in diameter from 20 to 225 nm. Incubation with EVs improved preimplantation embryonic development by increasing cell proliferation and decreasing apoptosis in blastocysts. Moreover, the implantation rate following ET was significantly higher in EV-supplemented embryos than in the control group [54]. This finding suggests that since EVs contain bioactive molecules from outgrowth embryos, they could enhance embryonic developmental competence and even implantation potential in mice. Giacomini et al. [54] showed that the EVs secreted from cultured embryos not only seemed to improve developmental competence by exchanging embryotrophic messages, but could also send bioactive molecules to the maternal endometrium, supporting a favorable endometrial environment for implantation.
EVs play a role in cell-to-cell communications because their cargo contains potentially bioactive molecules relevant for physiological responses and pathological conditions. Since miRNAs in EVs have been well characterized, bioinformatic research into the miRNA expression profiles of EVs will be helpful to explore their physiological functions and pathological biomarkers. The numerous suggested biomarkers that could be used to predict the developmental competence of embryos include miRNAs secreted from in vitro cultured pre- and peri-implantation embryos. In particular, the miRNA profile has been reported to show correlations with fertilization using IVF and intracytoplasmic sperm injection, chromosomal abnormalities of embryos, and pregnancy outcomes [55,56].
Recently, we performed the first profiling study on miRNAs of EVs from blastocysts, non-outgrowth embryos, and outgrowth embryos in mice [57]. A total of 3,163 miRNAs were detected in the blastocysts and outgrowth embryos, and the miRNA expression profiles were significantly different between non-outgrowth and outgrowth embryos. Ten miRNAs (let-7b, miR-23a, miR-27a, miR-92a, miR-183, miR200c, miR-291a, miR-425, miR-429 and miR-652) were identified as significant differentially expressed miRNAs in outgrowth embryos by microarray and in silico analysis. The expression of these miRNAs markedly changed during preimplantation embryo development. In particular, let-7b-5p, miR-200c-3p and miR-23a-3p were significantly upregulated in outgrowth embryos compared with blastocysts and non-outgrowth blastocysts [57]. This study suggested that differentially expressed miRNAs in outgrowth embryos compared with blastocysts and non-outgrowth embryos could be involved in embryo attachment and interactions between the embryo proper and maternal endometrium during the implantation process. We conclude that EVs secreted from outgrowth embryos could improve the developmental competence of in vitro cultured mouse preimplantation embryos. Findings of specific embryotrophic factors and miRNAs from outgrowth embryos might be valuable for advancing reproductive technologies in the future.
Conclusion
Blastocyst outgrowth has proven to be a useful and efficient model for investigating the adhesion and invasion of trophoblast cells during the implantation process of mammalian embryos. The developmental program of blastocysts and trophoblast cells is regulated by transcripts or proteins produced at an earlier stage by preimplantation embryos. Post-translational modifications of specific proteins, rather than biosynthesis, regulate the onset of trophoblast differentiation in preparation for blastocyst implantation and outgrowth in vitro. The regulation of blastocyst development and outgrowth adhesion independently of gene activation enables cells to adapt to alterations in the conditions of in vitro culture. It was recently suggested that spent embryo culture medium metabolites might be related to the ability of blastocysts to undergo outgrowth [58]. In a metabolite analysis of embryo culture medium, non-outgrowth blastocysts that lacked the ability to adhere in vitro had increased requirements for lactate and pyruvate, and showed a significant reduction of the pyruvate-alanine ratio. Thus, it was proposed that the aforementioned metabolites from the spent medium should be further analyzed using proper experimental models to substantiate their potential as biomarkers for predicting the implantation competence of embryos in clinical IVF-ET programs [58].
Supplementation of specific molecules in culture media could improve pre- and peri-implantation development. Treatment with the well-known appetite hormone leptin increased the blastocyst outgrowth rate of ICM during embryonic stem cell derivation [59]. This year, Truong and Gardner [60] reported that addition of a combination of three antioxidants (acetyl-L-carnitine, N-acetyl-L-cysteine, and α-lipoic acid) to vitrification and warming solutions resulted in a significant increase in the outgrowth area, which was correlated with higher fetal weight, crown rump length, and limb development after ET than were found in embryos that did not receive antioxidant treatment.
In many previous studies and our experiments, a significant correlation was found between blastocyst implantation in vitro by an outgrowth assay and implantation in utero by ET. This review suggests that the outgrowth blastocyst assay might be an alternative to animal experimentation involving ET in utero. Using the outgrowth assay could reduce the number of sacrificed animals needed to assess the developmental competence of peri- and postimplantation embryos [61]. Therefore, we suggest that the outgrowth model might be a cost- and time-effective alternative method to ET for evaluating effective culture conditions or treatments. To analyze placental and fetal development in utero, the ET approach will still be required. However, implementing screening tests using the outgrowth model described herein will effectively reduce time, cost, and the number of surgical procedures and sacrifices in animals.
Recently, we found that outgrowth embryos secreted EVs containing specific miRNAs. The function of miRNAs from outgrowth embryos should be elucidated in further research. An advanced outgrowth model and further culture of outgrowth embryos could provide a subtle and valuable research model of peri- and postimplantation development, with implications for progress in assisted reproductive technologies.
Notes
Conflict of interest
Jin Hyun Jun has been an associate editor of Journal of Clinical and Experimental Reproductive Medicine since 2018; however, he was not involved in the peer reviewer selection, evaluation, or decision process of this article. No other potential conflicts of interest relevant to this article were reported.
Author contributions
Conceptualization: JHJ. Data curation: JK, JL. Formal analysis: JHJ, JK. Methodology: JK, JL. Visualization: JHJ, JK. Writing–original draft: JK. Writing–review & editing: JHJ, JL.

